Region:Middle East
Author(s):Rebecca
Product Code:KRAD7351
Pages:86
Published On:December 2025
By Treatment Type:
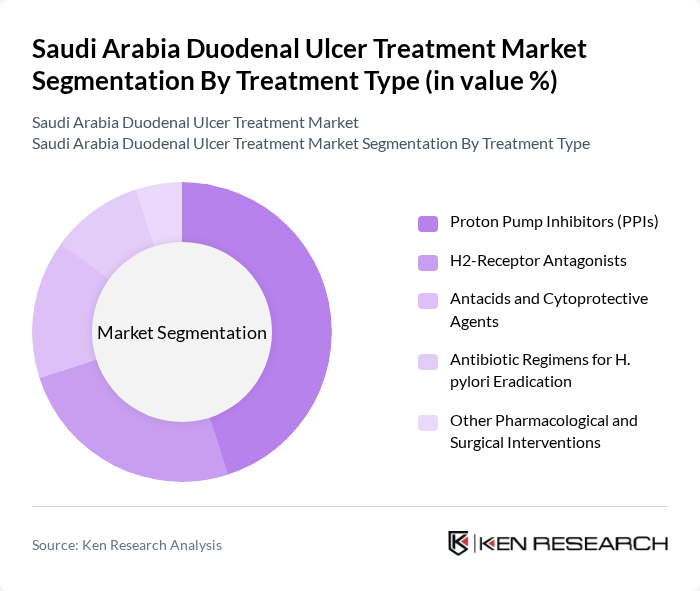
The treatment type segmentation includes various pharmacological and surgical interventions. The dominant subsegment is Proton Pump Inhibitors (PPIs), which are widely prescribed as first-line therapy in duodenal and broader peptic ulcer disease due to their effectiveness in suppressing gastric acid secretion and promoting mucosal healing. H2-Receptor Antagonists also hold a significant share, but their usage is gradually declining as PPIs have become the preferred choice in most clinical guidelines. Antibiotic regimens for H. pylori eradication, typically in combination with PPIs and other agents, are crucial for treating ulcers caused by this bacterium and for reducing recurrence rates. Antacids and Cytoprotective Agents, including agents such as sucralfate and prostaglandin analogues, are used mainly for symptomatic relief and mucosal protection, often as adjuncts rather than stand-alone curative therapies. Other pharmacological and surgical interventions, including endoscopic hemostasis and surgical procedures reserved for complicated or refractory cases, are less common but still play a role in comprehensive treatment plans, particularly in bleeding or perforated ulcers.
By Route of Administration:
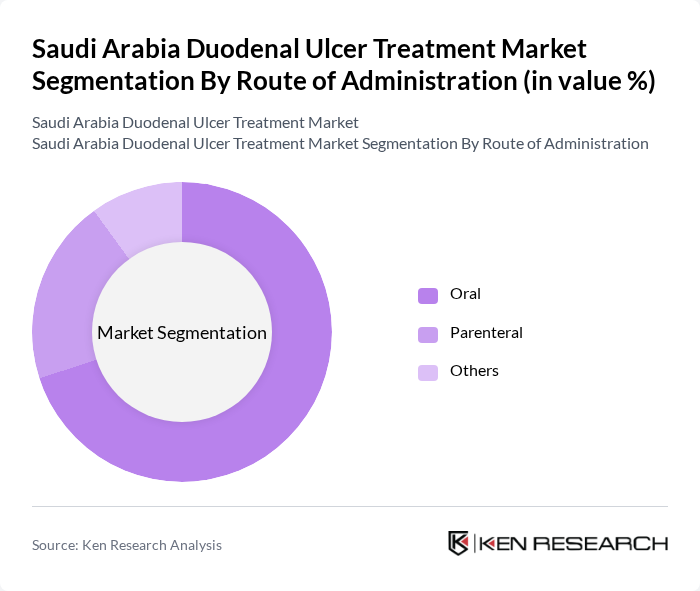
The route of administration segmentation highlights the methods through which treatments are delivered to patients. Oral administration is the most prevalent route, favored for its convenience, patient adherence, and the availability of multiple PPI, H2-blocker, antacid, and antibiotic formulations in tablet, capsule, and suspension forms. Parenteral administration, while less common, is utilized in hospital settings for acute or complicated cases, such as bleeding ulcers or when rapid acid suppression is required, or when patients are unable to take medications orally. Other routes, such as localized endoscopic delivery of hemostatic agents or adjunctive therapies, are less frequently employed but may be used in specific circumstances, particularly in interventional gastroenterology. The dominance of oral administration reflects patient preferences, cost-effectiveness, and the strong evidence base supporting oral regimens for both acute management and maintenance therapy.
The Saudi Arabia Duodenal Ulcer Treatment Market is characterized by a dynamic mix of regional and international players. Leading participants such as AstraZeneca plc, Pfizer Inc., Takeda Pharmaceutical Company Limited, GlaxoSmithKline plc (GSK), Sanofi S.A., Johnson & Johnson (Janssen), Novartis AG, Merck & Co., Inc. (MSD), AbbVie Inc., Bayer AG, Boehringer Ingelheim International GmbH, Amgen Inc., Astellas Pharma Inc., Eli Lilly and Company, Jamjoom Pharma, Tabuk Pharmaceuticals Manufacturing Company, Saudi Pharmaceutical Industries & Medical Appliances Corporation (SPIMACO) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the duodenal ulcer treatment market in Saudi Arabia appears promising, driven by ongoing advancements in medical technology and increased healthcare investments. The integration of telemedicine is expected to enhance patient access to specialists, particularly in underserved areas. Additionally, the focus on personalized medicine will likely lead to more effective treatment protocols tailored to individual patient needs, improving overall health outcomes and patient satisfaction in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Treatment Type | Proton Pump Inhibitors (PPIs) H2-Receptor Antagonists Antacids and Cytoprotective Agents Antibiotic Regimens for H. pylori Eradication Other Pharmacological and Surgical Interventions |
| By Route of Administration | Oral Parenteral Others |
| By End-User | Government Hospitals Private Hospitals Specialized Gastroenterology Clinics Homecare and Other Settings |
| By Patient Demographics | Age Group (Pediatrics, Adults, Geriatrics) Gender (Male, Female) Comorbidity Status (With/Without Chronic Conditions) |
| By Treatment Duration | Short-term Eradication and Healing Regimens (< 8 weeks) Maintenance and Long-term Therapy (? 8 weeks) Rescue and Refractory Therapy |
| By Geographic Region | Central Region (including Riyadh) Eastern Region (including Dammam and Khobar) Western Region (including Jeddah, Mecca, and Medina) Southern and Northern Regions |
| By Payer & Insurance Coverage | Government-funded Coverage (MOH and Public Schemes) Private Health Insurance Out-of-Pocket Payments Other Payers (Employer-based and Institutional Programs) |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gastroenterology Clinics | 100 | Gastroenterologists, Clinic Managers |
| Pharmacy Dispensing Practices | 80 | Pharmacists, Pharmacy Managers |
| Hospital Treatment Protocols | 70 | Hospital Administrators, Medical Directors |
| Patient Experience Surveys | 120 | Patients diagnosed with duodenal ulcers |
| Healthcare Policy Insights | 60 | Health Policy Analysts, Government Officials |
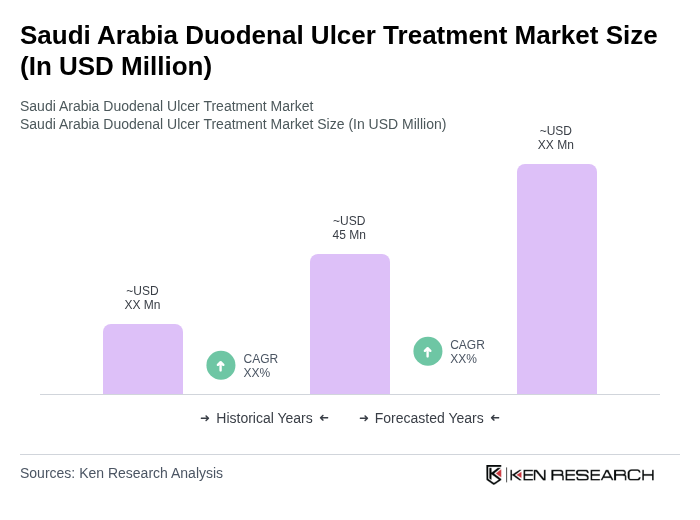
The Saudi Arabia Duodenal Ulcer Treatment Market is valued at approximately USD 45 million, based on a five-year historical analysis. This figure reflects the growing demand for effective treatment options due to the increasing prevalence of duodenal ulcers in the region.