Region:Middle East
Author(s):Dev
Product Code:KRAC8737
Pages:92
Published On:November 2025
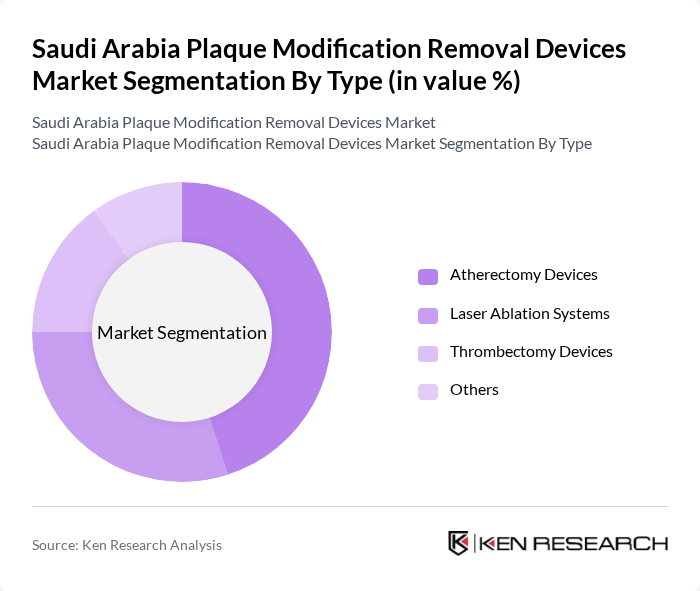
By Type:The market is segmented into various types of plaque modification removal devices, including atherectomy devices, laser ablation systems, thrombectomy devices, and others. Among these, atherectomy devices are gaining significant traction due to their efficiency and effectiveness in plaque removal. The increasing preference for minimally invasive and catheter-based interventions is driving the demand for these devices, making them the leading sub-segment in the market.
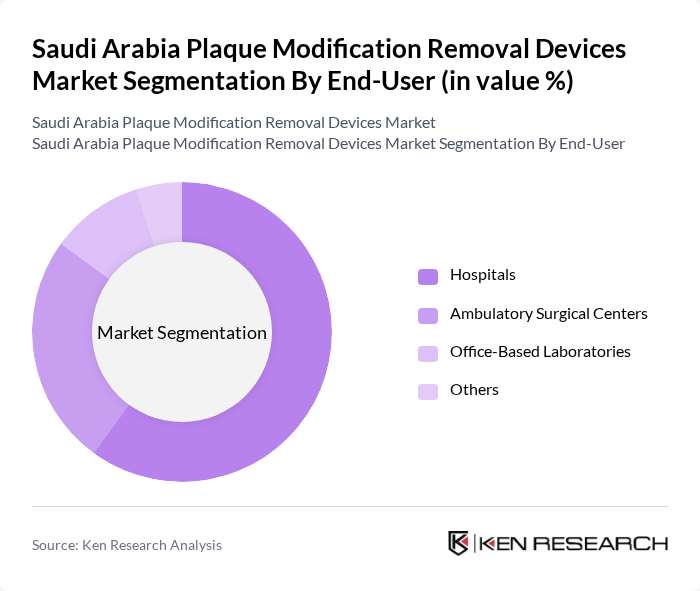
By End-User:The end-user segmentation includes hospitals, ambulatory surgical centers (ASCs), office-based laboratories, and others. Hospitals are the primary users of plaque modification removal devices, as they are equipped with the necessary infrastructure and trained professionals to utilize these advanced technologies. The growing adoption of outpatient cardiovascular procedures in ASCs and office-based settings is significantly contributing to the market's expansion.
The Saudi Arabia Plaque Modification Removal Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Dentsply Sirona, Straumann Group, 3M Company, Henry Schein, Inc., Patterson Companies, Inc., Carestream Health, Nobel Biocare, Align Technology, Inc., KaVo Kerr, Ivoclar Vivadent AG, GC Corporation, Vatech Co., Ltd., Planmeca Oy, Benco Dental, Biolase, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the plaque modification removal devices market in Saudi Arabia appears promising, driven by increasing investments in healthcare infrastructure and a growing emphasis on preventive dental care. As the government continues to promote dental health initiatives, the market is likely to see a surge in demand for innovative dental solutions. Furthermore, the integration of digital technologies in dental practices is expected to enhance patient experiences and treatment outcomes, fostering a more robust market environment.
| Segment | Sub-Segments |
|---|---|
| By Type | Ultrasonic Devices Laser Devices Handheld Devices Others |
| By End-User | Dental Clinics Hospitals Home Care Others |
| By Distribution Channel | Direct Sales Online Retail Distributors Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Application | Preventive Care Therapeutic Care Cosmetic Procedures Others |
| By Technology | Digital Imaging CAD/CAM Systems D Printing Others |
| By Policy Support | Subsidies for dental equipment Tax incentives for manufacturers Grants for research and development Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Dental Clinics | 120 | Dentists, Clinic Managers |
| Hospitals with Dental Departments | 80 | Healthcare Administrators, Dental Surgeons |
| Dental Equipment Distributors | 60 | Sales Managers, Product Specialists |
| Patient Focus Groups | 50 | Patients who have undergone plaque removal procedures |
| Regulatory Bodies | 40 | Health Policy Analysts, Regulatory Affairs Managers |
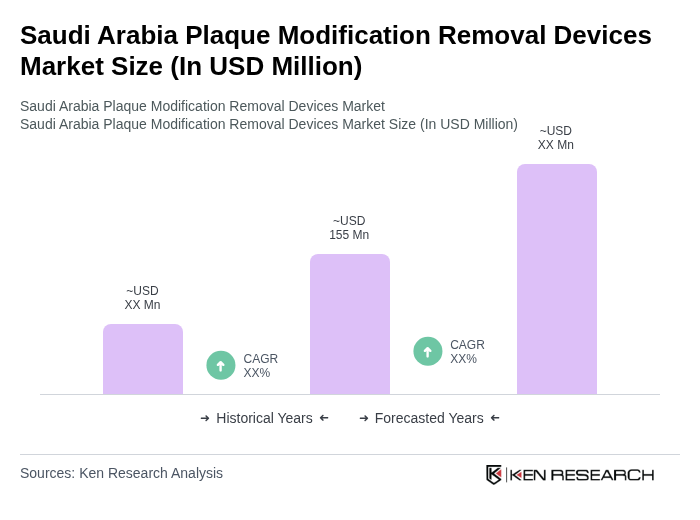
The Saudi Arabia Plaque Modification Removal Devices Market is valued at approximately USD 155 million, reflecting a significant growth trend driven by the increasing prevalence of cardiovascular diseases and advancements in catheter-based plaque removal technologies.