Region:Middle East
Author(s):Rebecca
Product Code:KRAB2053
Pages:84
Published On:January 2026
By Molecule Type:
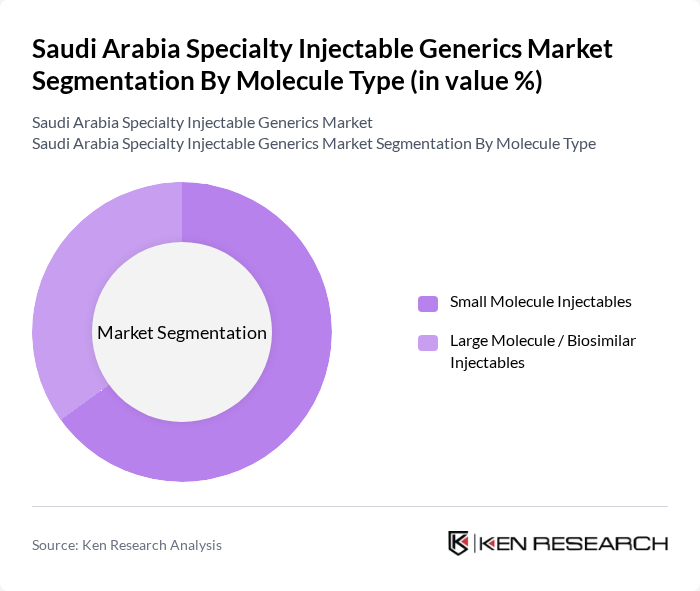
The market is segmented into two primary categories: Small Molecule Injectables and Large Molecule / Biosimilar Injectables. Small Molecule Injectables dominate the market due to their widespread use in high-volume therapeutic areas, including anti-infectives, analgesics, cardiovascular drugs, and perioperative medications, and their established cost-effectiveness, stable manufacturing processes, and broad inclusion on hospital formularies and public tenders. Large Molecule / Biosimilar Injectables are gaining traction, particularly in oncology, rheumatology, and other autoimmune diseases, as they offer advanced targeted and biologic therapies at lower cost than originators, supported by SFDA biosimilar guidelines, increasing payer acceptance, and greater inclusion of biosimilars in hospital and insurance reimbursement pathways.
By Therapeutic Class:
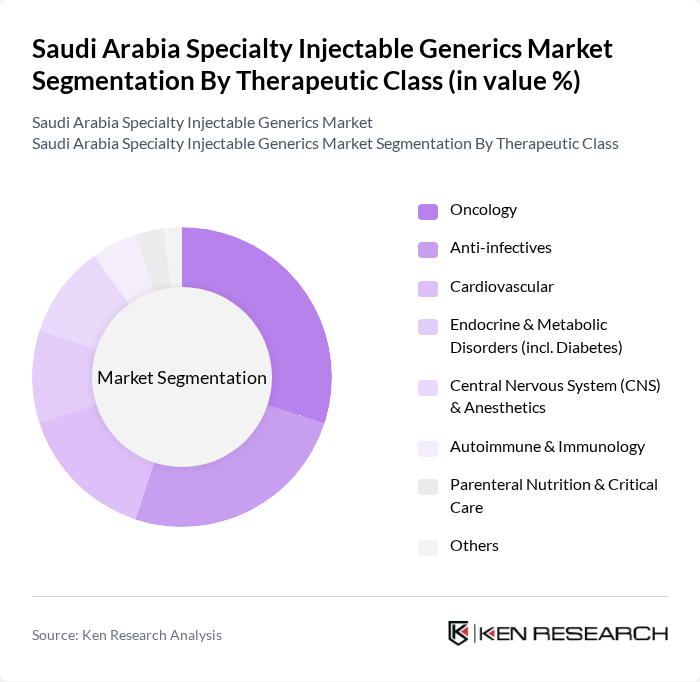
The therapeutic class segmentation includes Oncology, Anti-infectives, Cardiovascular, Endocrine & Metabolic Disorders (including Diabetes), Central Nervous System (CNS) & Anesthetics, Autoimmune & Immunology, Parenteral Nutrition & Critical Care, and Others. Oncology injectables lead the market due to the rising incidence of cancer in Saudi Arabia, increased availability of injectable chemotherapy, immunotherapy, and targeted biologics, and the expansion of oncology centers and cancer programs. Anti-infectives also hold a significant share, driven by the ongoing need for parenteral antibiotics and antifungals in hospital and critical care settings and their high utilization in public tenders. Demand for injectables in cardiovascular, diabetes, CNS, anesthetics, autoimmune and immunology, and critical care segments is growing as hospitals and payers emphasize rapid-acting parenteral therapies, broader insurance coverage for generics, and adoption of guideline-based treatment pathways to improve clinical outcomes and optimize total treatment costs.
The Saudi Arabia Specialty Injectable Generics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Saudi Pharmaceutical Industries and Medical Appliances Corporation (SPIMACO), Tabuk Pharmaceuticals, Jamjoom Pharma, Riyadh Pharma (Al Riyadh Pharmaceutical Industries Co.), AJA Pharmaceutical Industries Co., Saudi Chemical Company Holding – Saudi International Pharma (SIPH), Sudair Pharma Company, Jamjoom Medical Solutions (JMS), Gulf Pharmaceutical Industries (Julphar) – KSA Operations, Dar Al Dawa Development and Investment Company – KSA Operations, SPIMACO Addwaeih – Contract Manufacturing Partners, Global Multinational Partners (e.g., Pfizer, Novartis, GSK) – Injectable Generics/Biosimilars in KSA, Key Regional CDMOs and Fill-Finish Partners, Leading Hospital Pharmacy Groups (e.g., Dr. Sulaiman Al-Habib, Mouwasat) – High-volume Buyers, Emerging Local Sterile Injectables Start-ups and JV Platforms contribute to innovation, geographic expansion, and service delivery in this space.
The future of the specialty injectable generics market in Saudi Arabia appears promising, driven by ongoing healthcare reforms and increasing investments in research and development. As the government continues to promote generics through various initiatives, the market is likely to witness enhanced competition and innovation. Additionally, the growing trend towards personalized medicine and advanced manufacturing technologies will further shape the landscape, enabling more tailored treatment options for patients and improving overall healthcare outcomes.
| Segment | Sub-Segments |
|---|---|
| By Molecule Type | Small Molecule Injectables Large Molecule / Biosimilar Injectables |
| By Therapeutic Class | Oncology Anti-infectives Cardiovascular Endocrine & Metabolic Disorders (incl. Diabetes) Central Nervous System (CNS) & Anesthetics Autoimmune & Immunology Parenteral Nutrition & Critical Care Others |
| By Route of Administration | Intravenous (IV) Intramuscular (IM) Subcutaneous (SC) Others |
| By Container / Packaging Type | Vials Ampoules Prefilled Syringes & Pens Premix / Ready-to-use Bags Others |
| By Formulation Type | Ready-to-use Solutions Lyophilized Powders Emulsions and Lipid-based Injectables Suspensions Others |
| By End-User | Public Hospitals Private Hospitals Specialty Clinics & Day-care Centers Retail & Hospital Pharmacies Others |
| By Region | Northern & Central Region Western Region Eastern Region Southern Region |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Procurement Departments | 80 | Procurement Managers, Pharmacy Directors |
| Pharmaceutical Distributors | 70 | Sales Managers, Distribution Coordinators |
| Healthcare Professionals | 100 | Doctors, Nurses, Clinical Pharmacists |
| Patient Advocacy Groups | 50 | Patient Representatives, Healthcare Advocates |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
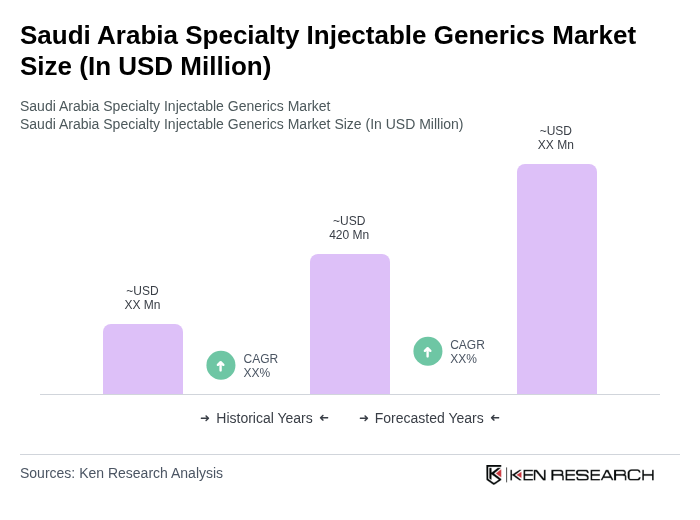
The Saudi Arabia Specialty Injectable Generics Market is valued at approximately USD 420 million, reflecting a significant growth driven by the increasing prevalence of chronic diseases, an aging population, and the demand for cost-effective treatment options.