Region:Middle East
Author(s):Dev
Product Code:KRAA3874
Pages:82
Published On:January 2026
By Product Type:
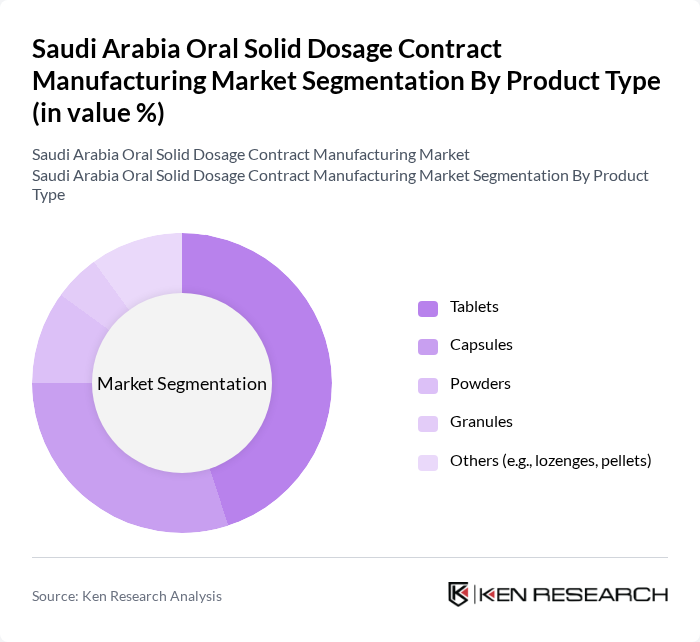
The product type segmentation includes Tablets, Capsules, Powders, Granules, and Others (e.g., lozenges, pellets). Among these, Tablets dominate the market due to their widespread use, ease of administration, and suitability for high-volume chronic therapies such as cardiovascular and metabolic conditions. The preference for tablets is driven by their stability, accurate dose uniformity, scalability in manufacturing, and cost-effectiveness. Capsules are also gaining traction, particularly in the nutraceutical and specialty prescription segments, as they can accommodate complex, multiparticulate, or moisture-sensitive formulations and enable faster product lifecycle management. Powders and Granules are utilized for specific therapeutic applications, pediatric and geriatric patients, and as intermediates for further tableting or encapsulation. The Others category, which includes lozenges, pellets, and orally disintegrating forms, caters to niche patient-centric and fast-dissolving formulations, contributing to the overall diversification of product offerings.
By Release / Formulation Type:
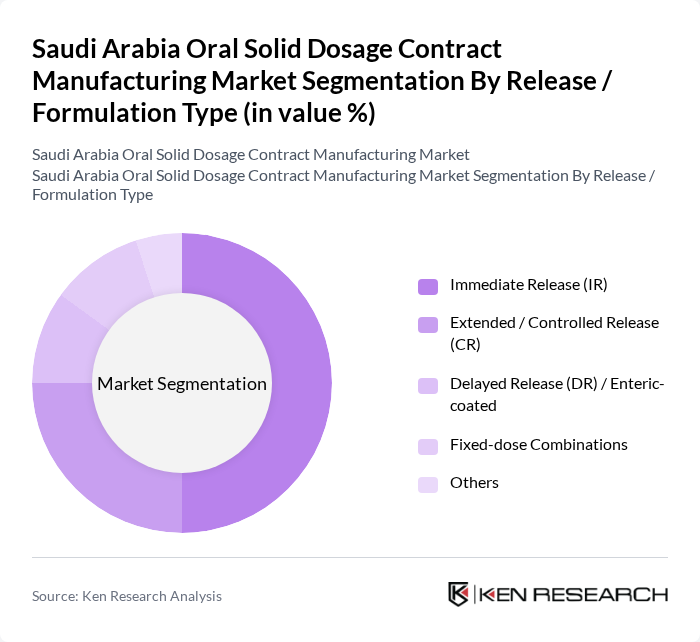
This segmentation includes Immediate Release (IR), Extended / Controlled Release (CR), Delayed Release (DR) / Enteric-coated, Fixed-dose Combinations, and Others. The Immediate Release (IR) formulations lead the market due to their rapid onset of action and suitability for a broad range of acute and chronic indications, making them the most commonly outsourced oral dosage form globally and in the region. Extended and Controlled Release formulations are gaining popularity for chronic conditions such as diabetes, hypertension, and psychiatric disorders, as they enable sustained therapeutic levels, improved adherence, and reduced dosing frequency, which encourages originator and generic companies to partner with CDMOs that have advanced formulation capabilities. Delayed Release and enteric?coated formulations are utilized to protect acid?labile drugs, target specific intestinal regions, and reduce gastrointestinal side effects, requiring specialized coating technologies that CDMOs increasingly provide. Fixed-dose Combinations are favored for their convenience in managing comorbidities and aligning with national treatment guidelines, especially in cardiovascular and metabolic diseases, and they support differentiation in the generics space. The Others category includes orally disintegrating tablets, multi-layer tablets, and abuse-deterrent or modified-release technologies that cater to specific patient needs and align with the broader trend toward patient-centric drug delivery.
The Saudi Arabia Oral Solid Dosage Contract Manufacturing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Saudi Pharmaceutical Industries and Medical Appliances Corporation (SPIMACO), Tabuk Pharmaceuticals Manufacturing Company, Jamjoom Pharma, Riyadh Pharma (Medical Application Company), Saudi Chemical Company Holding – AJA Pharma, Jamjoom Medical Industries, Sudair Pharma Company, Hikma Pharmaceuticals, Julphar Gulf Pharmaceutical Industries, Tabuk Pharmaceuticals Manufacturing Marketing Affiliates (Saudi), Pfizer Saudi Limited, Sanofi Saudi Arabia, Bayer Saudi Arabia, Merck Sharp & Dohme (MSD) Saudi Arabia, Other Emerging Saudi CDMOs contribute to innovation, geographic expansion, technology transfer, and service delivery in this space by investing in local manufacturing, quality upgrades, and broader portfolios of tablets and capsules for both branded and generic therapies.
The future of the Saudi Arabia oral solid dosage contract manufacturing market appears promising, driven by advancements in technology and increasing healthcare investments. As the government continues to support local manufacturing through incentives, companies are likely to adopt innovative production methods. Additionally, the growing focus on personalized medicine and biopharmaceuticals will create new avenues for growth, enabling manufacturers to cater to evolving patient needs and preferences in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Tablets Capsules Powders Granules Others (e.g., lozenges, pellets) |
| By Release / Formulation Type | Immediate Release (IR) Extended / Controlled Release (CR) Delayed Release (DR) / Enteric-coated Fixed-dose Combinations Others |
| By Service Type | Drug Product Development Commercial Manufacturing Clinical Trial Manufacturing Packaging and Labelling Others |
| By Customer Type | Large Pharmaceutical Companies Medium & Small Pharmaceutical Companies Biotechnology Companies Nutraceutical & OTC Companies Others |
| By Therapeutic Area | Cardiovascular Diseases Oncology Metabolic & Endocrine Disorders (e.g., diabetes) Neurological & CNS Disorders Infectious & Gastrointestinal Diseases Others |
| By Stage | Clinical Commercial |
| By Region | Central Region (incl. Riyadh) Western Region (incl. Makkah, Madinah) Eastern Region (incl. Dammam, Al Khobar) Northern Region Southern Region |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Contract Manufacturers | 90 | Operations Managers, Quality Assurance Heads |
| Regulatory Affairs Professionals | 80 | Regulatory Managers, Compliance Officers |
| Pharmaceutical Distributors | 70 | Supply Chain Managers, Distribution Heads |
| Healthcare Providers | 60 | Pharmacists, Hospital Procurement Officers |
| Market Analysts | 50 | Industry Analysts, Market Research Specialists |
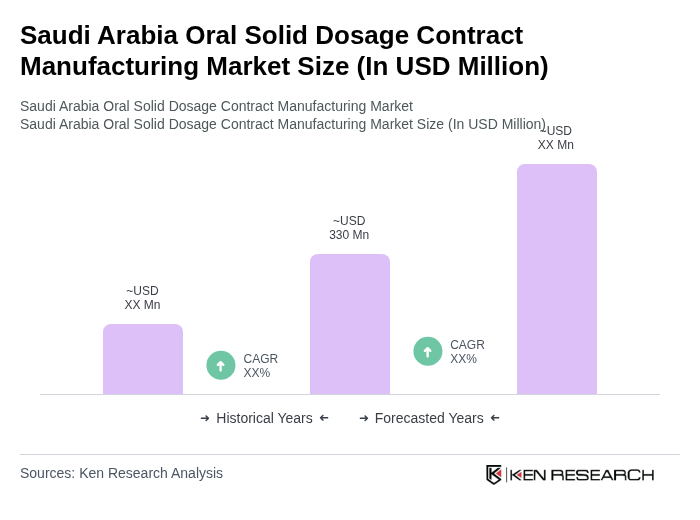
The Saudi Arabia Oral Solid Dosage Contract Manufacturing Market is valued at approximately USD 330 million, reflecting a historical analysis of revenues and their share within the broader pharmaceutical contract development and manufacturing organization (CDMO) market in the country.