About the Report
Base Year 2024Oman Sterile Barrier Systems Market Overview
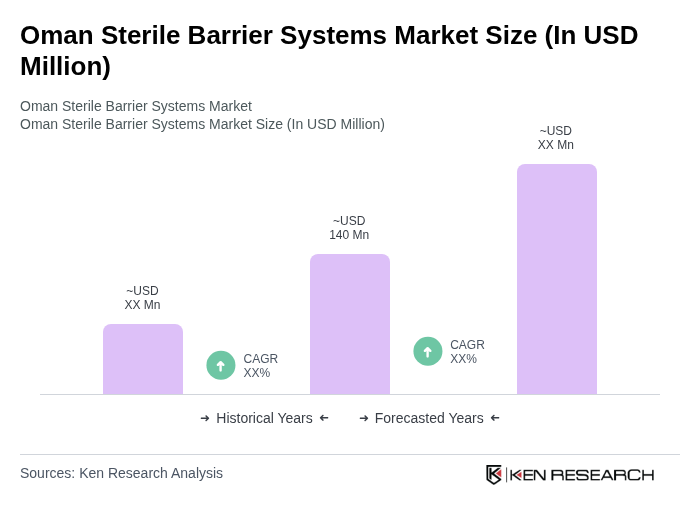
- The Oman Sterile Barrier Systems Market is valued at USD 140 million, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for safe and effective packaging solutions in the healthcare sector, particularly in hospitals and clinics, as well as the rising awareness of infection control measures. Additional market drivers include the expansion of healthcare infrastructure, adoption of advanced packaging technologies, and the rising demand for sterile pharmaceuticals and biologics, which require stringent contamination controls.
- Muscat and Salalah are the dominant cities in the Oman Sterile Barrier Systems Market due to their advanced healthcare infrastructure and concentration of medical facilities. The presence of major hospitals and surgical centers in these cities significantly contributes to the demand for sterile barrier systems, making them key players in the market.
- In 2023, the Omani government implemented the “Medical Devices and Products Packaging Regulation, 2023” issued by the Ministry of Health, Oman. This regulation mandates that all medical packaging must comply with ISO 11607 standards for sterility and safety. The regulation requires healthcare facilities to use packaging materials and systems certified for microbial barrier performance, traceability, and tamper evidence, thereby enhancing patient safety and driving market growth.
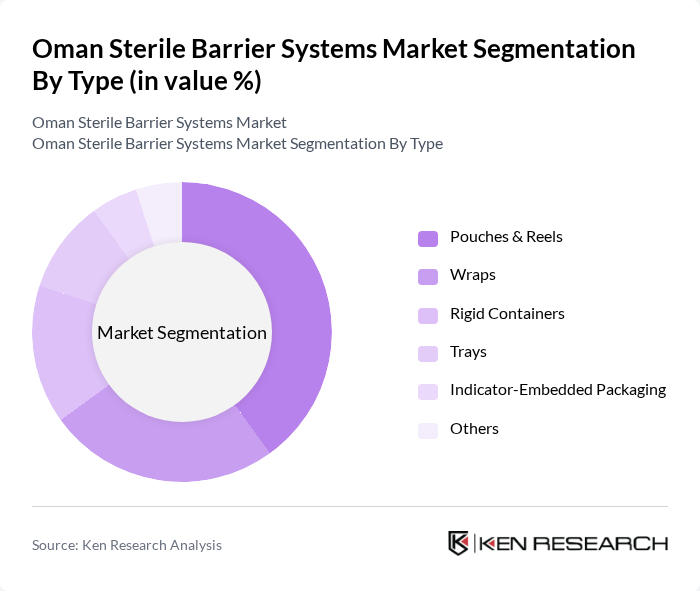
Oman Sterile Barrier Systems Market Segmentation
By Type:The market is segmented into various types of sterile barrier systems, including pouches & reels, wraps, rigid containers, trays, indicator-embedded packaging, and others. Among these, pouches & reels are the most widely used due to their versatility, ease of handling, and effectiveness in maintaining sterility. Wraps are also widely adopted, especially in surgical and procedural settings where rapid access to sterile instruments is essential. Rigid containers and trays are preferred for high-value surgical kits and sensitive items, while indicator-embedded packaging is increasingly used for compliance verification in sterilization processes.
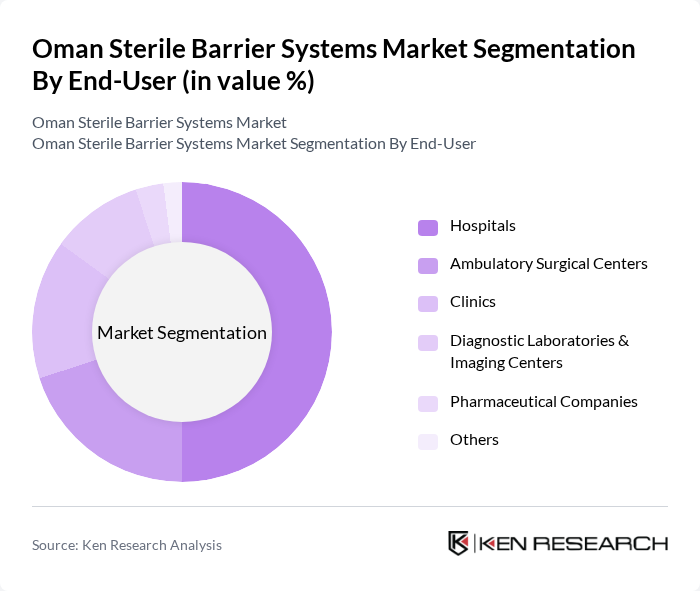
By End-User:The end-user segmentation includes hospitals, ambulatory surgical centers, clinics, diagnostic laboratories & imaging centers, pharmaceutical companies, and others. Hospitals are the leading end-users, driven by the high volume of surgical procedures, infection control protocols, and the need for reliable sterilization methods. Ambulatory surgical centers are expanding their adoption of advanced sterile barrier systems to enhance patient safety and operational efficiency. Diagnostic laboratories and imaging centers increasingly require sterile packaging for specimen collection and sensitive equipment, while pharmaceutical companies drive demand through the production of sterile drugs and biologics.
Oman Sterile Barrier Systems Market Competitive Landscape
The Oman Sterile Barrier Systems Market is characterized by a dynamic mix of regional and international players. Leading participants such as 3M Company, DuPont de Nemours, Inc., Medtronic plc, Johnson & Johnson (Ethicon), Becton, Dickinson and Company (BD), Cardinal Health, Inc., Terumo Corporation, Smith & Nephew plc, Stryker Corporation, Mölnlycke Health Care AB, Owens & Minor, Inc. (Halyard Health), Amcor plc, West Pharmaceutical Services, Inc., Sterimed Group, Steris plc contribute to innovation, geographic expansion, and service delivery in this space.
Oman Sterile Barrier Systems Market Industry Analysis
Growth Drivers
- Increasing Demand for Hygiene and Safety:The healthcare sector in Oman is experiencing a significant shift towards enhanced hygiene and safety protocols, driven by a growing awareness of infection control. In future, healthcare spending in Oman is projected to reach approximately OMR 1.65 billion, reflecting a 10% increase from the previous year. This surge in investment is expected to bolster the demand for sterile barrier systems, as healthcare facilities prioritize patient safety and infection prevention measures.
- Rising Prevalence of Hospital-Acquired Infections:The World Health Organization reported that hospital-acquired infections (HAIs) affect 7% of patients in developed countries and up to 10% in developing regions. In Oman, the incidence of HAIs is estimated at 8%, prompting healthcare providers to adopt stringent sterilization practices. This alarming trend is driving the demand for effective sterile barrier systems, as hospitals seek to mitigate infection risks and improve patient outcomes through enhanced packaging solutions.
- Technological Advancements in Packaging Materials:The sterile barrier systems market is witnessing rapid technological advancements, particularly in the development of innovative packaging materials. In future, the global market for advanced packaging materials is expected to exceed $350 billion, with a notable portion attributed to healthcare applications. These advancements are enabling the production of more effective, durable, and environmentally friendly sterile barrier systems, aligning with the growing demand for sustainable healthcare solutions in Oman.
Market Challenges
- High Costs Associated with Sterile Barrier Systems:The implementation of sterile barrier systems often incurs significant costs, which can be a barrier for many healthcare facilities in Oman. The average cost of a sterile barrier system can range from OMR 1,200 to OMR 5,500, depending on the complexity and materials used. This financial burden can deter smaller healthcare providers from adopting these essential systems, potentially compromising patient safety and infection control efforts.
- Stringent Regulatory Requirements:The sterile barrier systems market in Oman is subject to rigorous regulatory scrutiny, which can pose challenges for manufacturers and healthcare providers. Compliance with ISO standards and local healthcare regulations requires significant investment in quality assurance and testing processes. In future, the cost of compliance is projected to increase by 15%, further straining the resources of healthcare facilities and manufacturers, potentially limiting market growth.
Oman Sterile Barrier Systems Market Future Outlook
The future of the Oman sterile barrier systems market appears promising, driven by increasing healthcare investments and a heightened focus on patient safety. As the government continues to expand healthcare infrastructure, particularly in rural areas, the demand for advanced sterile barrier systems is expected to rise. Additionally, the integration of smart packaging technologies and sustainable materials will likely shape the market landscape, fostering innovation and enhancing the overall effectiveness of infection control measures in healthcare settings.
Market Opportunities
- Expansion of Healthcare Facilities in Rural Areas:The Omani government is investing heavily in healthcare infrastructure, with plans to establish over 25 new healthcare facilities in rural regions in future. This expansion presents a significant opportunity for sterile barrier system manufacturers to cater to the growing demand for infection control solutions in underserved areas, enhancing patient safety and healthcare quality.
- Development of Biodegradable Materials:The global shift towards sustainability is influencing the sterile barrier systems market, with increasing interest in biodegradable materials. In future, the biodegradable packaging market is projected to reach $25 billion. This trend presents an opportunity for manufacturers in Oman to innovate and develop eco-friendly sterile barrier systems, aligning with global sustainability goals and attracting environmentally conscious healthcare providers.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Pouches & Reels Wraps Rigid Containers Trays Indicator-Embedded Packaging Others |
| By End-User | Hospitals Ambulatory Surgical Centers Clinics Diagnostic Laboratories & Imaging Centers Pharmaceutical Companies Others |
| By Material | Plastics (e.g., Polypropylene, Polyethylene) Paper Tyvek® (High-Density Polyethylene) Foil Composites Others |
| By Application | Surgical Instruments Medical Devices Pharmaceuticals & Biologics Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | Muscat Salalah Sohar Nizwa Others |
| By Regulatory Compliance | CE Marking FDA Approval ISO Certification Gulf Cooperation Council (GCC) Regulatory Compliance Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Oman Standards and Metrology Institute)
Manufacturers and Producers of Sterile Barrier Systems
Distributors and Retailers of Medical Supplies
Healthcare Providers and Hospitals
Pharmaceutical Companies
Industry Associations (e.g., Oman Medical Association)
Financial Institutions and Banks
Players Mentioned in the Report:
3M Company
DuPont de Nemours, Inc.
Medtronic plc
Johnson & Johnson (Ethicon)
Becton, Dickinson and Company (BD)
Cardinal Health, Inc.
Terumo Corporation
Smith & Nephew plc
Stryker Corporation
Molnlycke Health Care AB
Owens & Minor, Inc. (Halyard Health)
Amcor plc
West Pharmaceutical Services, Inc.
Sterimed Group
Steris plc
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Oman Sterile Barrier Systems Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Oman Sterile Barrier Systems Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Oman Sterile Barrier Systems Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand forhygieneandsafetyin healthcare
3.1.2 Rising prevalence ofhospital-acquired infections
3.1.3 Technological advancements inpackaging materials
3.1.4 Growing healthcare infrastructure in Oman
3.2 Market Challenges
3.2.1 High costs associated withsterile barrier systems
3.2.2 Stringentregulatory requirements
3.2.3 Limited awareness among end-users
3.2.4 Competition fromnon-sterile alternatives
3.3 Market Opportunities
3.3.1 Expansion ofhealthcare facilitiesin rural areas
3.3.2 Increasing investments inmedical research
3.3.3 Development ofbiodegradable materials
3.3.4 Collaborations withinternational firms
3.4 Market Trends
3.4.1 Shift towardssustainable packaging solutions
3.4.2 Adoption ofsmart packaging technologies
3.4.3 Growing focus onpatient safety
3.4.4 Integration ofdigital technologiesin supply chains
3.5 Government Regulation
3.5.1 Compliance withISO standards
3.5.2 Implementation ofhealthcare safety regulations
3.5.3 Guidelines forsterilization processes
3.5.4 Monitoring ofimported materials
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Oman Sterile Barrier Systems Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Oman Sterile Barrier Systems Market Segmentation
8.1 By Type
8.1.1 Pouches & Reels
8.1.2 Wraps
8.1.3 Rigid Containers
8.1.4 Trays
8.1.5 Indicator-Embedded Packaging
8.1.6 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Ambulatory Surgical Centers
8.2.3 Clinics
8.2.4 Diagnostic Laboratories & Imaging Centers
8.2.5 Pharmaceutical Companies
8.2.6 Others
8.3 By Material
8.3.1 Plastics (e.g., Polypropylene, Polyethylene)
8.3.2 Paper
8.3.3 Tyvek® (High-Density Polyethylene)
8.3.4 Foil
8.3.5 Composites
8.3.6 Others
8.4 By Application
8.4.1 Surgical Instruments
8.4.2 Medical Devices
8.4.3 Pharmaceuticals & Biologics
8.4.4 Others
8.5 By Distribution Channel
8.5.1 Direct Sales
8.5.2 Distributors
8.5.3 Online Sales
8.5.4 Others
8.6 By Region
8.6.1 Muscat
8.6.2 Salalah
8.6.3 Sohar
8.6.4 Nizwa
8.6.5 Others
8.7 By Regulatory Compliance
8.7.1 CE Marking
8.7.2 FDA Approval
8.7.3 ISO Certification
8.7.4 Gulf Cooperation Council (GCC) Regulatory Compliance
8.7.5 Others
9. Oman Sterile Barrier Systems Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue (USD, Oman/Middle East)
9.2.4 Revenue Growth Rate (%)
9.2.5 Market Share (%)
9.2.6 Market Penetration Rate (Oman Healthcare Facilities Served)
9.2.7 Product Portfolio Breadth (Number of Sterile Barrier SKUs)
9.2.8 Regulatory Compliance Score (ISO, CE, FDA, GCC)
9.2.9 Distribution Network Coverage (Regions/Cities in Oman)
9.2.10 Customer Retention Rate (%)
9.2.11 Product Quality Index (Complaint Rate, Returns)
9.2.12 Pricing Competitiveness (vs. Market Average)
9.2.13 Innovation Rate (New Product Launches per Year)
9.2.14 Brand Recognition Score (Survey/Market Study)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 3M Company
9.5.2 DuPont de Nemours, Inc.
9.5.3 Medtronic plc
9.5.4 Johnson & Johnson (Ethicon)
9.5.5 Becton, Dickinson and Company (BD)
9.5.6 Cardinal Health, Inc.
9.5.7 Terumo Corporation
9.5.8 Smith & Nephew plc
9.5.9 Stryker Corporation
9.5.10 Mölnlycke Health Care AB
9.5.11 Owens & Minor, Inc. (Halyard Health)
9.5.12 Amcor plc
9.5.13 West Pharmaceutical Services, Inc.
9.5.14 Sterimed Group
9.5.15 Steris plc
10. Oman Sterile Barrier Systems Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Defense
10.1.3 Ministry of Education
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Sector Investments
10.2.2 Research and Development Expenditure
10.2.3 Facility Upgrades
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Laboratories
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training and Education Needs
10.4.2 Infrastructure Readiness
10.4.3 Financial Preparedness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics
10.5.2 User Feedback Mechanisms
10.5.3 Future Expansion Plans
10.5.4 Others
11. Oman Sterile Barrier Systems Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from health and safety organizations in Oman
- Review of market studies published by local and international research firms
- Examination of government publications and regulations related to sterile barrier systems
Primary Research
- Interviews with key stakeholders in the healthcare sector, including hospital procurement managers
- Surveys conducted with manufacturers of sterile barrier systems and packaging solutions
- Field interviews with regulatory bodies overseeing medical device compliance in Oman
Validation & Triangulation
- Cross-validation of data from multiple sources, including trade associations and market players
- Triangulation of findings from primary interviews with secondary data insights
- Sanity checks through expert panel discussions with industry veterans
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on healthcare expenditure trends in Oman
- Segmentation of the market by end-user categories such as hospitals, clinics, and laboratories
- Incorporation of growth rates from regional healthcare market forecasts
Bottom-up Modeling
- Collection of sales data from leading manufacturers of sterile barrier systems in Oman
- Estimation of market share based on production capacity and distribution channels
- Volume and pricing analysis based on product types and applications
Forecasting & Scenario Analysis
- Multi-variable forecasting using healthcare trends, population growth, and disease prevalence
- Scenario analysis based on potential regulatory changes and technological advancements
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Procurement Departments | 60 | Procurement Managers, Supply Chain Coordinators |
| Manufacturers of Sterile Barrier Systems | 40 | Product Managers, R&D Directors |
| Healthcare Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
| End-users in Clinical Settings | 50 | Clinical Managers, Infection Control Officers |
| Distributors of Medical Packaging | 40 | Sales Managers, Logistics Coordinators |
Frequently Asked Questions
What is the current value of the Oman Sterile Barrier Systems Market?
The Oman Sterile Barrier Systems Market is valued at approximately USD 140 million, reflecting a significant growth driven by the increasing demand for effective packaging solutions in the healthcare sector, particularly in hospitals and clinics.