About the Report
Base Year 2024Listen to the audio summary
Saudi Arabia MEA Hip Knee Reconstruction Devices Market Overview
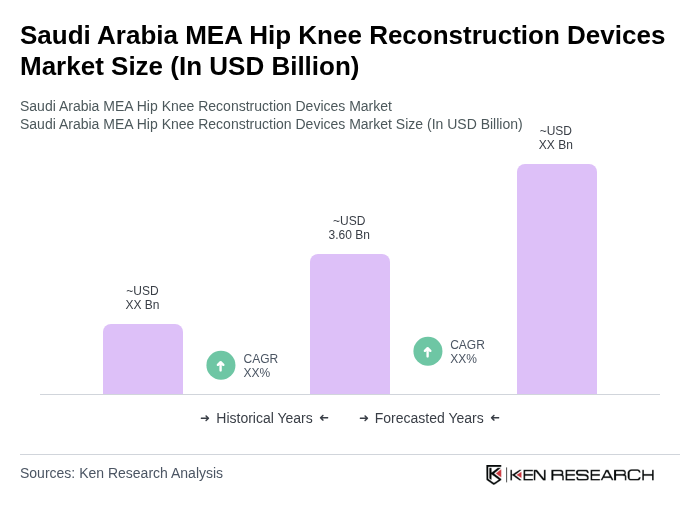
- The Saudi Arabia MEA Hip Knee Reconstruction Devices Market is valued at USD 3.60 billion, based on a five-year historical analysis. This growth is primarily driven by the rising prevalence of musculoskeletal disorders, particularly osteoarthritis, and an increase in trauma-related cases requiring orthopedic interventions. The expansion of healthcare infrastructure and the adoption of advanced surgical technologies, such as robotic-assisted systems, further propel the demand for hip and knee reconstruction solutions in the region.
- Saudi Arabia leads the MEA market, supported by its advanced healthcare facilities and growing investments in specialized medical centers under broader regional healthcare development strategies. Major cities like Riyadh and Jeddah are at the forefront, benefiting from enhanced healthcare services and a focus on orthopedic care, which drives market growth.
- An important regulatory development includes the Medical Devices Interim Regulation introduced by the Saudi Food and Drug Authority, which mandates the registration of joint replacement devices in the national registry and enforces stringent quality and safety standards for hip and knee implants.
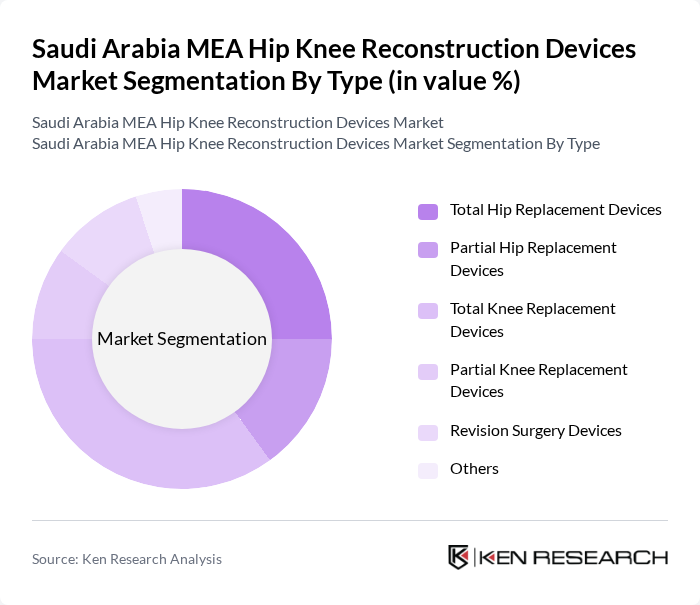
Saudi Arabia MEA Hip Knee Reconstruction Devices Market Segmentation
By Type:The market is segmented into various types of devices, including Total Hip Replacement Devices, Partial Hip Replacement Devices, Total Knee Replacement Devices, Partial Knee Replacement Devices, Revision Surgery Devices, and Others. Among these, Total Knee Replacement Devices are currently dominating the market due to the high incidence of knee-related disorders and the increasing number of surgical procedures performed. The growing elderly population and rising awareness about advanced surgical options are also contributing to the demand for these devices.
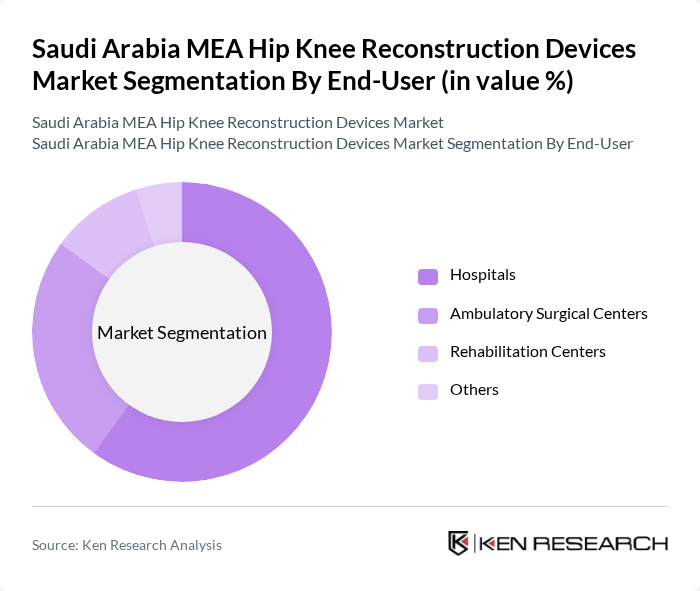
By End-User:The end-user segmentation includes Hospitals, Ambulatory Surgical Centers, Rehabilitation Centers, and Others. Hospitals are the leading end-user segment, accounting for a significant share of the market. This dominance is attributed to the comprehensive services offered by hospitals, including advanced surgical procedures and post-operative care, which are essential for patients undergoing hip and knee reconstruction surgeries.
Saudi Arabia MEA Hip Knee Reconstruction Devices Market Competitive Landscape
The Saudi Arabia MEA Hip Knee Reconstruction Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Johnson & Johnson, Stryker Corporation, Zimmer Biomet, Medtronic, Smith & Nephew, DePuy Synthes, B. Braun Melsungen AG, Arthrex, NuVasive, Conmed Corporation, Orthofix Medical Inc., Aesculap Implant Systems, Exactech, RTI Surgical, Medacta International contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia MEA Hip Knee Reconstruction Devices Market Industry Analysis
Growth Drivers
- Non-Oil GDP Expansion and Economic Diversification:Saudi Arabia's non-oil real GDP increased by SAR 566 billion, reaching SAR 4.5 trillion in the future, with a growth rate of 4.5 percent in the future. This expansion in non-oil sectors, including healthcare, retail, and construction, supports increased demand for hip and knee reconstruction devices. The diversification strategy enhances economic activity, leading to higher healthcare investments and device market growth.
- Robust Fiscal Investment in Vision 2030 Projects:The Saudi government allocated SAR 1.285 trillion for expenditures in the future, following a revised fiscal deficit of SAR 115 billion in the future. This substantial investment in healthcare infrastructure under Vision 2030 is expected to drive demand for advanced orthopedic devices, including hip and knee implants, as the government prioritizes health sector improvements.
- Surge in Medical Devices Market & Health Technology Funding:The Saudi Medical Devices market was valued at USD 6.29 billion in the future, with USD 1.5 billion earmarked for health technology under Vision 2030. This significant funding enhances the adoption of advanced orthopedic devices, including hip and knee reconstruction technologies, driven by increased healthcare spending and innovation in medical technology.
Market Challenges
- Stricter Regulatory Approval Timelines:The approval process for new medical devices in Saudi Arabia typically takes between 6 to 12 months. These lengthy regulatory timelines can delay the launch of advanced hip and knee reconstruction devices, hindering market growth and limiting patient access to innovative treatments. Compliance with evolving regulations adds complexity to the market entry process.
- High Fiscal Deficit Straining Public Budget:The fiscal deficit for the future was revised to SAR 115 billion, with a projected deficit of SAR 101 billion for the future. This financial strain may limit future healthcare spending, potentially delaying infrastructure projects and procurement of orthopedic devices, thereby affecting market growth and innovation in the hip and knee reconstruction sector.
Saudi Arabia MEA Hip Knee Reconstruction Devices Market Future Outlook
Emerging technologies such as digital health integration and AI-enabled diagnostics are reshaping healthcare delivery, expected to complement orthopedic device use. A shift toward value-based care and home-based monitoring suggests rising interest in less invasive, patient-specific reconstruction solutions. Market consolidation and partnerships are likely to grow, as Saudi Arabia aligns with international regulatory standards and seeks to enhance local manufacturing capacity, fostering innovation and accessibility in the orthopedic device market.
Market Opportunities
- Orthopedic Devices Segment Expansion:The orthopedic devices revenue is projected to reach USD 707.79 million in the future, indicating a focused opportunity within the hip and knee reconstruction sub-market. This growth reflects the increasing demand for orthopedic solutions driven by an aging population and rising chronic conditions, presenting a lucrative market for device manufacturers.
- Regulatory Harmonization Enabling Market Entry:The adoption of the SFDA’s risk-based classification system aligns with international standards, simplifying the entry for devices approved elsewhere. This regulatory modernization is expected to accelerate the adoption of advanced reconstruction devices, enhancing market dynamics and fostering innovation in the orthopedic sector.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Total Hip Replacement Devices Partial Hip Replacement Devices Total Knee Replacement Devices Partial Knee Replacement Devices Revision Surgery Devices Others |
| By End-User | Hospitals Ambulatory Surgical Centers Rehabilitation Centers Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Material | Metal-based Devices Polymer-based Devices Ceramic-based Devices Others |
| By Surgical Technique | Open Surgery Minimally Invasive Surgery Robotic-assisted Surgery Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Policy Support | Subsidies for Medical Devices Tax Exemptions for Manufacturers Grants for Research and Development Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Importers
Industry Associations
Financial Institutions
Players Mentioned in the Report:
Johnson & Johnson
Stryker Corporation
Zimmer Biomet
Medtronic
Smith & Nephew
DePuy Synthes
B. Braun Melsungen AG
Arthrex
NuVasive
Conmed Corporation
Orthofix Medical Inc.
Aesculap Implant Systems
Exactech
RTI Surgical
Medacta International
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia MEA Hip Knee Reconstruction Devices Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia MEA Hip Knee Reconstruction Devices Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia MEA Hip Knee Reconstruction Devices Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of orthopedic disorders
3.1.2 Advancements in surgical techniques
3.1.3 Rising geriatric population
3.1.4 Government initiatives for healthcare improvement
3.2 Market Challenges
3.2.1 High cost of advanced devices
3.2.2 Limited awareness among healthcare providers
3.2.3 Regulatory hurdles
3.2.4 Competition from alternative treatments
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Increasing medical tourism
3.3.3 Development of innovative products
3.3.4 Collaborations with local distributors
3.4 Market Trends
3.4.1 Shift towards minimally invasive surgeries
3.4.2 Growing demand for personalized medicine
3.4.3 Integration of digital technologies in healthcare
3.4.4 Focus on patient-centric care
3.5 Government Regulation
3.5.1 Medical device registration requirements
3.5.2 Compliance with international standards
3.5.3 Pricing regulations for medical devices
3.5.4 Policies promoting local manufacturing
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia MEA Hip Knee Reconstruction Devices Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia MEA Hip Knee Reconstruction Devices Market Segmentation
8.1 By Type
8.1.1 Total Hip Replacement Devices
8.1.2 Partial Hip Replacement Devices
8.1.3 Total Knee Replacement Devices
8.1.4 Partial Knee Replacement Devices
8.1.5 Revision Surgery Devices
8.1.6 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Ambulatory Surgical Centers
8.2.3 Rehabilitation Centers
8.2.4 Others
8.3 By Region
8.3.1 Central Region
8.3.2 Eastern Region
8.3.3 Western Region
8.3.4 Southern Region
8.4 By Material
8.4.1 Metal-based Devices
8.4.2 Polymer-based Devices
8.4.3 Ceramic-based Devices
8.4.4 Others
8.5 By Surgical Technique
8.5.1 Open Surgery
8.5.2 Minimally Invasive Surgery
8.5.3 Robotic-assisted Surgery
8.5.4 Others
8.6 By Distribution Channel
8.6.1 Direct Sales
8.6.2 Distributors
8.6.3 Online Sales
8.6.4 Others
8.7 By Policy Support
8.7.1 Subsidies for Medical Devices
8.7.2 Tax Exemptions for Manufacturers
8.7.3 Grants for Research and Development
8.7.4 Others
9. Saudi Arabia MEA Hip Knee Reconstruction Devices Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Product Innovation Rate
9.2.7 Distribution Efficiency
9.2.8 Pricing Strategy
9.2.9 Brand Recognition
9.2.10 Customer Satisfaction Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Johnson & Johnson
9.5.2 Stryker Corporation
9.5.3 Zimmer Biomet
9.5.4 Medtronic
9.5.5 Smith & Nephew
9.5.6 DePuy Synthes
9.5.7 B. Braun Melsungen AG
9.5.8 Arthrex
9.5.9 NuVasive
9.5.10 Conmed Corporation
9.5.11 Orthofix Medical Inc.
9.5.12 Aesculap Implant Systems
9.5.13 Exactech
9.5.14 RTI Surgical
9.5.15 Medacta International
10. Saudi Arabia MEA Hip Knee Reconstruction Devices Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.1.4 Compliance Requirements
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends in Healthcare Facilities
10.2.2 Spending on Medical Equipment
10.2.3 Budgeting for Surgical Procedures
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges Faced by Hospitals
10.3.2 Issues in Ambulatory Surgical Centers
10.3.3 Rehabilitation Center Needs
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training and Education Needs
10.4.2 Technology Acceptance Levels
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Success Metrics
10.5.2 Case Studies of Successful Implementations
10.5.3 Future Expansion Opportunities
10.5.4 Others
11. Saudi Arabia MEA Hip Knee Reconstruction Devices Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Solutions
9.2 Export Entry Strategy
9.2.1 Target Countries Identification
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements Analysis
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnerships Evaluation
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability Strategies
14. Potential Partner List
14.1 Distributors Identification
14.2 Joint Ventures Opportunities
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup Steps
15.1.2 Market Entry Strategies
15.1.3 Growth Acceleration Tactics
15.1.4 Scale & Stabilize Approaches
15.2 Key Activities and Milestones
15.2.1 Milestone Identification
15.2.2 Activity Scheduling
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from healthcare associations and medical device regulatory bodies in Saudi Arabia
- Review of published articles and journals focusing on hip and knee reconstruction technologies
- Examination of demographic and epidemiological data related to orthopedic conditions in the region
Primary Research
- Interviews with orthopedic surgeons and healthcare professionals specializing in joint reconstruction
- Surveys conducted with hospital procurement departments regarding device purchasing trends
- Focus groups with patients who have undergone hip or knee reconstruction surgeries
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and industry reports
- Triangulation of data from healthcare providers, manufacturers, and regulatory agencies
- Sanity checks through feedback from a panel of orthopedic specialists
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and orthopedic device spending
- Segmentation of the market by device type, including total hip and knee implants
- Incorporation of growth rates from historical data and projected healthcare trends
Bottom-up Modeling
- Collection of sales data from leading manufacturers of hip and knee reconstruction devices
- Estimation of unit sales based on surgical volume and device usage rates
- Cost analysis of devices to determine average selling prices across different segments
Forecasting & Scenario Analysis
- Utilization of time-series analysis to project future market growth based on historical trends
- Scenario modeling based on potential changes in healthcare policies and patient demographics
- Development of best-case, worst-case, and most-likely scenarios through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Orthopedic Surgeons | 100 | Surgeons specializing in hip and knee procedures |
| Hospital Procurement Managers | 80 | Managers responsible for purchasing medical devices |
| Patients Post-Surgery | 75 | Patients who have undergone hip or knee reconstruction |
| Healthcare Policy Makers | 50 | Officials involved in healthcare regulations and policies |
| Medical Device Manufacturers | 60 | Executives from companies producing orthopedic devices |
Frequently Asked Questions
What is the current value of the Saudi Arabia MEA Hip Knee Reconstruction Devices Market?
The Saudi Arabia MEA Hip Knee Reconstruction Devices Market is valued at approximately USD 3.60 billion, reflecting significant growth driven by the rising prevalence of musculoskeletal disorders and advancements in healthcare infrastructure and surgical technologies.