About the Report
Base Year 2024Saudi Arabia Point of Care Molecular Diagnostics Market Overview
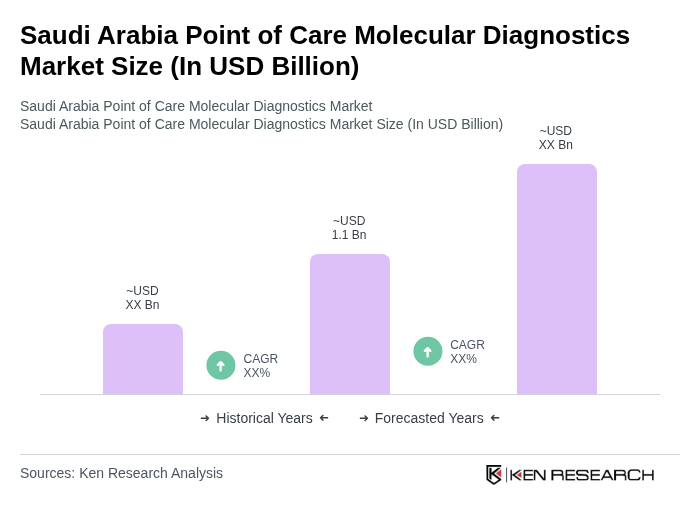
- The Saudi Arabia Point of Care Molecular Diagnostics Market is valued at USD 1.1 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of infectious diseases, the rising demand for rapid diagnostic tests, and advancements in molecular technologies that enhance the accuracy and speed of diagnostics. The market is further supported by significant government investment in healthcare infrastructure and digital health initiatives under Vision 2030, which prioritize innovation, capacity expansion, and integration of advanced diagnostic tools .
- Key cities such as Riyadh, Jeddah, and Dammam dominate the market due to their advanced healthcare infrastructure, high population density, and the presence of leading healthcare institutions. These urban centers are also focal points for medical research and innovation, contributing to the growth of the diagnostics market. The adoption of smart city health solutions, including IoT-enabled diagnostics and telemedicine, further strengthens the position of these cities as healthcare innovation hubs .
- In 2023, the Saudi Food and Drug Authority (SFDA) implemented the “Medical Devices Interim Regulation” (issued by the Saudi Food and Drug Authority, 2023), which streamlines the approval process for point-of-care molecular diagnostic devices. This regulation mandates that all molecular diagnostic devices undergo conformity assessment and registration with the SFDA prior to market entry, ensuring that healthcare providers can access timely and effective testing options while maintaining safety and performance standards .
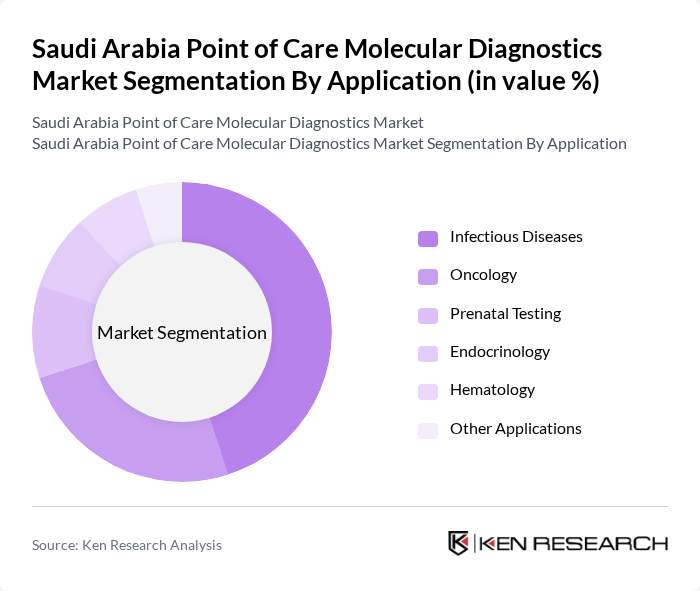
Saudi Arabia Point of Care Molecular Diagnostics Market Segmentation
By Application:The application segment includes various areas such as infectious diseases, oncology, prenatal testing, endocrinology, hematology, and other applications. Among these, infectious diseases are the leading sub-segment due to the rising incidence of viral and bacterial infections, which necessitate rapid and accurate diagnostic solutions. The demand for oncology testing is also growing, driven by increased awareness, early detection initiatives, and the integration of molecular diagnostics into cancer care pathways. The diverse applications of molecular diagnostics cater to a wide range of healthcare needs, making this segment crucial for market growth .
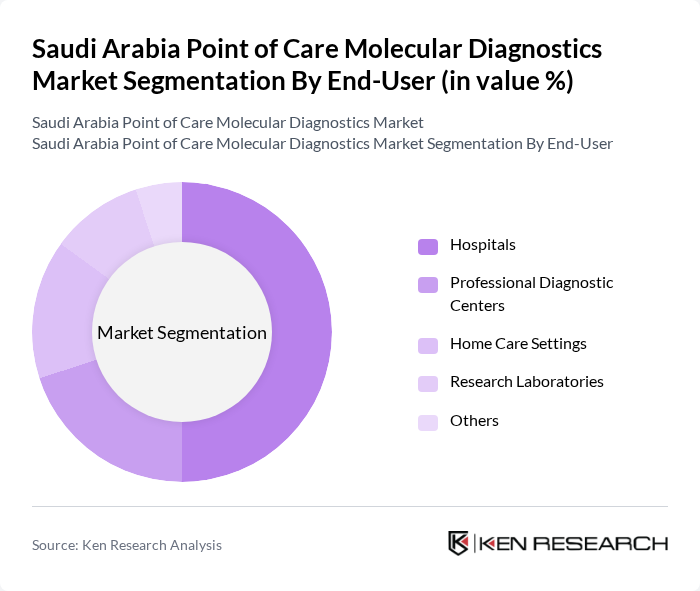
By End-User:The end-user segment comprises hospitals, professional diagnostic centers, home care settings, research laboratories, and others. Hospitals are the dominant end-user in this market, driven by the increasing adoption of point-of-care testing to enhance patient care and reduce turnaround times for diagnostic results. Professional diagnostic centers are also gaining traction as they provide specialized testing services. The growing trend of home care settings for diagnostics is expected to further diversify the market, catering to patient convenience and accessibility. The integration of digital health platforms and telemedicine is also expanding the reach of molecular diagnostics to non-traditional care settings .
Saudi Arabia Point of Care Molecular Diagnostics Market Competitive Landscape
The Saudi Arabia Point of Care Molecular Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics, Qiagen N.V., BioMérieux S.A., Danaher Corporation, Hologic, Inc., Bayer AG, Bio-Rad Laboratories, Inc., Agilent Technologies, Inc., OraSure Technologies, Inc., Nova Biomedical, Nipro Corporation contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia Point of Care Molecular Diagnostics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Infectious Diseases:The rise in infectious diseases in Saudi Arabia is significant, with reported cases of tuberculosis reaching approximately 1,500 annually and a notable increase in viral infections like COVID-19. The World Health Organization reported that infectious diseases account for about 30% of total deaths in the region. This growing health crisis drives the demand for point-of-care molecular diagnostics, which provide rapid and accurate results, essential for timely treatment and containment of outbreaks.
- Rising Demand for Rapid Diagnostic Solutions:The urgency for rapid diagnostic solutions is underscored by the healthcare system's need to manage patient loads effectively. In future, the average turnaround time for traditional diagnostics is projected to be around 48 hours, while point-of-care tests can deliver results in under an hour. This efficiency is crucial in emergency settings, where timely diagnosis can significantly impact patient outcomes, thus propelling the market for molecular diagnostics in Saudi Arabia.
- Technological Advancements in Molecular Diagnostics:The Saudi Arabian market is witnessing rapid technological advancements, with investments in innovative diagnostic tools increasing by 15% annually. The introduction of next-generation sequencing and CRISPR technology is enhancing the accuracy and speed of molecular diagnostics. These advancements are supported by a growing number of research institutions, with over 50 active research projects focused on diagnostics, fostering a conducive environment for market growth and innovation.
Market Challenges
- High Costs of Advanced Diagnostic Equipment:The initial investment required for advanced molecular diagnostic equipment can be prohibitive, with costs ranging from $50,000 to $200,000 per unit. This financial barrier limits access for smaller healthcare facilities and clinics, which may struggle to adopt these technologies. As a result, the high costs hinder the widespread implementation of point-of-care molecular diagnostics, particularly in rural areas where healthcare resources are already limited.
- Limited Awareness Among Healthcare Professionals:Despite the benefits of molecular diagnostics, there remains a significant knowledge gap among healthcare professionals in Saudi Arabia. A survey indicated that only 40% of practitioners are familiar with the latest molecular diagnostic technologies. This lack of awareness can lead to underutilization of available diagnostic tools, ultimately affecting patient care and delaying the adoption of innovative solutions in the healthcare system.
Saudi Arabia Point of Care Molecular Diagnostics Market Future Outlook
The future of the point-of-care molecular diagnostics market in Saudi Arabia appears promising, driven by ongoing technological innovations and a growing emphasis on personalized medicine. As healthcare providers increasingly adopt AI-driven diagnostic tools, the efficiency and accuracy of testing are expected to improve significantly. Furthermore, the integration of telemedicine is likely to enhance access to diagnostics, particularly in remote areas, fostering a more inclusive healthcare environment and addressing the rising demand for rapid testing solutions.
Market Opportunities
- Expansion of Telemedicine and Remote Diagnostics:The surge in telemedicine adoption presents a unique opportunity for point-of-care molecular diagnostics. With over 60% of the population now using telehealth services, integrating diagnostic testing into these platforms can enhance patient access to timely care, particularly in underserved regions, thereby driving market growth and improving health outcomes.
- Collaborations with Local Healthcare Providers:Strategic partnerships with local healthcare providers can facilitate the distribution and implementation of molecular diagnostics. By leveraging existing healthcare networks, companies can enhance their market presence and ensure that advanced diagnostic solutions reach a broader patient base, ultimately contributing to improved healthcare delivery across Saudi Arabia.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Application | Infectious Diseases Oncology Prenatal Testing Endocrinology Hematology Other Applications |
| By End-User | Hospitals Professional Diagnostic Centers Home Care Settings Research Laboratories Others |
| By Testing Mode | Prescription-based Testing OTC Testing |
| By Technology | PCR Technology Isothermal Amplification Microarray Technology Others |
| By Region | Northern and Central Region Eastern Region Western Region Southern Region |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Laboratory Service Providers
Industry Associations (e.g., Saudi Health Council)
Financial Institutions
Players Mentioned in the Report:
Abbott Laboratories
Roche Diagnostics
Qiagen N.V.
BioMerieux S.A.
Danaher Corporation
Hologic, Inc.
Bayer AG
Bio-Rad Laboratories, Inc.
Agilent Technologies, Inc.
OraSure Technologies, Inc.
Nova Biomedical
Nipro Corporation
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Point of Care Molecular Diagnostics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Point of Care Molecular Diagnostics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Point of Care Molecular Diagnostics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of infectious diseases
3.1.2 Rising demand for rapid diagnostic solutions
3.1.3 Technological advancements in molecular diagnostics
3.1.4 Government initiatives to enhance healthcare infrastructure
3.2 Market Challenges
3.2.1 High costs of advanced diagnostic equipment
3.2.2 Limited awareness among healthcare professionals
3.2.3 Regulatory hurdles in product approvals
3.2.4 Competition from traditional diagnostic methods
3.3 Market Opportunities
3.3.1 Expansion of telemedicine and remote diagnostics
3.3.2 Collaborations with local healthcare providers
3.3.3 Development of point-of-care testing for chronic diseases
3.3.4 Investment in research and development for innovative solutions
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Integration of AI in diagnostic processes
3.4.3 Growing focus on preventive healthcare
3.4.4 Increasing adoption of home-based testing kits
3.5 Government Regulation
3.5.1 Implementation of quality control standards
3.5.2 Guidelines for the approval of new diagnostic tests
3.5.3 Policies promoting local manufacturing of diagnostics
3.5.4 Regulations on data privacy and patient consent
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Point of Care Molecular Diagnostics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Point of Care Molecular Diagnostics Market Segmentation
8.1 By Application
8.1.1 Infectious Diseases
8.1.2 Oncology
8.1.3 Prenatal Testing
8.1.4 Endocrinology
8.1.5 Hematology
8.1.6 Other Applications
8.2 By End-User
8.2.1 Hospitals
8.2.2 Professional Diagnostic Centers
8.2.3 Home Care Settings
8.2.4 Research Laboratories
8.2.5 Others
8.3 By Testing Mode
8.3.1 Prescription-based Testing
8.3.2 OTC Testing
8.4 By Technology
8.4.1 PCR Technology
8.4.2 Isothermal Amplification
8.4.3 Microarray Technology
8.4.4 Others
8.5 By Region
8.5.1 Northern and Central Region
8.5.2 Eastern Region
8.5.3 Western Region
8.5.4 Southern Region
8.6 By Distribution Channel
8.6.1 Direct Sales
8.6.2 Distributors
8.6.3 Online Sales
8.6.4 Others
9. Saudi Arabia Point of Care Molecular Diagnostics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Company Size (Large, Medium, or Small)
9.2.3 Revenue Growth Rate (%)
9.2.4 Market Penetration Rate in Saudi Arabia (%)
9.2.5 Product Portfolio Breadth (Number of POC MDx Platforms)
9.2.6 Regulatory Approvals in Saudi Arabia
9.2.7 Distribution Network Strength
9.2.8 Pricing Competitiveness (Premium/Mid-tier/Value)
9.2.9 R&D Investment in Molecular Diagnostics (%)
9.2.10 Strategic Partnerships in Middle East Region
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Abbott Laboratories
9.5.2 Roche Diagnostics
9.5.3 Qiagen N.V.
9.5.4 BioMérieux S.A.
9.5.5 Danaher Corporation
9.5.6 Hologic, Inc.
9.5.7 Bayer AG
9.5.8 Bio-Rad Laboratories, Inc.
9.5.9 Agilent Technologies, Inc.
9.5.10 OraSure Technologies, Inc.
9.5.11 Nova Biomedical
9.5.12 Nipro Corporation
10. Saudi Arabia Point of Care Molecular Diagnostics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Education
10.1.3 Ministry of Defense
10.1.4 Others
10.2 Corporate Spend on Healthcare Infrastructure
10.2.1 Hospital Infrastructure Investments
10.2.2 Diagnostic Center Technology Upgrades
10.2.3 Home Care Diagnostic Equipment
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Diagnostic Labs
10.3.3 Home Care Providers
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training and Education Needs
10.4.2 Infrastructure Readiness
10.4.3 Financial Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 ROI Measurement Techniques
10.5.2 Use Case Development
10.5.3 Long-term Sustainability Plans
10.5.4 Others
11. Saudi Arabia Point of Care Molecular Diagnostics Market Future Size, 2025-2033
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from health authorities and industry associations in Saudi Arabia
- Review of published articles and white papers on molecular diagnostics and point-of-care testing
- Examination of government health policies and funding initiatives related to diagnostics
Primary Research
- Interviews with healthcare professionals, including lab managers and clinicians in hospitals
- Surveys targeting procurement officers in healthcare institutions and diagnostic companies
- Field interviews with key opinion leaders in the molecular diagnostics sector
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including industry reports and expert opinions
- Triangulation of market data with sales figures and healthcare expenditure trends
- Sanity checks conducted through expert panel reviews and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on national healthcare spending and diagnostics budget
- Segmentation of the market by product type, application, and end-user demographics
- Incorporation of growth rates from historical data and projected healthcare trends
Bottom-up Modeling
- Collection of sales data from leading point-of-care molecular diagnostic manufacturers
- Estimation of market share based on product volume and pricing strategies
- Analysis of distribution channels and their impact on market penetration
Forecasting & Scenario Analysis
- Development of forecasting models using historical growth rates and market drivers
- Scenario analysis based on potential regulatory changes and technological advancements
- Creation of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Laboratories | 100 | Laboratory Managers, Clinical Pathologists |
| Private Diagnostic Centers | 60 | Center Directors, Medical Technologists |
| Healthcare Providers | 50 | General Practitioners, Specialists |
| Government Health Agencies | 40 | Policy Makers, Health Program Coordinators |
| Research Institutions | 40 | Research Scientists, Academic Professors |
Frequently Asked Questions
What is the current value of the Saudi Arabia Point of Care Molecular Diagnostics Market?
The Saudi Arabia Point of Care Molecular Diagnostics Market is valued at approximately USD 1.1 billion, reflecting significant growth driven by the rising prevalence of infectious diseases and advancements in molecular technologies.