Region:Middle East
Author(s):Shubham
Product Code:KRAD1879
Pages:97
Published On:December 2025
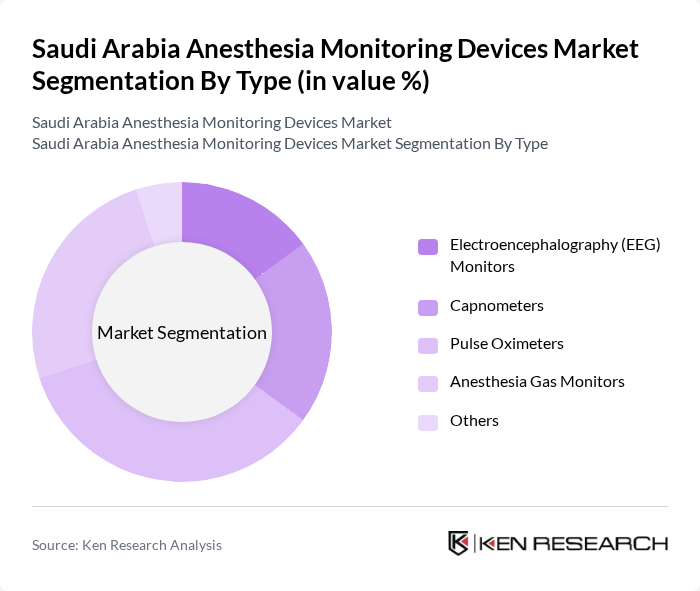
By Type:The anesthesia monitoring devices market is segmented into various types, including Electroencephalography (EEG) Monitors, Capnometers, Pulse Oximeters, Anesthesia Gas Monitors, and Others. Among these, Pulse Oximeters are dominating the market due to their widespread use in hospitals and surgical centers for continuous monitoring of oxygen saturation levels in patients. The increasing prevalence of respiratory diseases and the need for real-time monitoring during surgeries further enhance the demand for these devices.
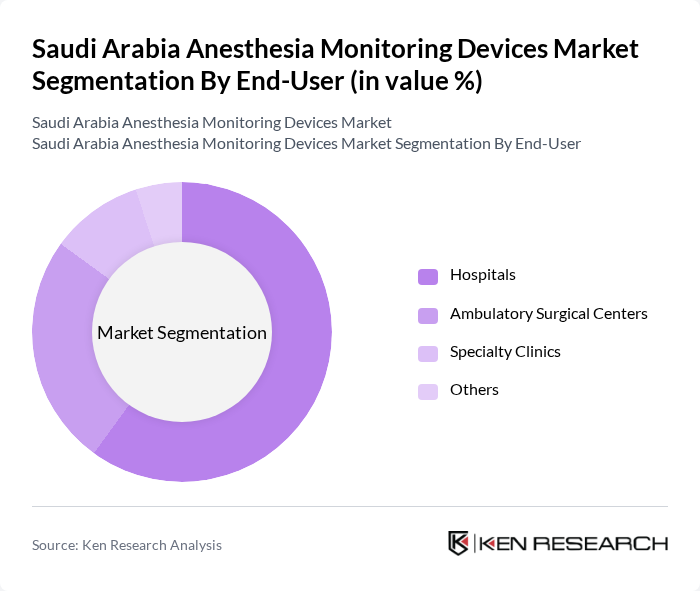
By End-User:The market is segmented by end-users, including Hospitals, Ambulatory Surgical Centers, Specialty Clinics, and Others. Hospitals are the leading end-user segment, driven by the increasing number of surgical procedures and the need for advanced monitoring systems to ensure patient safety. The growing trend of outpatient surgeries in Ambulatory Surgical Centers is also contributing to the market's expansion.
The Saudi Arabia Anesthesia Monitoring Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as GE Healthcare, Philips Healthcare, Drägerwerk AG, Mindray Medical International Limited, Siemens Healthineers, Masimo Corporation, Nihon Kohden Corporation, Baxter International Inc., Covidien (Medtronic), Spacelabs Healthcare, Stryker Corporation, Welch Allyn (Hillrom), Intersurgical, Ecolab, and Cardinal Health contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Saudi Arabia anesthesia monitoring devices market appears promising, driven by technological advancements and increased healthcare investments. Emerging technologies, such as AI integration in monitoring systems, are expected to enhance patient safety and operational efficiency. Additionally, the expansion of telemedicine services, evidenced by over 2 million consultations handled by Seha Virtual Hospital in future, indicates a growing trend towards remote monitoring solutions, further supporting the market's growth trajectory.
| Segment | Sub-Segments |
|---|---|
| By Type | Electroencephalography (EEG) Monitors Capnometers Pulse Oximeters Anesthesia Gas Monitors Others |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Clinics Others |
| By Application | General Surgery Cardiac Surgery Orthopedic Surgery Others |
| By Technology | Digital Monitors Analog Monitors Wireless Monitors Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Policy Support | Government Subsidies Tax Incentives Regulatory Support Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Anesthesia Departments | 100 | Anesthesiologists, Nurse Anesthetists |
| Medical Device Distributors | 75 | Sales Managers, Product Specialists |
| Healthcare Procurement Offices | 80 | Procurement Officers, Supply Chain Managers |
| Regulatory Bodies | 50 | Policy Makers, Compliance Officers |
| Clinical Research Organizations | 60 | Clinical Researchers, Medical Advisors |
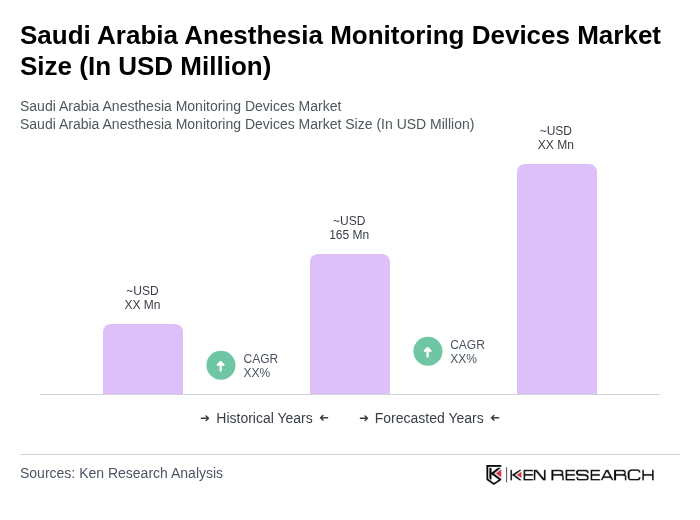
The Saudi Arabia Anesthesia Monitoring Devices Market is valued at approximately USD 165 million. This valuation reflects the increasing demand for advanced monitoring systems driven by a rise in surgical procedures and a focus on patient safety.