Region:Middle East
Author(s):Geetanshi
Product Code:KRAD1219
Pages:99
Published On:November 2025
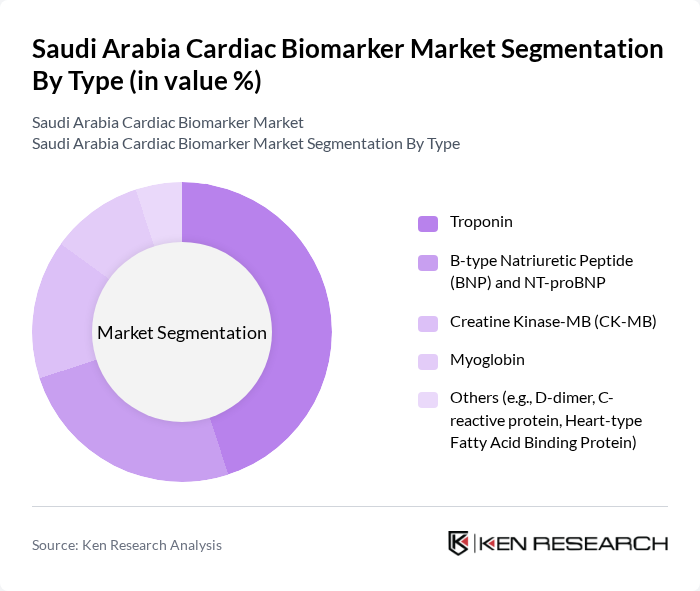
By Type:The market is segmented into various types of cardiac biomarkers, including Troponin, B-type Natriuretic Peptide (BNP) and NT-proBNP, Creatine Kinase-MB (CK-MB), Myoglobin, and Others (e.g., D-dimer, C-reactive protein, Heart-type Fatty Acid Binding Protein). Among these,Troponinis the leading sub-segment due to its high specificity and sensitivity in diagnosing myocardial infarction, making it the preferred choice for clinicians. The increasing incidence of heart diseases and the need for accurate, rapid diagnostic tools further bolster the demand for Troponin tests. Point-of-care testing and integration with digital health platforms are enhancing accessibility and clinical decision-making for cardiac events .
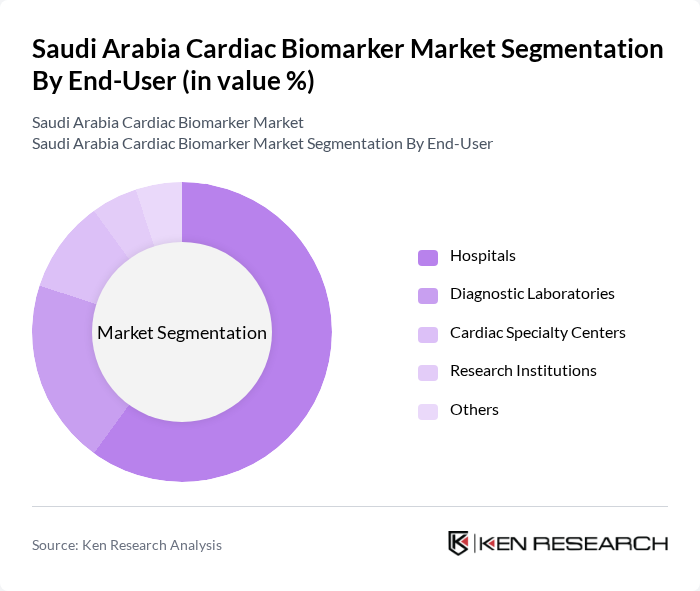
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Cardiac Specialty Centers, Research Institutions, and Others.Hospitalsare the dominant end-user segment, driven by the increasing number of cardiac patients requiring immediate diagnostic services. The integration of advanced diagnostic technologies and multidisciplinary heart teams in hospitals enhances the efficiency of cardiac care, leading to a higher demand for cardiac biomarkers. Government funding for tertiary hospitals and the expansion of cath-lab infrastructure further reinforce hospital dominance in this segment .
The Saudi Arabia Cardiac Biomarker Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, Beckman Coulter, Ortho Clinical Diagnostics, bioMérieux, Thermo Fisher Scientific, QuidelOrtho Corporation, PerkinElmer, Fujirebio, Sysmex Corporation, Hologic, Inc., DiaSorin S.p.A., Shenzhen Mindray Bio-Medical Electronics Co., Ltd., ACON Laboratories, Inc., Randox Laboratories Ltd., Alere Inc. (now part of Abbott), Werfen (Instrumentation Laboratory), Tosoh Corporation, Saudi Diagnostics Holding Company (SDH) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the cardiac biomarker market in Saudi Arabia appears promising, driven by technological advancements and increased healthcare investments. The integration of artificial intelligence in diagnostics is expected to enhance the accuracy and efficiency of biomarker testing. Additionally, the growing emphasis on preventive healthcare will likely lead to increased screening and early diagnosis initiatives, ultimately improving patient outcomes and reducing the burden of cardiovascular diseases across the nation.
| Segment | Sub-Segments |
|---|---|
| By Type | Troponin B-type Natriuretic Peptide (BNP) and NT-proBNP Creatine Kinase-MB (CK-MB) Myoglobin Others (e.g., D-dimer, C-reactive protein, Heart-type Fatty Acid Binding Protein) |
| By End-User | Hospitals Diagnostic Laboratories Cardiac Specialty Centers Research Institutions Others |
| By Application | Acute Coronary Syndrome (ACS) Heart Failure Myocardial Infarction Congestive Heart Failure Others (e.g., Risk Stratification, Prognosis) |
| By Distribution Channel | Direct Sales Online Sales Distributors Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Technology | Immunoassays Mass Spectrometry PCR-based Techniques Point-of-Care Testing Others (e.g., Chemiluminescence, ELISA) |
| By Pricing Model | Premium Pricing Competitive Pricing Value-based Pricing Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiologist Insights | 60 | Cardiologists, Heart Specialists |
| Hospital Procurement Managers | 50 | Procurement Managers, Supply Chain Managers |
| Laboratory Technicians | 40 | Lab Technicians, Biomedical Engineers |
| Patient Awareness Surveys | 100 | Cardiac Patients, General Public |
| Healthcare Policy Makers | 40 | Health Administrators, Policy Analysts |
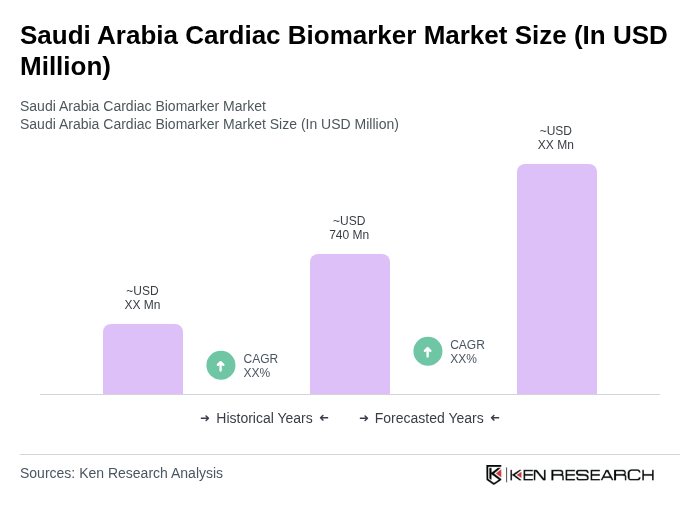
The Saudi Arabia Cardiac Biomarker Market is valued at approximately USD 740 million, driven by the increasing prevalence of cardiovascular diseases and advancements in diagnostic technologies, alongside government investments in healthcare infrastructure.