Region:Middle East
Author(s):Geetanshi
Product Code:KRAD7210
Pages:96
Published On:December 2025
By Test Type:The market is segmented into various test types, including Molecular Diagnostics, Immunoassays & Clinical Chemistry, Hematology & Coagulation Tests, Microbiology & Infectious Disease LDTs, Histology / Cytology & Tissue-based LDTs, Flow Cytometry-based LDTs, Mass Spectrometry-based LDTs, and Other Specialized LDTs. Among these, Molecular Diagnostics is a leading segment due to its critical role in the early detection of infectious and genetic diseases and the growing adoption of PCR and NGS technologies in Saudi hospital and reference laboratories. The increasing focus on precision medicine and personalized healthcare, particularly in oncology, rare diseases, and pharmacogenomics, is driving the demand for molecular diagnostics, making it a key growth driver in the market.

By End-User:The market is segmented by end-users, including Hospital-Based Laboratories, Independent Diagnostic Laboratories, Academic & Research Institutions, Reference & Specialty Laboratories, and Others. Hospital-Based Laboratories dominate the market due to their extensive patient base, integration into large public and private hospital networks, and the need for comprehensive diagnostic services supporting complex clinical specialties. The integration of advanced technologies such as high?throughput molecular analyzers, automation, and digital pathology in these facilities enhances their testing capabilities, making them the preferred choice for patients and healthcare providers alike.

The Saudi Arabia Pacific Laboratory Developed Tests Market is characterized by a dynamic mix of regional and international players. Leading participants such as Al Borg Medical Laboratories, King Faisal Specialist Hospital & Research Centre, Dr. Sulaiman Al Habib Medical Group, Saudi German Hospitals Group, Dallah Health Company, Al-Moosa Specialist Hospital, Al Nahdi Medical Company, Al-Dar Medical Services, King Saud University Medical City, King Abdulaziz Medical City (National Guard Health Affairs), Saudi Biotech Company, Mayo Clinic Laboratories, Eurofins Scientific, Quest Diagnostics, Labcorp contribute to innovation, geographic expansion, and service delivery in this space.
The future of the laboratory developed tests market in Saudi Arabia appears promising, driven by ongoing advancements in technology and a growing emphasis on personalized healthcare. As the healthcare sector continues to evolve, the integration of artificial intelligence and machine learning in diagnostics is expected to enhance test accuracy and efficiency. Additionally, the expansion of telemedicine services will facilitate remote diagnostics, making laboratory developed tests more accessible to patients across the region, ultimately improving health outcomes and patient engagement.
| Segment | Sub-Segments |
|---|---|
| By Test Type | Molecular Diagnostics (PCR, RT-PCR, qPCR) Immunoassays & Clinical Chemistry Hematology & Coagulation Tests Microbiology & Infectious Disease LDTs Histology / Cytology & Tissue-based LDTs Flow Cytometry-based LDTs Mass Spectrometry-based LDTs Other Specialized LDTs (e.g., toxicology, pharmacogenomics) |
| By End-User | Hospital-Based Laboratories Independent Diagnostic Laboratories Academic & Research Institutions Reference & Specialty Laboratories Others |
| By Clinical Application | Infectious Diseases (including respiratory & hospital-acquired infections) Oncology & Hemato-oncology Genetic & Genomic Testing (including carrier & pharmacogenomic tests) Cardiometabolic & Endocrine Disorders Autoimmune & Inflammatory Diseases Therapeutic Drug Monitoring & Toxicology Others |
| By Technology Platform | PCR & Real-Time PCR Next-Generation Sequencing (NGS) Microarray & Gene Expression Profiling Immunoassay Platforms (CLIA, ELISA, etc.) Flow Cytometry Platforms Mass Spectrometry Platforms Others |
| By Test Complexity & Development Model | High-Complexity LDTs Moderate-Complexity LDTs Laboratory–Industry Co-developed LDTs In-house Developed LDTs |
| By Region | Central Region (including Riyadh) Eastern Region (including Dammam, Khobar) Western Region (including Jeddah, Makkah, Madinah) Southern & Northern Regions |
| By Policy & Reimbursement Environment | Government Funding & Subsidy Support Public & Private Insurance Reimbursement Coverage Research & Innovation Grants Public–Private Partnership (PPP) Frameworks Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Laboratory Managers | 100 | Laboratory Directors, Quality Assurance Managers |
| Healthcare Providers | 80 | Physicians, Nurse Practitioners |
| Diagnostic Test Manufacturers | 70 | Product Managers, Sales Directors |
| Regulatory Affairs Specialists | 60 | Compliance Officers, Regulatory Managers |
| Public Health Officials | 60 | Epidemiologists, Health Policy Analysts |
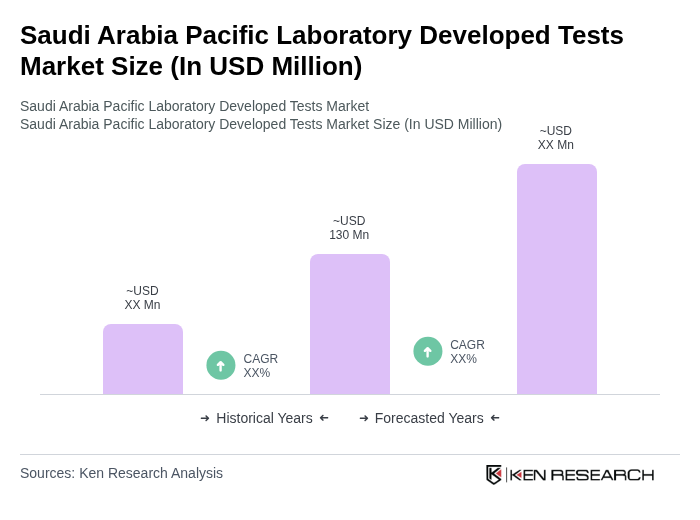
The Saudi Arabia Pacific Laboratory Developed Tests Market is valued at approximately USD 130 million, reflecting a significant growth driven by the increasing prevalence of chronic diseases, advancements in diagnostic technologies, and a focus on personalized medicine.