Region:Middle East
Author(s):Dev
Product Code:KRAA3754
Pages:86
Published On:January 2026
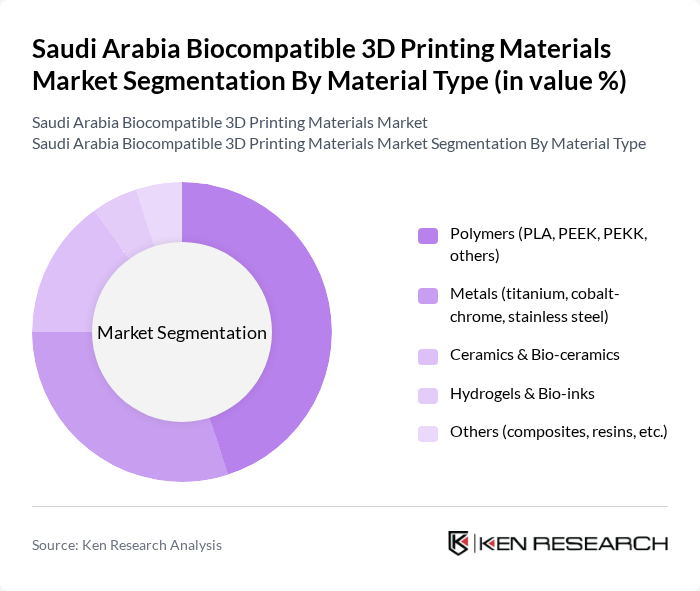
By Material Type:The biocompatible 3D printing materials market is segmented into various material types, including polymers, metals, ceramics, hydrogels, and others. Among these, polymers, particularly PLA and PEEK, dominate the market due to their versatility, ease of use, and favorable biocompatibility profiles for medical models, surgical guides, dental splints, and certain implantable components. Metals, especially titanium and cobalt-chrome alloys, are also significant due to their strength, fatigue resistance, and long-term stability in orthopedic, spinal, and dental implant applications. The demand for ceramics and bio-ceramics is growing, driven by their use in dental crowns, bridges, maxillofacial reconstructions, and bone graft substitutes, while hydrogels and bio-inks are increasingly used in research settings for tissue engineering, regenerative medicine, and drug-delivery constructs.
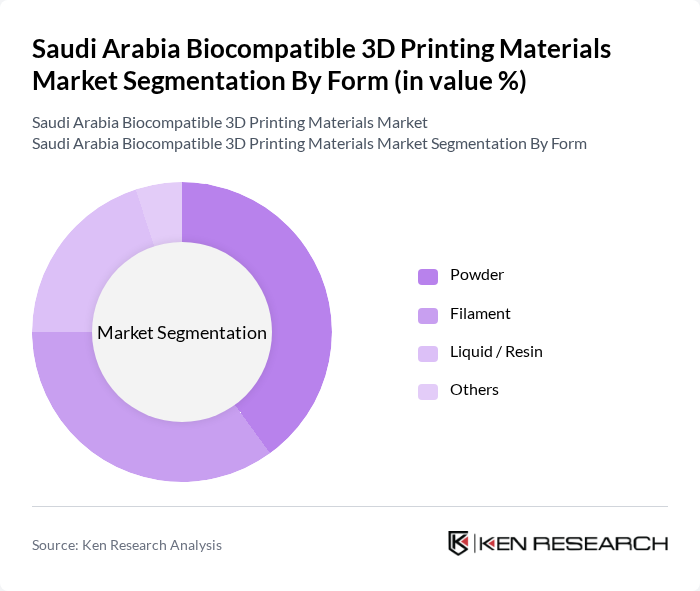
By Form:The market is also segmented by the form of materials used in 3D printing, including powders, filaments, liquids/resins, and others. The powder form is gaining traction due to its compatibility with various 3D printing technologies such as selective laser melting (SLM), selective laser sintering (SLS), and electron beam melting (EBM), particularly in metal and ceramic medical applications. Filaments are widely used in polymer-based 3D printing, especially fused filament fabrication (FFF/FDM) systems adopted by hospitals, universities, and dental labs for anatomical models, guides, and prosthetic components. Liquid/resin forms are essential for technologies like stereolithography (SLA) and digital light processing (DLP), which are extensively used for high-precision dental models, aligner molds, surgical guides, and hearing-aid shells. The choice of form is influenced by the specific requirements of the application, regulatory classification of the end-use device, surface finish needs, and the desired mechanical and biological properties of the final product.
The Saudi Arabia Biocompatible 3D Printing Materials Market is characterized by a dynamic mix of regional and international players. Leading participants such as Stratasys Ltd., 3D Systems Corporation, Materialise NV, EOS GmbH, SLM Solutions Group AG, Renishaw plc, HP Inc., Formlabs Inc., Ultimaker B.V. (UltiMaker), BASF SE, Covestro AG, Arkema S.A., SABIC (Saudi Basic Industries Corporation), Evonik Industries AG, Mitsubishi Chemical Group Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the biocompatible 3D printing materials market in Saudi Arabia appears promising, driven by technological advancements and increasing healthcare demands. As the government continues to invest in healthcare infrastructure, the integration of 3D printing in medical applications is expected to expand. Additionally, the focus on personalized medicine and sustainable practices will likely shape the market landscape, encouraging the development of innovative materials and processes that meet evolving healthcare needs while enhancing patient care.
| Segment | Sub-Segments |
|---|---|
| By Material Type | Polymers (PLA, PEEK, PEKK, others) Metals (titanium, cobalt-chrome, stainless steel) Ceramics & Bio-ceramics Hydrogels & Bio-inks Others (composites, resins, etc.) |
| By Form | Powder Filament Liquid / Resin Others |
| By Application | Implants & Prostheses Surgical Guides & Instruments Dental Applications Tissue Engineering & Regenerative Medicine Drug Delivery & Personalized Medicine Anatomical Models & Prototyping Others |
| By End-Use Sector | Hospitals & Specialized Clinics Dental Labs & Dental Clinics Medical Device & Implant Manufacturers Research & Academic Institutions Contract Manufacturing & Service Bureaus Others |
| By 3D Printing Technology | Stereolithography (SLA) & Digital Light Processing (DLP) Fused Deposition Modeling (FDM) / Fused Filament Fabrication (FFF) Selective Laser Sintering (SLS) Powder Bed Fusion (including DMLS/SLM) Material Jetting & Binder Jetting Others |
| By Biocompatibility & Regulatory Class | Short-term Contact (Class I / IIa) Long-term & Implantable (Class IIb / III) Sterilizable vs Non-sterilizable Materials Certified vs Non-certified Medical Grades |
| By Distribution Channel | Direct Sales (OEM to End User) Authorized Distributors Online & E-commerce Platforms Value-added Resellers & Integrators Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers Using 3D Printing | 120 | Surgeons, Medical Device Developers |
| Manufacturers of Biocompatible Materials | 90 | Product Managers, R&D Directors |
| Regulatory Bodies and Standards Organizations | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Academic Institutions Conducting Research | 70 | Researchers, Professors in Biomedical Engineering |
| End-users of 3D Printed Medical Devices | 80 | Patients, Healthcare Administrators |
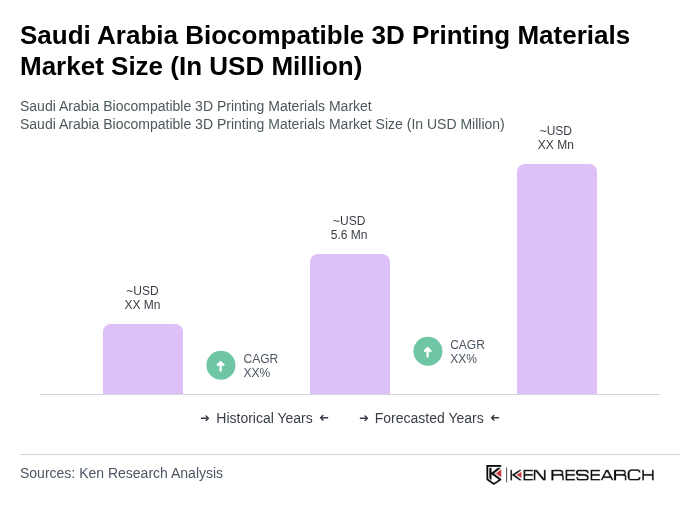
The Saudi Arabia Biocompatible 3D Printing Materials Market is valued at approximately USD 5.6 million, reflecting growth driven by advancements in medical technology and increasing demand for personalized healthcare solutions.