Region:Middle East
Author(s):Rebecca
Product Code:KRAB2111
Pages:84
Published On:January 2026
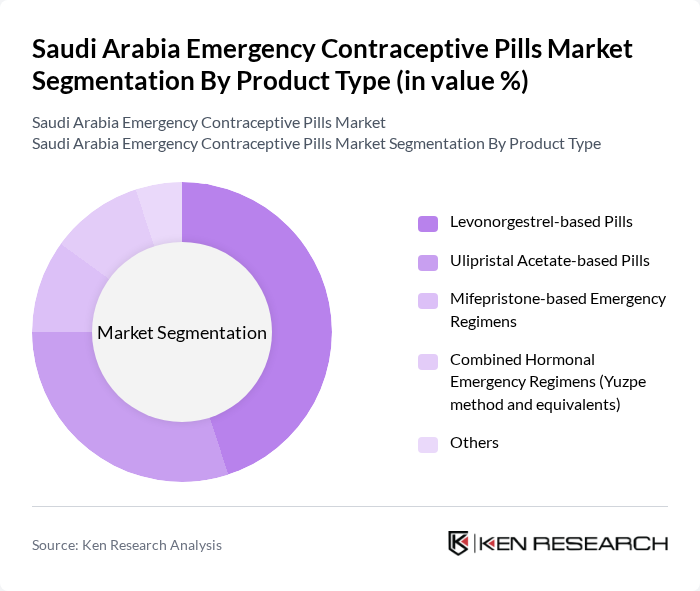
By Product Type:The market is segmented into various product types, including Levonorgestrel-based Pills, Ulipristal Acetate-based Pills, Mifepristone-based Emergency Regimens, Combined Hormonal Emergency Regimens (Yuzpe method and equivalents), and Others. Among these, Levonorgestrel-based Pills are the most widely used globally due to their established efficacy, WHO-recommended dosing, and broad inclusion in national formularies, and they similarly account for the majority of emergency contraception use in Middle East and Asia-Pacific markets. They are preferred by consumers for their ease of use, availability in both branded and generic versions, and quick action, making them a dominant choice in emergency contraceptive options.
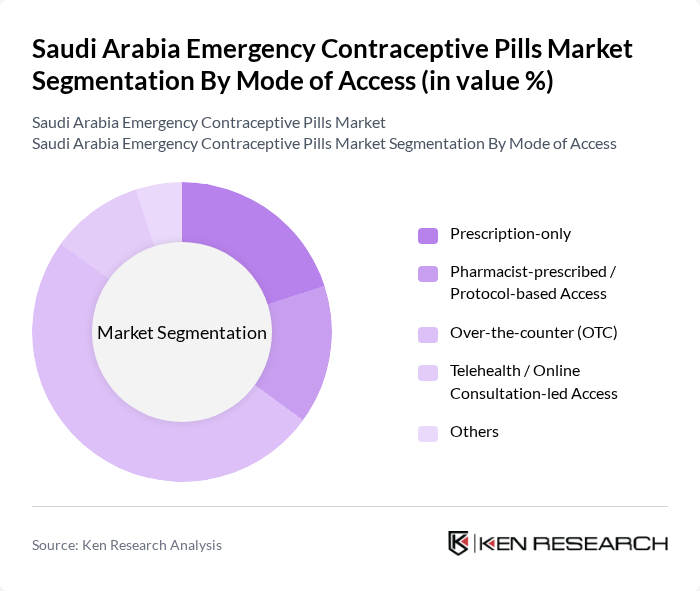
By Mode of Access:The market is also segmented by mode of access, which includes Prescription-only, Pharmacist-prescribed / Protocol-based Access, Over-the-counter (OTC), Telehealth / Online Consultation-led Access, and Others. The Over-the-counter (OTC) segment has gained significant traction globally as more countries authorize non-prescription access to levonorgestrel emergency contraceptive pills, although in Saudi Arabia prescription-based and pharmacist-mediated access remain the primary channels within the SFDA regulatory framework. This shift toward faster access in many markets, together with the growth of digital health platforms and e-pharmacies, reflects changing consumer behavior towards seeking immediate and discreet solutions for reproductive health needs.
The Saudi Arabia Emergency Contraceptive Pills Market is characterized by a dynamic mix of regional and international players. Leading participants such as Bayer AG, HRA Pharma (Perrigo Company plc), Gedeon Richter Plc., Sun Pharmaceutical Industries Ltd., Cipla Ltd., Lupin Limited, Dr. Reddy’s Laboratories Ltd., Mankind Pharma Ltd., Teva Pharmaceutical Industries Ltd., Pfizer Inc., Glenmark Pharmaceuticals Ltd., Aurobindo Pharma Ltd., Julphar (Gulf Pharmaceutical Industries), Tabuk Pharmaceuticals Manufacturing Company, SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corporation) contribute to innovation, geographic expansion, and service delivery in this space, supported by global portfolios in emergency contraception and broader hormonal contraceptive segments.
The future of the emergency contraceptive pills market in Saudi Arabia appears promising, driven by increasing health awareness and government support. As telemedicine services expand, access to reproductive health consultations will improve, facilitating greater awareness and usage of emergency contraceptives. Additionally, the trend towards over-the-counter availability is likely to enhance accessibility, making these products more readily available to women. This evolving landscape suggests a positive trajectory for the market in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Levonorgestrel-based Pills Ulipristal Acetate-based Pills Mifepristone-based Emergency Regimens Combined Hormonal Emergency Regimens (Yuzpe method and equivalents) Others |
| By Mode of Access | Prescription-only Pharmacist-prescribed / Protocol-based Access Over-the-counter (OTC) Telehealth / Online Consultation-led Access Others |
| By Distribution Channel | Hospital Pharmacies Retail / Community Pharmacies Online Pharmacies & E-commerce Platforms Clinics & Primary Health Centers Others |
| By End-User | Women of Reproductive Age (15–24 years) Women of Reproductive Age (25–34 years) Women of Reproductive Age (35–44 years) Healthcare Providers & Institutions Others |
| By Prescriber Type | OB-GYN Specialists General Practitioners / Family Physicians Pharmacists NGOs & Community Health Workers Others |
| By Region | Central Region Eastern Region Western Region Southern Region Northern Region |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Provider Insights | 100 | Gynecologists, Family Physicians |
| Pharmacy Distribution Feedback | 80 | Pharmacists, Pharmacy Managers |
| Consumer Awareness Surveys | 120 | Women aged 18-45, Health Educators |
| Cultural Perception Studies | 60 | Community Health Workers, Social Workers |
| Market Trend Analysis | 70 | Market Analysts, Public Health Officials |
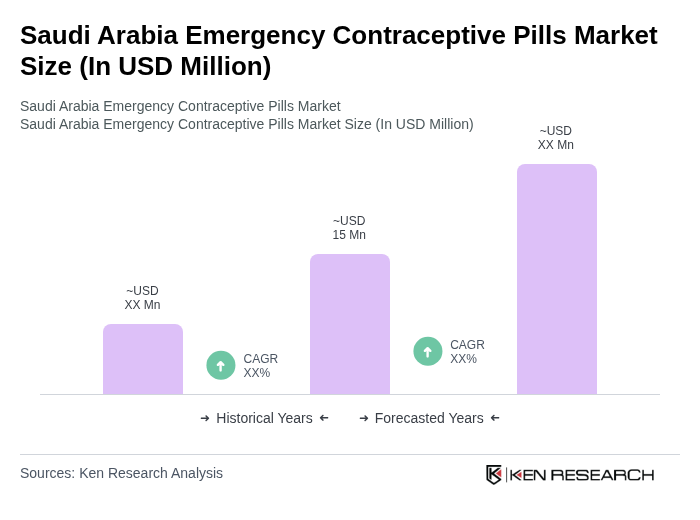
The Saudi Arabia Emergency Contraceptive Pills Market is valued at approximately USD 15 million, reflecting a significant increase in usage driven by rising awareness of reproductive health and family planning solutions among women.