Region:Middle East
Author(s):Rebecca
Product Code:KRAD2762
Pages:97
Published On:November 2025
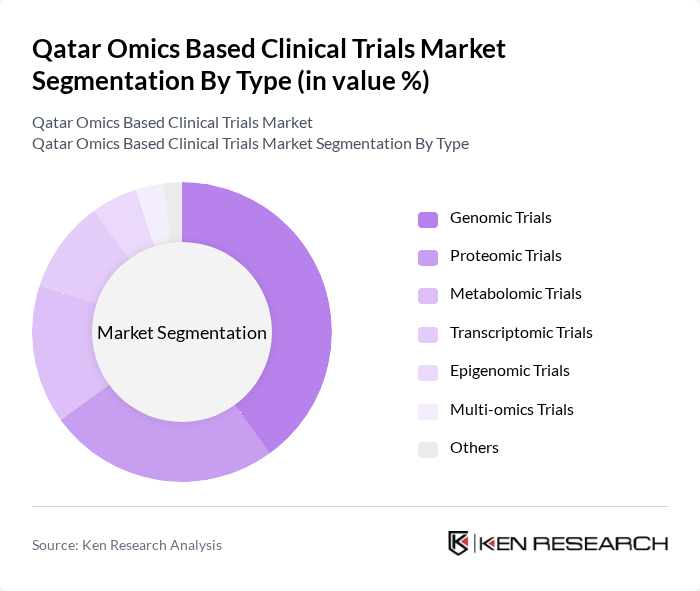
By Type:The market is segmented into various types of omics-based trials, including genomic, proteomic, metabolomic, transcriptomic, epigenomic, multi-omics, and others. Genomic trials are currently leading the market due to the increasing focus on personalized medicine and the ability to tailor treatments based on genetic profiles. The demand for genomic data in clinical trials is driven by advancements in sequencing technologies and a growing understanding of genetic factors in disease progression.
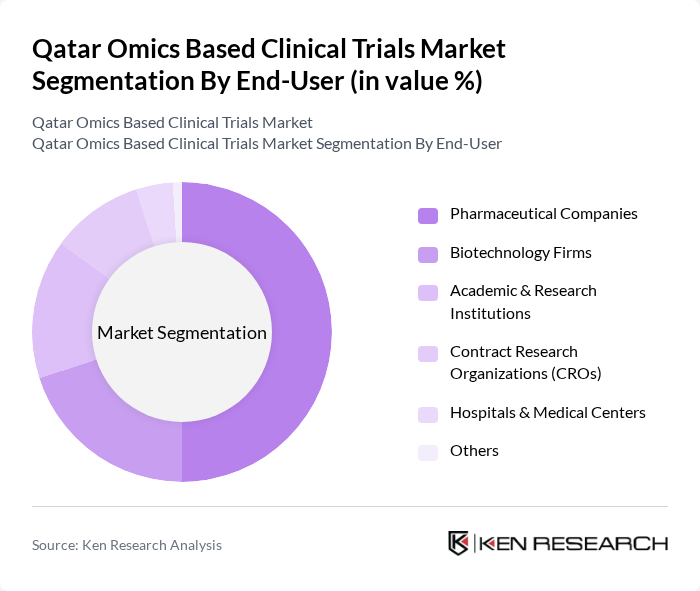
By End-User:The end-user segmentation includes pharmaceutical companies, biotechnology firms, academic and research institutions, contract research organizations (CROs), hospitals and medical centers, and others. Pharmaceutical companies are the dominant end-users, as they are increasingly leveraging omics technologies to enhance drug discovery and development processes. The integration of omics data allows these companies to identify potential drug targets and biomarkers, significantly improving the efficiency of clinical trials.
The Qatar Omics Based Clinical Trials Market is characterized by a dynamic mix of regional and international players. Leading participants such as Qatar Biomedical Research Institute (QBRI), Hamad Medical Corporation (HMC), Qatar University, Sidra Medicine, Qatar Foundation, Gulf Pharmaceutical Industries (Julphar), Doha Clinic Hospital, Qatar Clinical Research Center, Al Ahli Hospital, Dallah Hospital, Al Emadi Hospital, Weill Cornell Medicine – Qatar, Qatar Ministry of Public Health, Qatar National Research Fund (QNRF), Qatar Science and Technology Park (QSTP) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Qatar omics-based clinical trials market appears promising, driven by technological advancements and increased government investment. As genomic technologies continue to evolve, the integration of artificial intelligence and machine learning will enhance trial efficiency and data analysis. Furthermore, the shift towards decentralized clinical trials is expected to improve patient recruitment and retention, ultimately leading to more successful outcomes. These trends indicate a robust growth trajectory for the market, fostering innovation and collaboration within the healthcare sector.
| Segment | Sub-Segments |
|---|---|
| By Type | Genomic Trials Proteomic Trials Metabolomic Trials Transcriptomic Trials Epigenomic Trials Multi-omics Trials Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic & Research Institutions Contract Research Organizations (CROs) Hospitals & Medical Centers Others |
| By Therapeutic Area | Oncology Cardiovascular Diseases Neurological Disorders Infectious Diseases Rare Genetic Disorders Others |
| By Phase of Clinical Trials | Phase I Phase II Phase III Phase IV Observational Studies Others |
| By Region | Doha Al Rayyan Umm Salal Al Wakrah Al Khor Others |
| By Funding Source | Government Grants Private Investments Public-Private Partnerships Venture Capital International Funding Agencies Others |
| By Data Management Approach | Centralized Data Management Decentralized Data Management Hybrid Data Management Cloud-Based Data Management Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Research Organizations (CROs) | 60 | Clinical Trial Managers, Research Scientists |
| Biotechnology Firms | 50 | Product Development Leads, Regulatory Affairs Specialists |
| Healthcare Providers | 40 | Oncologists, Genetic Counselors |
| Regulatory Bodies | 40 | Policy Makers, Compliance Officers |
| Academic Institutions | 45 | Research Professors, Graduate Students in Biomedical Fields |
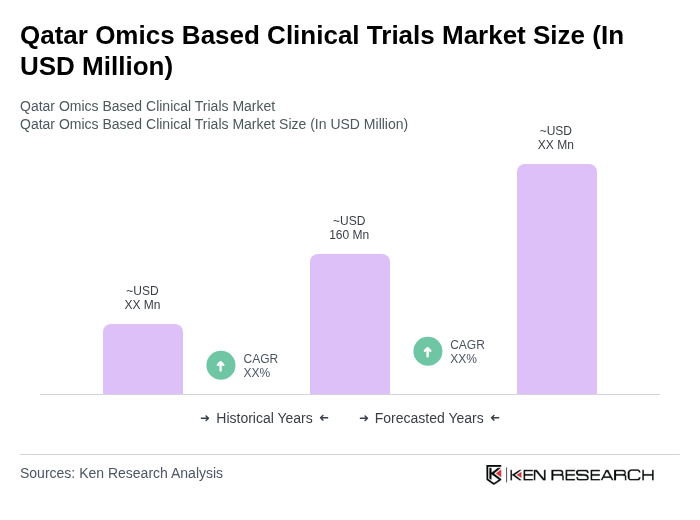
The Qatar Omics Based Clinical Trials Market is valued at approximately USD 160 million, reflecting significant growth driven by advancements in personalized medicine, biotechnology investments, and the demand for innovative therapeutic solutions.