Region:Middle East
Author(s):Dev
Product Code:KRAB2349
Pages:96
Published On:January 2026
By Test Type:The test type segmentation includes various methods used for noninvasive prenatal testing. The dominant subsegment is Cell-free DNA (cfDNA) testing, which is preferred due to its high accuracy and reliability in detecting common fetal aneuploidies such as trisomy 21, 18, and 13, often with sensitivities and specificities above those of traditional serum screening. Ultrasound-based screening, Biochemical screening, and Combined first-trimester screening remain significant as part of standard prenatal care pathways, and are frequently used in conjunction with cfDNA testing for comprehensive risk assessment, especially in public health settings. The increasing preference for cfDNA testing is driven by its noninvasive nature, earlier testing window, improving reimbursement in parts of the Middle East and Africa, and growing awareness among expectant parents and clinicians regarding the clinical utility of advanced prenatal genetic screening.

By Indication:The indication segmentation focuses on the specific genetic conditions that noninvasive prenatal testing aims to detect. Trisomy 21 (Down syndrome) is the leading subsegment, as it is the most common chromosomal abnormality detected through prenatal screening programs worldwide and is a key clinical focus of cfDNA-based NIPT panels. Other significant indications include Trisomy 18 and Trisomy 13, which are routinely incorporated in standard NIPT offerings, Sex chromosome aneuploidies such as Turner and Klinefelter syndromes, and selected Microdeletions and other genetic disorders where extended panels are available. The high clinical relevance of Down syndrome, combined with rising awareness in the Middle East regarding the benefits of early risk identification and counseling, contributes to the dominance of this subsegment in the overall indication mix.

The Qatar Noninvasive Prenatal Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Natera, Inc., Illumina, Inc., Roche Diagnostics, Laboratory Corporation of America Holdings (Labcorp), Quest Diagnostics Incorporated, PerkinElmer, Inc., Eurofins Scientific SE, Myriad Genetics, Inc., BGI Genomics Co., Ltd., MedGenome Labs Ltd., Agilent Technologies, Inc., Siemens Healthineers AG, Thermo Fisher Scientific Inc., and local/regional prenatal genetic testing laboratories in Qatar contribute to innovation, geographic expansion, and service delivery in this space.
The future of the NIPT market in Qatar appears promising, driven by ongoing advancements in technology and increasing healthcare investments. As the government continues to enhance healthcare infrastructure, more facilities are expected to offer NIPT services, improving accessibility. Additionally, the integration of artificial intelligence in testing processes is anticipated to further enhance accuracy and efficiency, making NIPT more appealing to expectant mothers. These trends indicate a robust growth trajectory for the market in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Test Type | Cell-free DNA (cfDNA) testing Ultrasound-based screening Biochemical screening Combined first-trimester screening |
| By Indication | Trisomy 21 (Down syndrome) Trisomy 18 & Trisomy 13 Sex chromosome aneuploidies Microdeletions and other genetic disorders |
| By Gestational Age | –12 weeks –24 weeks –36 weeks |
| By Risk Category | High?risk pregnancies Average / low?risk pregnancies |
| By End-User | Hospitals Diagnostic laboratories Specialized fertility & prenatal clinics Others |
| By Technology | Next-generation sequencing (NGS) PCR-based methods Microarray and other technologies |
| By Geographic Distribution | Doha & Al Rayyan Other municipalities |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Obstetricians and Gynecologists | 60 | Healthcare Providers, Specialists in Prenatal Care |
| Expectant Parents | 120 | Parents-to-be, Couples considering NIPT |
| Laboratory Technicians | 40 | Technical Staff, Laboratory Managers |
| Healthcare Policy Makers | 40 | Government Officials, Health Administrators |
| Insurance Providers | 50 | Insurance Analysts, Policy Underwriters |
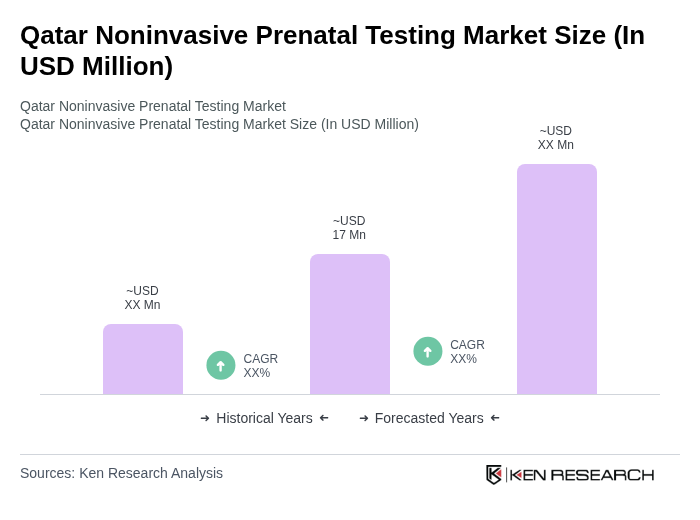
The Qatar Noninvasive Prenatal Testing market is valued at approximately USD 17 million, reflecting a significant growth trend driven by increased awareness of prenatal health and advancements in testing technologies.