About the Report
Base Year 2024Listen to the audio summary
UAE In Vitro Diagnostics Market Overview
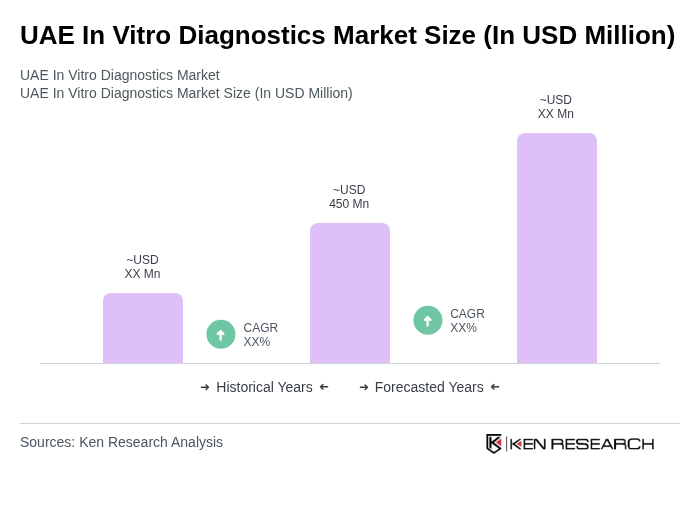
- The UAE In Vitro Diagnostics Market is valued at USD 450 million, based on a five-year historical analysis. This growth is primarily driven by the rising prevalence of chronic and infectious diseases, such as diabetes, cardiovascular conditions, and cancer, which have increased the demand for early detection and routine testing. Additionally, advancements in diagnostic technologies and the expansion of healthcare infrastructure have further propelled market growth.
- Key cities dominating the market include Abu Dhabi and Dubai, where sophisticated healthcare systems and AI-enabled laboratories enhance diagnostic adoption. The presence of advanced medical facilities and a focus on value-based healthcare contribute to the UAE's leadership in the IVD market. These emirates are also attractive for medical tourism, further boosting demand for in vitro diagnostics.
- In 2024, the UAE government announced a strategic initiative to enhance the regulatory framework for in vitro diagnostics. This includes the establishment of a unified AI-powered digital platform aimed at streamlining healthcare professional licensing across federal and local entities, thereby improving efficiency and aligning with the nation’s vision for a more integrated healthcare system.
UAE In Vitro Diagnostics Market Segmentation
By Type:
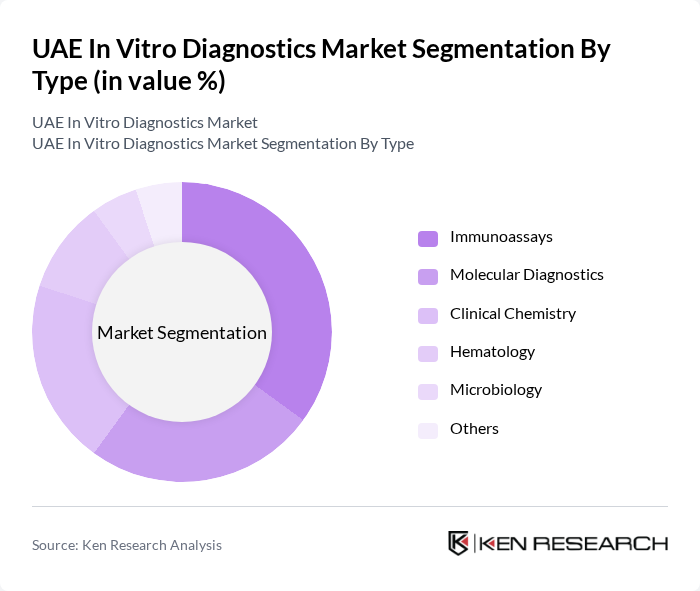
The in vitro diagnostics market is segmented into various types, including Immunoassays, Molecular Diagnostics, Clinical Chemistry, Hematology, Microbiology, and Others. Among these, Immunoassays are the leading subsegment, driven by their widespread application in disease diagnosis and monitoring. The increasing demand for rapid and accurate testing methods has led to significant advancements in immunoassay technologies, making them a preferred choice in clinical settings.
By End-User:
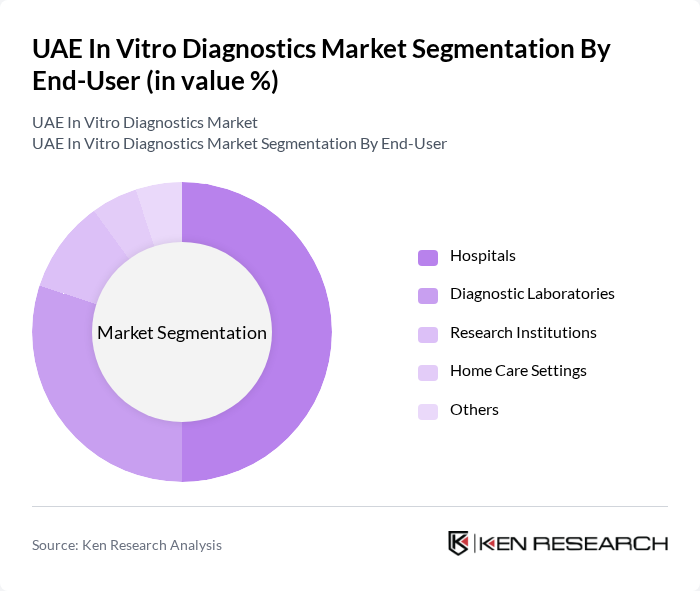
The end-user segmentation includes Hospitals, Diagnostic Laboratories, Research Institutions, Home Care Settings, and Others. Hospitals are the dominant end-user segment, accounting for a significant share of the market. This is attributed to the increasing number of hospital admissions and the growing emphasis on diagnostic testing as part of patient care protocols. Hospitals are also investing in advanced diagnostic technologies to improve patient outcomes.
UAE In Vitro Diagnostics Market Competitive Landscape
The UAE In Vitro Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Thermo Fisher Scientific, Bio-Rad Laboratories, Becton, Dickinson and Company, QIAGEN, Hologic, Inc., Ortho Clinical Diagnostics, Agilent Technologies, PerkinElmer, Inc., Sysmex Corporation, Mindray Medical International Limited, DiaSorin S.p.A., Grifols S.A. contribute to innovation, geographic expansion, and service delivery in this space.
UAE In Vitro Diagnostics Market Industry Analysis
Growth Drivers
- Robust Economic Growth & Diversification:The UAE's GDP reached **1.77 trillion dirhams** (approximately USD 481.4 billion) in future, with non-oil sectors contributing **75.5%**. This broad-based economic growth, particularly in tourism, financial services, and healthcare, enhances healthcare spending. As healthcare expenditure rises, the demand for in vitro diagnostics (IVD) tools increases, driven by the need for advanced diagnostic solutions in a diversifying economy.
- Rising Healthcare Budget Allocation:The UAE government allocated an annual healthcare budget of **USD 1.36 billion** in future, up from USD 1.33 billion in future. This increase reflects a commitment to enhancing public healthcare infrastructure, which directly supports the adoption of IVD technologies. As funding grows, healthcare facilities can invest in advanced diagnostic tools, improving patient outcomes and expanding diagnostic capabilities across the region.
- Growing Prevalence of Chronic & Infectious Diseases:The IVD market generated **USD 287.28 million** in future, driven by a significant disease burden, with nearly **30%** of the population being diabetic or pre-diabetic according to the Dubai Health Authority. This high prevalence of chronic diseases necessitates early detection and routine monitoring, thereby increasing the demand for IVD tools that facilitate timely diagnosis and management of health conditions.
Market Challenges
- High Capital Costs for Advanced IVD Systems:Advanced diagnostic systems, such as AI-driven and molecular platforms, can cost upwards of **USD 150,000 per unit**. These high capital costs pose a significant barrier to entry for smaller clinics and diagnostic centers, limiting their ability to adopt cutting-edge technologies. Consequently, this restricts the overall growth of the IVD market, as many facilities cannot afford the latest diagnostic equipment.
- Shortage of Specialized Diagnostic Workforce:As of future, the UAE employed just over **4,000** healthcare professionals specializing in diagnostics. This shortage of skilled personnel in areas such as molecular diagnostics and bioinformatics hampers the scalability of the IVD market. The lack of trained professionals limits the effective implementation and operation of advanced diagnostic technologies, thereby constraining market growth potential.
UAE In Vitro Diagnostics Market Future Outlook
The future of the UAE In Vitro Diagnostics market appears promising, driven by advancements in AI and genomics that enhance diagnostic accuracy and efficiency. The integration of AI in diagnostic processes is expected to streamline workflows, while the expansion of point-of-care testing and home-based diagnostic kits will increase accessibility. Additionally, ongoing investments in healthcare infrastructure and technology will further support the growth of the IVD market, positioning the UAE as a leader in innovative diagnostic solutions.
Market Opportunities
- Rapidly Growing Molecular Diagnostics Segment:The molecular diagnostics segment is projected to grow at **7.59% CAGR**, driven by increasing demand for precision medicine and genomic initiatives. This growth presents significant opportunities for companies to develop and market advanced diagnostic solutions tailored to the needs of the UAE population.
- Software & Service Solutions Integration:The software and service segment is expected to grow at **9.35% CAGR**, the fastest among product categories. This growth offers opportunities for companies to provide middleware, AI analytics, and interoperability solutions, enhancing the overall efficiency and effectiveness of diagnostic processes in the UAE healthcare system.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Immunoassays Molecular Diagnostics Clinical Chemistry Hematology Microbiology Others |
| By End-User | Hospitals Diagnostic Laboratories Research Institutions Home Care Settings Others |
| By Application | Infectious Diseases Cancer Diagnostics Cardiovascular Diseases Genetic Testing Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Technology | PCR Technology Next-Generation Sequencing Microarray Technology Others |
| By Sample Type | Blood Samples Urine Samples Tissue Samples Saliva Samples Others |
| By Region | Abu Dhabi Dubai Sharjah Ajman Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health and Prevention, Dubai Health Authority)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Laboratory Service Providers
Industry Associations (e.g., UAE Medical Device Association)
Financial Institutions
Players Mentioned in the Report:
Roche Diagnostics
Abbott Laboratories
Siemens Healthineers
Thermo Fisher Scientific
Bio-Rad Laboratories
Becton, Dickinson and Company
QIAGEN
Hologic, Inc.
Ortho Clinical Diagnostics
Agilent Technologies
PerkinElmer, Inc.
Sysmex Corporation
Mindray Medical International Limited
DiaSorin S.p.A.
Grifols S.A.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. UAE In Vitro Diagnostics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 UAE In Vitro Diagnostics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. UAE In Vitro Diagnostics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of chronic diseases
3.1.2 Rising demand for early diagnosis
3.1.3 Technological advancements in diagnostic tools
3.1.4 Government initiatives to enhance healthcare infrastructure
3.2 Market Challenges
3.2.1 High costs of advanced diagnostic equipment
3.2.2 Stringent regulatory requirements
3.2.3 Limited reimbursement policies
3.2.4 Competition from alternative diagnostic methods
3.3 Market Opportunities
3.3.1 Expansion of telemedicine and remote diagnostics
3.3.2 Growing focus on personalized medicine
3.3.3 Increasing investment in R&D
3.3.4 Collaborations with international diagnostic firms
3.4 Market Trends
3.4.1 Shift towards point-of-care testing
3.4.2 Integration of AI in diagnostic processes
3.4.3 Rise in home-based diagnostic kits
3.4.4 Emphasis on sustainable and eco-friendly practices
3.5 Government Regulation
3.5.1 Implementation of new health technology assessment frameworks
3.5.2 Updates to medical device registration processes
3.5.3 Enhanced guidelines for clinical laboratory standards
3.5.4 Regulations on data privacy and patient consent
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. UAE In Vitro Diagnostics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. UAE In Vitro Diagnostics Market Segmentation
8.1 By Type
8.1.1 Immunoassays
8.1.2 Molecular Diagnostics
8.1.3 Clinical Chemistry
8.1.4 Hematology
8.1.5 Microbiology
8.1.6 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Diagnostic Laboratories
8.2.3 Research Institutions
8.2.4 Home Care Settings
8.2.5 Others
8.3 By Application
8.3.1 Infectious Diseases
8.3.2 Cancer Diagnostics
8.3.3 Cardiovascular Diseases
8.3.4 Genetic Testing
8.3.5 Others
8.4 By Distribution Channel
8.4.1 Direct Sales
8.4.2 Distributors
8.4.3 Online Sales
8.4.4 Others
8.5 By Technology
8.5.1 PCR Technology
8.5.2 Next-Generation Sequencing
8.5.3 Microarray Technology
8.5.4 Others
8.6 By Sample Type
8.6.1 Blood Samples
8.6.2 Urine Samples
8.6.3 Tissue Samples
8.6.4 Saliva Samples
8.6.5 Others
8.7 By Region
8.7.1 Abu Dhabi
8.7.2 Dubai
8.7.3 Sharjah
8.7.4 Ajman
8.7.5 Others
9. UAE In Vitro Diagnostics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Product Innovation Rate
9.2.8 Distribution Efficiency
9.2.9 Brand Recognition
9.2.10 Customer Satisfaction Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Roche Diagnostics
9.5.2 Abbott Laboratories
9.5.3 Siemens Healthineers
9.5.4 Thermo Fisher Scientific
9.5.5 Bio-Rad Laboratories
9.5.6 Becton, Dickinson and Company
9.5.7 QIAGEN
9.5.8 Hologic, Inc.
9.5.9 Ortho Clinical Diagnostics
9.5.10 Agilent Technologies
9.5.11 PerkinElmer, Inc.
9.5.12 Sysmex Corporation
9.5.13 Mindray Medical International Limited
9.5.14 DiaSorin S.p.A.
9.5.15 Grifols S.A.
10. UAE In Vitro Diagnostics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Preferred Suppliers
10.1.3 Procurement Processes
10.1.4 Evaluation Criteria
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Diagnostic Facilities
10.2.2 Spending on Technology Upgrades
10.2.3 Budget for Training and Development
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges in Supply Chain Management
10.3.2 Issues with Equipment Maintenance
10.3.3 Demand for Faster Turnaround Times
10.4 User Readiness for Adoption
10.4.1 Awareness of New Technologies
10.4.2 Training Needs
10.4.3 Infrastructure Readiness
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Metrics for Success
10.5.2 Opportunities for Upselling
10.5.3 Long-term Value Assessment
11. UAE In Vitro Diagnostics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from healthcare authorities in the UAE
- Review of published articles and white papers on in vitro diagnostics trends
- Examination of regulatory frameworks and guidelines from the UAE Ministry of Health
Primary Research
- Interviews with laboratory managers and directors in hospitals and clinics
- Surveys with key opinion leaders in the diagnostics field
- Focus groups with healthcare professionals to understand user needs and preferences
Validation & Triangulation
- Cross-validation of findings with industry reports and expert opinions
- Triangulation of data from primary interviews and secondary sources
- Sanity checks through feedback from a panel of healthcare experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure data
- Segmentation of the market by product type and application area
- Incorporation of growth rates from government health initiatives and funding
Bottom-up Modeling
- Collection of sales data from leading in vitro diagnostics manufacturers
- Estimation of market penetration rates for various diagnostic tests
- Volume and pricing analysis based on historical sales trends
Forecasting & Scenario Analysis
- Multi-variable forecasting using demographic and epidemiological data
- Scenario analysis based on potential regulatory changes and technological advancements
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Laboratories | 100 | Laboratory Managers, Quality Control Officers |
| Hospitals and Healthcare Facilities | 80 | Healthcare Administrators, Medical Directors |
| Diagnostic Equipment Suppliers | 60 | Sales Managers, Product Specialists |
| Research Institutions | 50 | Research Scientists, Lab Technicians |
| Regulatory Bodies | 30 | Regulatory Affairs Managers, Compliance Officers |
Frequently Asked Questions
What is the current value of the UAE In Vitro Diagnostics Market?
The UAE In Vitro Diagnostics Market is valued at approximately USD 450 million, reflecting significant growth driven by the rising prevalence of chronic and infectious diseases, advancements in diagnostic technologies, and the expansion of healthcare infrastructure.