Region:Middle East
Author(s):Dev
Product Code:KRAE0036
Pages:92
Published On:December 2025
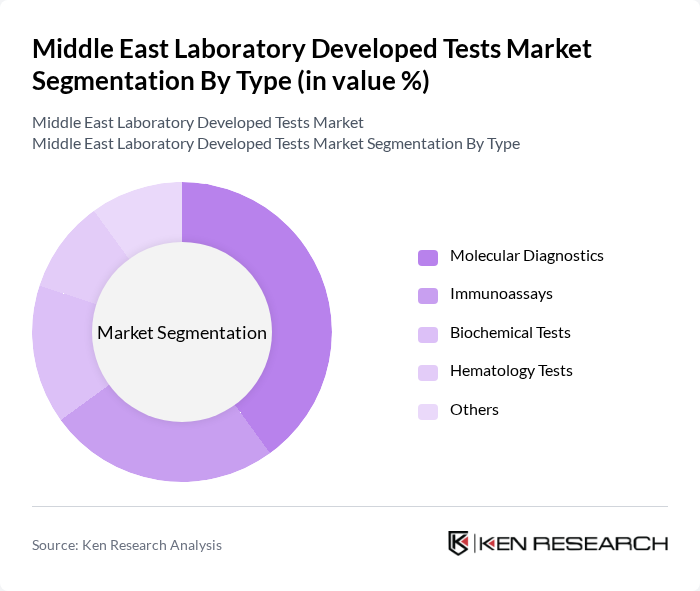
By Type:The market is segmented into various types, including Molecular Diagnostics, Immunoassays, Biochemical Tests, Hematology Tests, and Others. Among these, Molecular Diagnostics is the leading sub-segment due to its ability to provide precise and rapid results, which are crucial for effective patient management. The increasing adoption of personalized medicine and the growing prevalence of infectious diseases further bolster the demand for this technology.
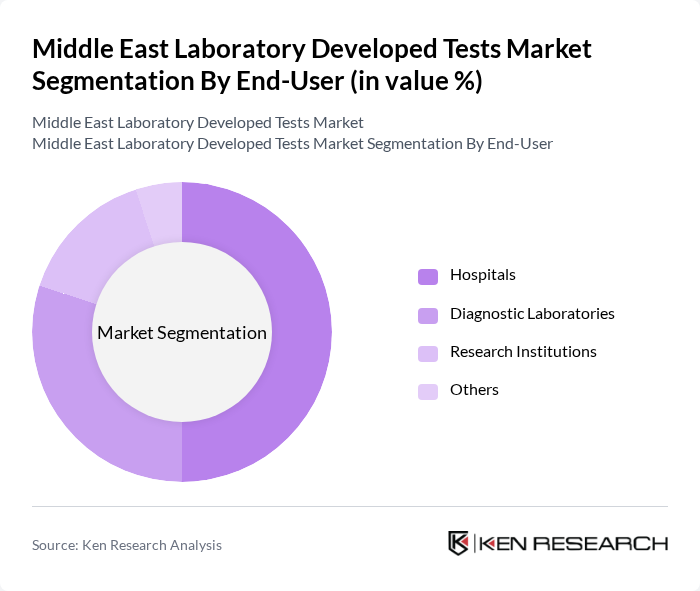
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Research Institutions, and Others. Hospitals are the dominant end-user segment, driven by the increasing number of patients requiring diagnostic testing and the need for timely results in clinical decision-making. The integration of advanced laboratory services within hospitals enhances their operational efficiency and patient care.
The Middle East Laboratory Developed Tests Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Thermo Fisher Scientific, Bio-Rad Laboratories, QIAGEN, Hologic, Inc., PerkinElmer, Inc., Becton, Dickinson and Company, Agilent Technologies, Genomic Health, Illumina, Inc., Cepheid, Luminex Corporation, Myriad Genetics contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Middle East laboratory developed tests market appears promising, driven by ongoing advancements in technology and a growing emphasis on personalized healthcare solutions. As healthcare infrastructure expands, particularly in emerging economies, the accessibility of LDTs is expected to improve. Additionally, the integration of digital health technologies will facilitate remote diagnostics and monitoring, further enhancing patient engagement. These trends indicate a robust market trajectory, with significant opportunities for innovation and collaboration in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Molecular Diagnostics Immunoassays Biochemical Tests Hematology Tests Others |
| By End-User | Hospitals Diagnostic Laboratories Research Institutions Others |
| By Application | Infectious Diseases Genetic Testing Cancer Diagnostics Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Others |
| By Technology | PCR Technology Next-Generation Sequencing Microarray Technology Others |
| By Region | GCC Countries Levant Region North Africa Others |
| By Policy Support | Government Grants Tax Incentives Research Funding Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Laboratories | 150 | Laboratory Managers, Medical Technologists |
| Healthcare Providers | 100 | Physicians, Pathologists |
| Regulatory Bodies | 50 | Regulatory Affairs Specialists, Compliance Officers |
| Diagnostic Equipment Manufacturers | 80 | Product Managers, Sales Directors |
| Research Institutions | 70 | Research Scientists, Lab Directors |
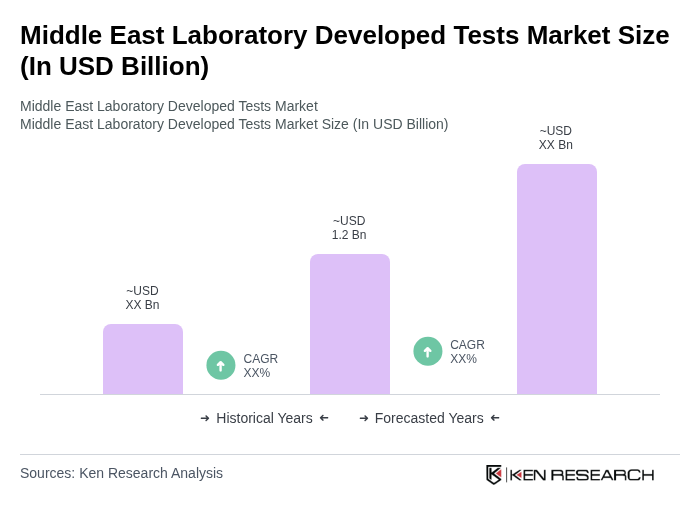
The Middle East Laboratory Developed Tests Market is valued at approximately USD 1.2 billion, reflecting a significant growth driven by the rising prevalence of chronic diseases, advancements in diagnostic technologies, and an increasing focus on personalized medicine.