Region:Middle East
Author(s):Shubham
Product Code:KRAA8679
Pages:82
Published On:November 2025
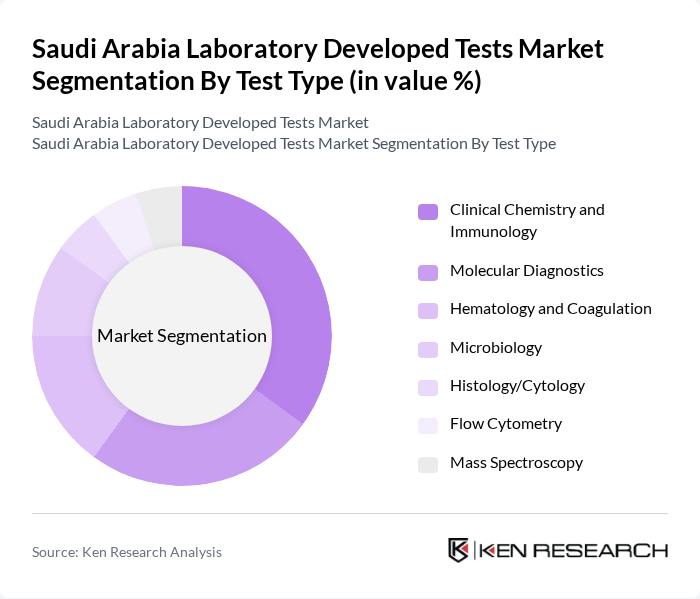
By Test Type:The market is segmented into various test types, including Clinical Chemistry and Immunology, Molecular Diagnostics, Hematology and Coagulation, Microbiology, Histology/Cytology, Flow Cytometry, and Mass Spectroscopy. Among these, Clinical Chemistry and Immunology is the leading segment due to its extensive application in routine diagnostics and the growing demand for biochemical tests. The increasing prevalence of metabolic disorders and the need for timely disease detection further bolster this segment's dominance. Molecular Diagnostics is also experiencing rapid growth, driven by technological advancements and the rising burden of genetic and infectious diseases.
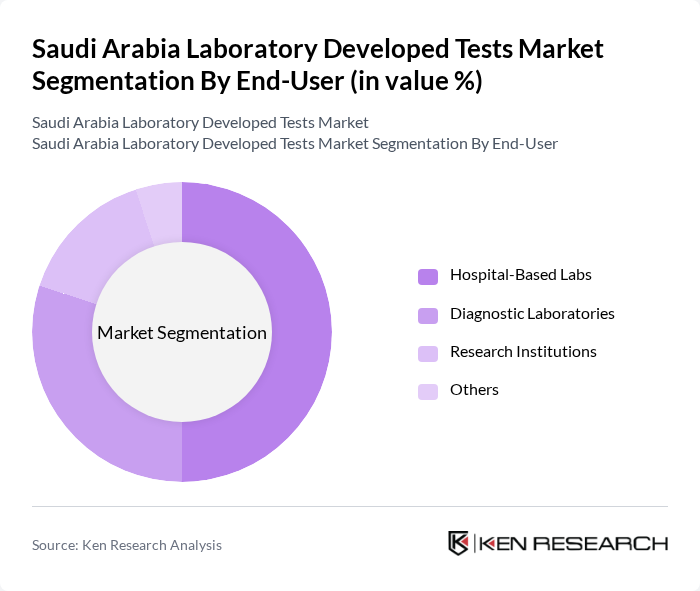
By End-User:The market is segmented by end-users, including Hospital-Based Labs, Diagnostic Laboratories, Research Institutions, and Others. Hospital-Based Labs dominate the market due to their comprehensive testing capabilities and the increasing number of patients seeking diagnostic services. The integration of advanced technologies in these labs enhances their efficiency and accuracy, making them the preferred choice for healthcare providers. Diagnostic Laboratories are also expanding rapidly, supported by collaborations with international organizations and the adoption of new testing platforms.
The Saudi Arabia Laboratory Developed Tests Market is characterized by a dynamic mix of regional and international players. Leading participants such as Al Borg Laboratories, King Faisal Specialist Hospital & Research Centre, Eurofins Scientific, Mayo Clinic Laboratories, Dr. Sulaiman Al Habib Medical Group, Saudi German Hospitals Group, Al Nahdi Medical Company, Al-Dar Medical Services, Dallah Hospital, Al-Moosa Specialist Hospital, Al-Salam International Hospital, Saudi Biotech Company contribute to innovation, geographic expansion, and service delivery in this space.
The future of the laboratory developed tests market in Saudi Arabia appears promising, driven by technological advancements and a growing emphasis on personalized healthcare. As the healthcare infrastructure expands, more laboratories will be equipped to develop innovative tests. Additionally, the integration of artificial intelligence in diagnostics is expected to enhance test accuracy and efficiency. These trends will likely lead to increased adoption of laboratory developed tests, improving patient outcomes and driving market growth in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Test Type | Clinical Chemistry and Immunology Molecular Diagnostics Hematology and Coagulation Microbiology Histology/Cytology Flow Cytometry Mass Spectroscopy |
| By End-User | Hospital-Based Labs Diagnostic Laboratories Research Institutions Others |
| By Therapeutic Area | Genetic Disorders Infections Oncology Metabolic Disorders Others |
| By Product Type | Consumables Instruments Software and Services |
| By Distribution Channel | Direct Tender Direct Sales Distributors Online Sales |
| By Sample Type | Blood Urine Tissue Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Laboratories | 100 | Laboratory Managers, Quality Assurance Officers |
| Healthcare Providers | 70 | Physicians, Diagnostic Test Coordinators |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Research Institutions | 60 | Research Scientists, Clinical Researchers |
| Industry Associations | 50 | Policy Makers, Industry Analysts |
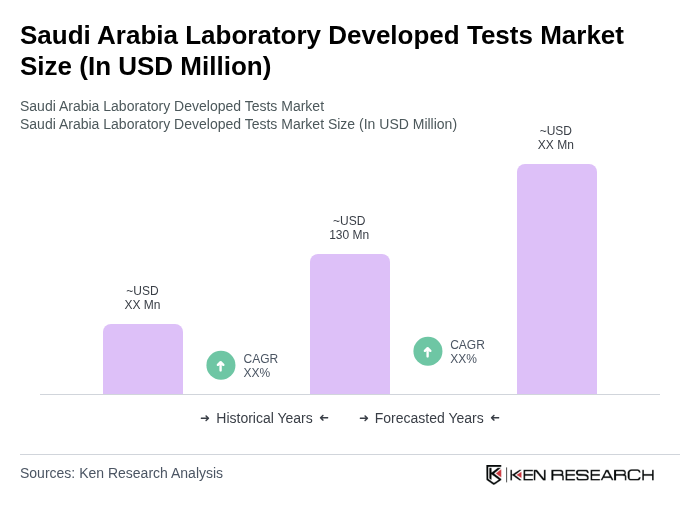
The Saudi Arabia Laboratory Developed Tests Market is valued at approximately USD 130 million, reflecting a significant growth driven by the increasing prevalence of chronic diseases, advancements in diagnostic technologies, and a focus on personalized medicine.