About the Report
Base Year 2024Saudi Arabia Sterile Barrier Systems Market Overview
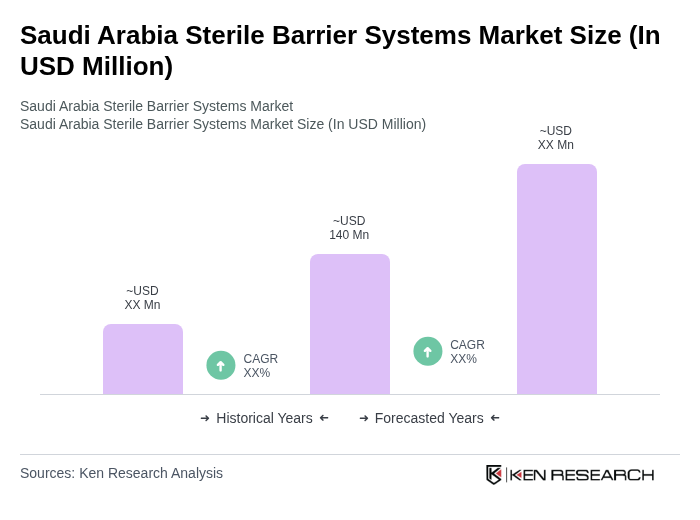
- The Saudi Arabia Sterile Barrier Systems Market is valued at USD 140 million, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for safe and effective medical packaging solutions, particularly in the healthcare sector, where the need for sterilization and contamination prevention is paramount. The market is further supported by the expansion of healthcare infrastructure, rising surgical procedures, and the localization of pharmaceutical manufacturing under Vision 2030, which emphasizes compliance with international sterility standards .
- Key cities such as Riyadh, Jeddah, and Dammam dominate the market due to their advanced healthcare infrastructure and concentration of hospitals and medical facilities. These urban centers are pivotal in driving the demand for sterile barrier systems, as they host a significant number of healthcare providers and pharmaceutical companies. The rise of medical tourism in these cities also contributes to the adoption of high-standard sterilization and packaging solutions .
- The “Medical Devices Regulation, 2021” issued by the Saudi Food and Drug Authority (SFDA) mandates that all medical packaging, including sterile barrier systems, must comply with specific sterilization standards aligned with international norms such as ISO 11607. This regulation requires manufacturers and importers to ensure product sterility, implement validated sterilization processes, and obtain SFDA certification before market entry, thereby enhancing patient safety and product quality across healthcare settings .
Saudi Arabia Sterile Barrier Systems Market Segmentation
By Product Type:
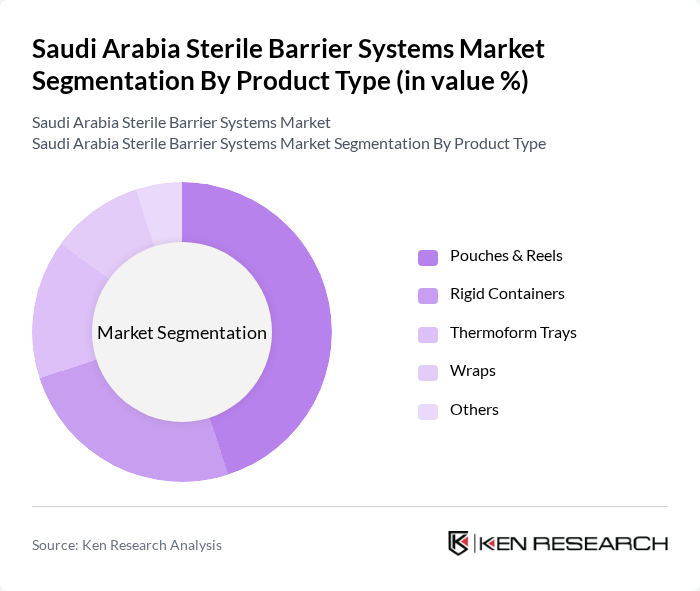
The product type segmentation includes Pouches & Reels, Rigid Containers, Thermoform Trays, Wraps, and Others. Among these, Pouches & Reels hold the largest market share due to their versatility, ease of use, and effectiveness in preventing contamination for a wide array of surgical instruments and single-use medical devices. Rigid Containers are the fastest-growing segment, favored for their durability, reusability, and ability to maintain sterility during extended storage and transportation. The increasing demand for sustainable and eco-friendly packaging solutions is also driving growth in paper-based wraps and innovative barrier materials, as healthcare providers seek to reduce environmental impact while maintaining high sterility standards .
By End-User:
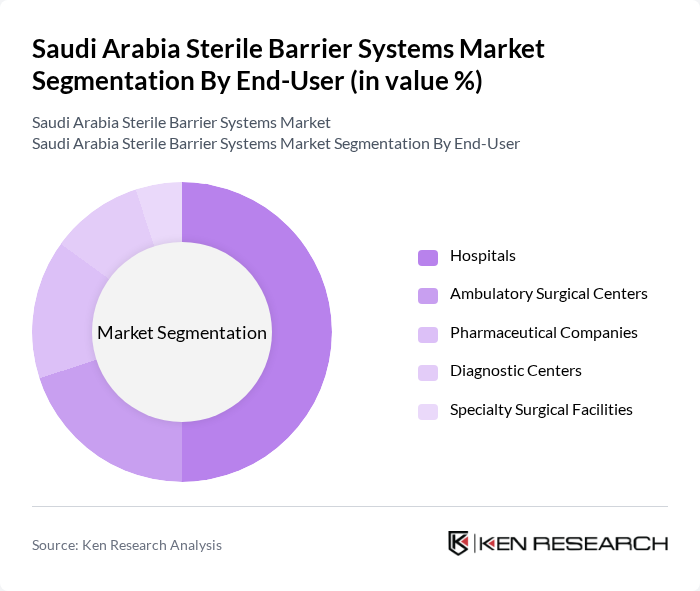
The end-user segmentation includes Hospitals, Ambulatory Surgical Centers, Pharmaceutical Companies, Diagnostic Centers, and Specialty Surgical Facilities. Hospitals remain the leading end-user, accounting for the largest market share due to their extensive use of sterile barrier systems for surgical instruments, reusable devices, and infection control protocols. Ambulatory Surgical Centers are the fastest-growing segment, propelled by the increasing volume of outpatient procedures and the need for efficient, single-use sterilization solutions. The pharmaceutical sector also drives demand, particularly for sterile packaging of pre-filled syringes, injectable drugs, and biologics, as local manufacturing expands to meet regulatory and export requirements .
Saudi Arabia Sterile Barrier Systems Market Competitive Landscape
The Saudi Arabia Sterile Barrier Systems Market is characterized by a dynamic mix of regional and international players. Leading participants such as 3M Company, STERIS Corporation, Amcor plc, Cardinal Health, Inc., DuPont de Nemours, Inc., Medline Industries, LP, Innovia Medical Company (Summit Medical LLC), Sklar Instruments, ASP (Fortive Corporation), West Pharmaceutical Services, Inc., Sealed Air Corporation, Ecolab Inc., Becton, Dickinson and Company, Terumo Corporation, Smith & Nephew plc contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia Sterile Barrier Systems Market Industry Analysis
Growth Drivers
- Increasing Demand for Healthcare Products:The healthcare sector in Saudi Arabia is projected to grow significantly, with government spending expected to reachUSD 53 billionin future. This surge in investment is driven by a growing population and an increase in chronic diseases, leading to a higher demand for sterile barrier systems. The rise in healthcare expenditure is anticipated to enhance the procurement of advanced medical devices, thereby boosting the sterile barrier systems market.
- Rising Awareness of Infection Control:The Saudi Ministry of Health has reported a 30% increase in infection control training programs since 2022. This heightened awareness among healthcare professionals and institutions is leading to a greater emphasis on using sterile barrier systems to prevent hospital-acquired infections. As hospitals adopt stricter infection control protocols, the demand for effective packaging solutions is expected to rise, further propelling market growth.
- Government Initiatives for Healthcare Infrastructure:The Saudi Vision 2030 initiative aims to enhance healthcare infrastructure, with an investment ofUSD 13.3 billionallocated for new hospitals and clinics in future. This initiative is expected to increase the number of healthcare facilities, thereby driving the demand for sterile barrier systems. The government's commitment to improving healthcare access and quality will create a favorable environment for market expansion.
Market Challenges
- High Cost of Sterile Barrier Systems:The average cost of sterile barrier systems in Saudi Arabia is approximately SAR 1,500 per unit, which poses a significant barrier for smaller healthcare facilities. This high cost can limit the adoption of advanced packaging solutions, particularly in rural areas where budgets are constrained. As a result, many facilities may opt for less effective alternatives, hindering overall market growth.
- Stringent Regulatory Requirements:Compliance with the Saudi Food and Drug Authority (SFDA) regulations can be challenging for manufacturers, as the approval process for new sterile barrier systems can take up to 18 months. This lengthy process can delay product launches and increase operational costs. Additionally, the need for continuous compliance with evolving regulations can strain resources, particularly for smaller companies in the market.
Saudi Arabia Sterile Barrier Systems Market Future Outlook
The future of the Saudi Arabia sterile barrier systems market appears promising, driven by ongoing investments in healthcare infrastructure and a growing emphasis on infection control. As the government continues to support healthcare advancements, the market is likely to witness increased adoption of innovative packaging solutions. Furthermore, the integration of smart technologies and sustainable practices will shape the industry, enhancing efficiency and environmental responsibility in packaging processes.
Market Opportunities
- Expansion of Healthcare Facilities:With the government planning to establish over 50 new healthcare facilities in future, there is a significant opportunity for sterile barrier systems manufacturers. This expansion will create a demand for high-quality packaging solutions, enabling companies to capture a larger market share and enhance their product offerings.
- Development of Eco-Friendly Packaging Solutions:As environmental concerns grow, there is an increasing demand for eco-friendly packaging in the healthcare sector. Companies that invest in developing biodegradable and recyclable sterile barrier systems can tap into this emerging market trend, appealing to environmentally conscious consumers and healthcare providers.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Pouches & Reels (45.07% market share) Rigid Containers (fastest growing segment) Thermoform Trays Wraps Others |
| By End-User | Hospitals (49.54% market share) Ambulatory Surgical Centers (fastest growing segment) Pharmaceutical Companies Diagnostic Centers Specialty Surgical Facilities |
| By Material | High-Barrier Plastics Paper-Based Materials (sustainable options) Multilayer Co-extruded Films Composite Materials Others |
| By Sterilization Method | Autoclaving Ethylene Oxide (ETO) Radiation Sterilization Others |
| By Application | Surgical Instruments Single-Use Medical Devices Pre-filled Syringes Pharmaceutical Products Others |
| By Distribution Channel | Direct Sales to Healthcare Facilities Medical Device Distributors Pharmaceutical Distributors Online Sales Others |
| By Regional Market | Central Region (Riyadh) Eastern Region (Dammam) Western Region (Jeddah) Southern Region |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Pharmaceutical Companies
Packaging Solution Providers
Industry Associations and Trade Organizations
Players Mentioned in the Report:
3M Company
STERIS Corporation
Amcor plc
Cardinal Health, Inc.
DuPont de Nemours, Inc.
Medline Industries, LP
Innovia Medical Company (Summit Medical LLC)
Sklar Instruments
ASP (Fortive Corporation)
West Pharmaceutical Services, Inc.
Sealed Air Corporation
Ecolab Inc.
Becton, Dickinson and Company
Terumo Corporation
Smith & Nephew plc
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Sterile Barrier Systems Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Sterile Barrier Systems Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Sterile Barrier Systems Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for healthcare products
3.1.2 Rising awareness of infection control
3.1.3 Government initiatives for healthcare infrastructure
3.1.4 Technological advancements in packaging
3.2 Market Challenges
3.2.1 High cost of sterile barrier systems
3.2.2 Stringent regulatory requirements
3.2.3 Limited local manufacturing capabilities
3.2.4 Competition from alternative packaging solutions
3.3 Market Opportunities
3.3.1 Expansion of healthcare facilities
3.3.2 Growth in the pharmaceutical sector
3.3.3 Increasing export potential
3.3.4 Development of eco-friendly packaging solutions
3.4 Market Trends
3.4.1 Shift towards sustainable packaging
3.4.2 Integration of smart technologies
3.4.3 Customization of packaging solutions
3.4.4 Focus on supply chain transparency
3.5 Government Regulation
3.5.1 Compliance with Saudi Food and Drug Authority (SFDA) standards
3.5.2 Implementation of ISO standards
3.5.3 Regulations on import and export of medical devices
3.5.4 Guidelines for environmental sustainability
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Sterile Barrier Systems Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Sterile Barrier Systems Market Segmentation
8.1 By Product Type
8.1.1 Pouches & Reels (45.07% market share)
8.1.2 Rigid Containers (fastest growing segment)
8.1.3 Thermoform Trays
8.1.4 Wraps
8.1.5 Others
8.2 By End-User
8.2.1 Hospitals (49.54% market share)
8.2.2 Ambulatory Surgical Centers (fastest growing segment)
8.2.3 Pharmaceutical Companies
8.2.4 Diagnostic Centers
8.2.5 Specialty Surgical Facilities
8.3 By Material
8.3.1 High-Barrier Plastics
8.3.2 Paper-Based Materials (sustainable options)
8.3.3 Multilayer Co-extruded Films
8.3.4 Composite Materials
8.3.5 Others
8.4 By Sterilization Method
8.4.1 Autoclaving
8.4.2 Ethylene Oxide (ETO)
8.4.3 Radiation Sterilization
8.4.4 Others
8.5 By Application
8.5.1 Surgical Instruments
8.5.2 Single-Use Medical Devices
8.5.3 Pre-filled Syringes
8.5.4 Pharmaceutical Products
8.5.5 Others
8.6 By Distribution Channel
8.6.1 Direct Sales to Healthcare Facilities
8.6.2 Medical Device Distributors
8.6.3 Pharmaceutical Distributors
8.6.4 Online Sales
8.6.5 Others
8.7 By Regional Market
8.7.1 Central Region (Riyadh)
8.7.2 Eastern Region (Dammam)
8.7.3 Western Region (Jeddah)
8.7.4 Southern Region
9. Saudi Arabia Sterile Barrier Systems Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Company Size Classification (Large, Medium, or Small)
9.2.3 Revenue Growth Rate (CAGR %)
9.2.4 Market Penetration Rate (%)
9.2.5 Product Portfolio Breadth (Number of SKUs)
9.2.6 Pricing Strategy (Premium/Mid-tier/Value)
9.2.7 Regulatory Certifications (ISO, FDA, SFDA)
9.2.8 Distribution Network Reach (Geographic Coverage %)
9.2.9 Innovation Index (R&D Investment as % of Revenue)
9.2.10 Customer Satisfaction Score (NPS or equivalent)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 3M Company
9.5.2 STERIS Corporation
9.5.3 Amcor plc
9.5.4 Cardinal Health, Inc.
9.5.5 DuPont de Nemours, Inc.
9.5.6 Medline Industries, LP
9.5.7 Innovia Medical Company (Summit Medical LLC)
9.5.8 Sklar Instruments
9.5.9 ASP (Fortive Corporation)
9.5.10 West Pharmaceutical Services, Inc.
9.5.11 Sealed Air Corporation
9.5.12 Ecolab Inc.
9.5.13 Becton, Dickinson and Company
9.5.14 Terumo Corporation
9.5.15 Smith & Nephew plc
10. Saudi Arabia Sterile Barrier Systems Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Defense
10.1.3 Ministry of Education
10.1.4 Ministry of Industry and Mineral Resources
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Pharmaceutical Sector Expenditure
10.2.3 Research and Development Funding
10.2.4 Equipment and Supplies Budgeting
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Pharmaceutical Companies
10.3.4 Research Laboratories
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.4.2 Technology Acceptance Levels
10.4.3 Budget Constraints
10.4.4 Regulatory Compliance Awareness
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics Tracking
10.5.2 User Feedback Mechanisms
10.5.3 Scalability of Solutions
10.5.4 Long-term Cost Savings Analysis
11. Saudi Arabia Sterile Barrier Systems Market Future Size, 2025-2033
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Customer Segmentation
1.5 Key Partnerships
1.6 Cost Structure Evaluation
1.7 Competitive Advantage Assessment
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Audience Identification
2.4 Communication Channels
2.5 Marketing Budget Allocation
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 E-commerce Integration
3.4 Logistics and Supply Chain Management
3.5 Distribution Partnerships
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
4.4 Customer Willingness to Pay
4.5 Price Sensitivity Assessment
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments Analysis
5.3 Emerging Trends Identification
5.4 Future Needs Forecasting
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Feedback Mechanisms
6.4 Relationship Management Strategies
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Customer-Centric Innovations
7.4 Competitive Differentiation
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
8.4 Marketing Campaigns
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging Considerations
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Tim
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from Saudi Arabian health authorities and medical device associations
- Review of published market studies and white papers on sterile barrier systems
- Examination of regulatory frameworks and compliance guidelines from the Saudi Food and Drug Authority (SFDA)
Primary Research
- Interviews with key opinion leaders in the healthcare sector, including hospital procurement managers
- Surveys with manufacturers and suppliers of sterile barrier systems to gather insights on market trends
- Field interviews with healthcare professionals to understand usage patterns and preferences
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including trade publications and market analytics
- Triangulation of insights from primary interviews with secondary data trends
- Sanity checks conducted through expert panel reviews comprising industry veterans
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total healthcare expenditure in Saudi Arabia as a basis for sterile barrier system demand
- Segmentation of market size by end-user categories such as hospitals, clinics, and laboratories
- Incorporation of growth rates from government healthcare initiatives and investments
Bottom-up Modeling
- Volume estimates based on sales data from leading manufacturers of sterile barrier systems
- Cost analysis derived from pricing models of various sterile barrier products
- Calculation of market size based on unit sales and average selling prices
Forecasting & Scenario Analysis
- Multi-variable forecasting using historical growth rates and projected healthcare trends
- Scenario analysis based on potential regulatory changes and technological advancements
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Procurement Departments | 45 | Procurement Managers, Supply Chain Coordinators |
| Medical Device Manufacturers | 38 | Product Managers, Sales Directors |
| Healthcare Facilities (Clinics & Labs) | 42 | Facility Managers, Clinical Directors |
| Regulatory Bodies and Compliance Officers | 28 | Regulatory Affairs Specialists, Compliance Managers |
| Healthcare Professionals (Doctors & Nurses) | 47 | Surgeons, Infection Control Specialists |
Frequently Asked Questions
What is the current value of the Saudi Arabia Sterile Barrier Systems Market?
The Saudi Arabia Sterile Barrier Systems Market is valued at approximately USD 140 million, reflecting a significant demand for effective medical packaging solutions, particularly in the healthcare sector, driven by the need for sterilization and contamination prevention.