Region:Asia
Author(s):Dev
Product Code:KRAE3093
Pages:95
Published On:March 2026
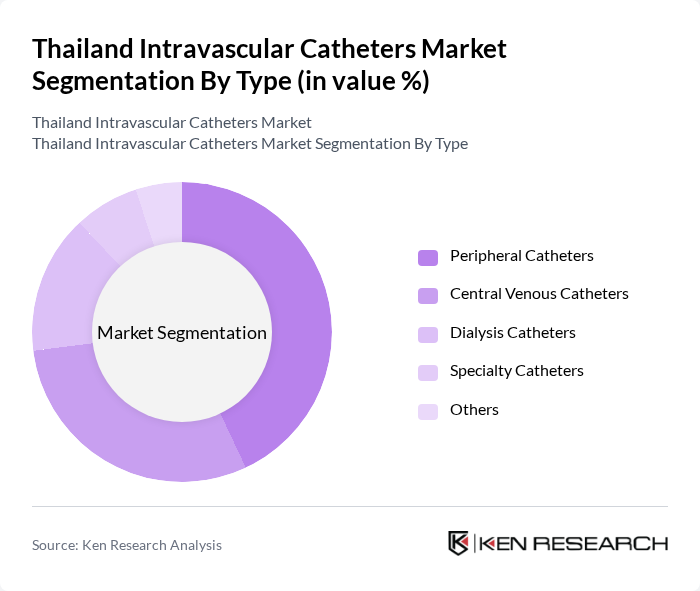
By Type:The market is segmented into various types of intravascular catheters, including Peripheral Catheters, Central Venous Catheters, Dialysis Catheters, Specialty Catheters, and Others. Among these, Peripheral Catheters are the most widely used due to their ease of insertion and lower risk of complications. Central Venous Catheters are also significant, particularly in critical care settings, where long-term access is required. The demand for Specialty Catheters is growing as healthcare providers seek tailored solutions for specific patient needs.
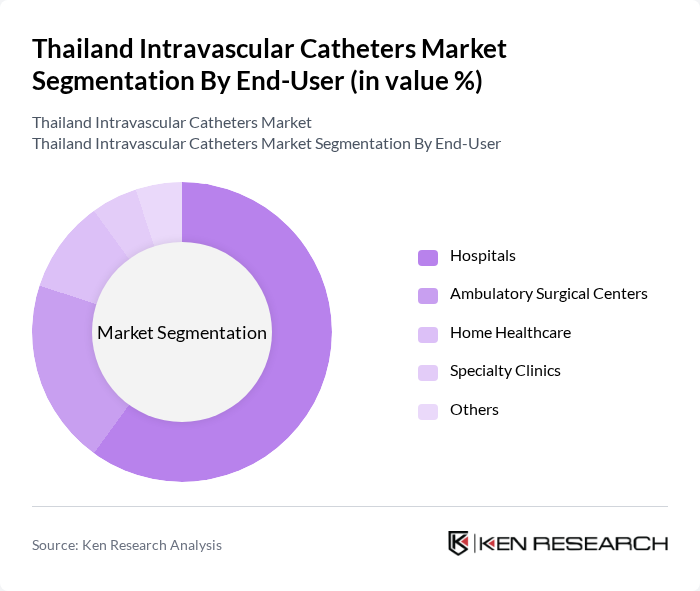
By End-User:The end-user segmentation includes Hospitals, Ambulatory Surgical Centers, Home Healthcare, Specialty Clinics, and Others. Hospitals are the primary end-users, accounting for a significant portion of the market due to the high volume of procedures requiring intravascular access. Ambulatory Surgical Centers are also gaining traction as they provide outpatient services, which increases the demand for various types of catheters. Home Healthcare is emerging as a vital segment, driven by the trend towards at-home patient care.
The Thailand Intravascular Catheters Market is characterized by a dynamic mix of regional and international players. Leading participants such as B. Braun Melsungen AG, Medtronic plc, Boston Scientific Corporation, Smiths Medical, Teleflex Incorporated, Terumo Corporation, Cook Medical, Johnson & Johnson, Cardinal Health, ConvaTec Group PLC, Fresenius Kabi AG, Halyard Health, Inc., AngioDynamics, Inc., Merit Medical Systems, Inc., Medline Industries, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the intravascular catheters market in Thailand appears promising, driven by ongoing advancements in healthcare technology and increasing healthcare investments. As the population ages, the demand for effective chronic disease management will rise, leading to greater catheter utilization. Additionally, the integration of digital health technologies is expected to enhance patient monitoring and safety, further propelling market growth. The focus on minimally invasive procedures will also create new avenues for catheter innovation and adoption in clinical practice.
| Segment | Sub-Segments |
|---|---|
| By Type | Peripheral Catheters Central Venous Catheters Dialysis Catheters Specialty Catheters Others |
| By End-User | Hospitals Ambulatory Surgical Centers Home Healthcare Specialty Clinics Others |
| By Material | Polyurethane Silicone PVC Others |
| By Application | Oncology Cardiology Gastroenterology Others |
| By Region | Bangkok Central Region Northern Region Southern Region Northeastern Region |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Pricing Model | Premium Pricing Competitive Pricing Value-Based Pricing Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Procurement Departments | 100 | Procurement Managers, Supply Chain Directors |
| Clinical Usage in Surgical Departments | 80 | Surgeons, Anesthesiologists |
| Medical Device Distributors | 60 | Sales Managers, Distribution Coordinators |
| Healthcare Policy Makers | 50 | Health Economists, Policy Analysts |
| Patient Care and Nursing Staff | 70 | Nurse Managers, Clinical Coordinators |
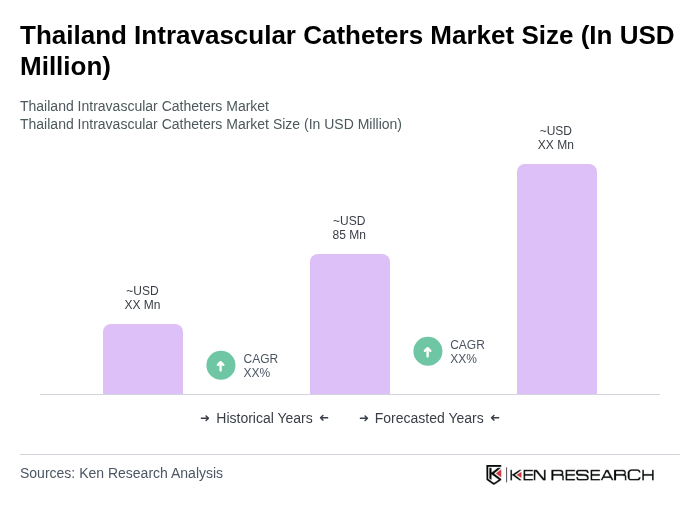
The Thailand Intravascular Catheters Market is valued at approximately USD 75 million, reflecting a five-year historical analysis. This growth is driven by factors such as the increasing prevalence of chronic diseases and advancements in medical technology.