Region:Middle East
Author(s):Rebecca
Product Code:KRAD1505
Pages:83
Published On:November 2025
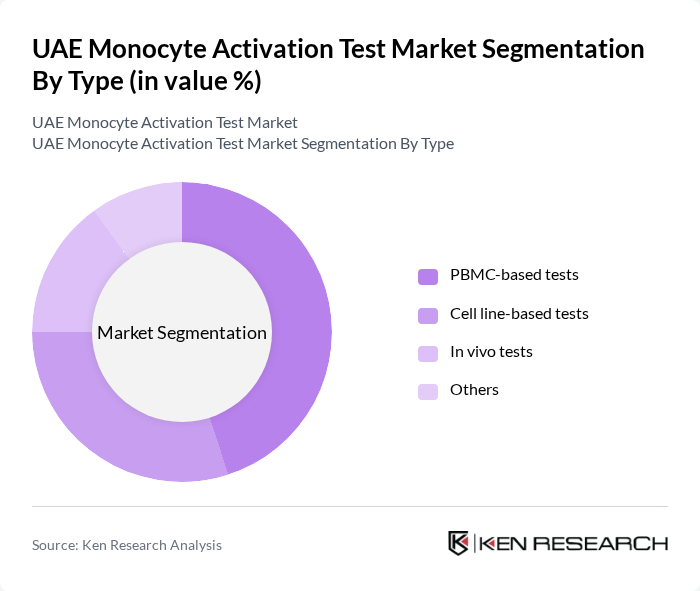
By Type:The market is segmented into PBMC-based tests, cell line-based tests, in vivo tests, and others. PBMC-based tests are gaining traction due to their reliability and relevance in assessing immune responses, providing a physiologically accurate model for pyrogen detection in vaccines and biologics. The increasing focus on personalized medicine and the need for accurate immunotoxicity testing are driving the demand for these tests, as they offer human-relevant results and faster turnaround times compared to traditional animal-based assays.
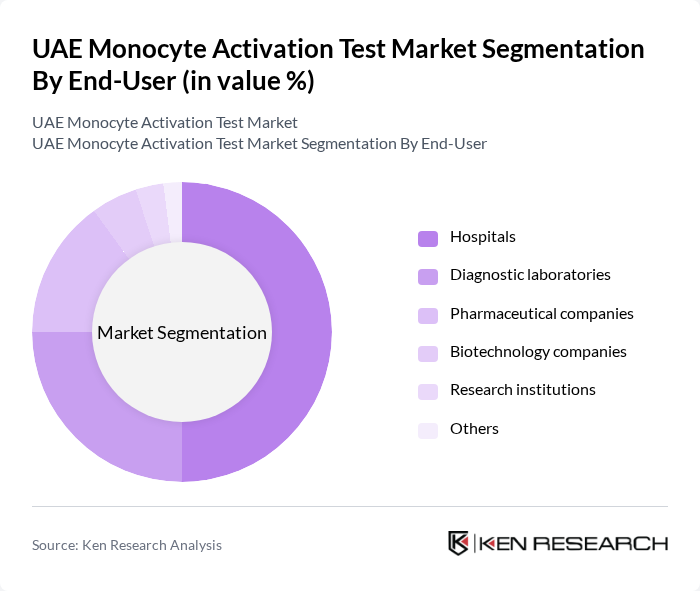
By End-User:The end-user segmentation includes hospitals, diagnostic laboratories, pharmaceutical companies, biotechnology companies, research institutions, and others. Hospitals are the leading end-users due to their extensive testing requirements and the increasing number of patients seeking advanced diagnostic services. The growing trend of personalized medicine is also influencing hospitals to adopt more sophisticated testing methods. Pharmaceutical companies are also major users, relying on MAT for drug development and safety testing, while biotechnology firms are rapidly increasing adoption due to regulatory demand for human-relevant assays.
The UAE Monocyte Activation Test Market is characterized by a dynamic mix of regional and international players. Leading participants such as Bio-Rad Laboratories, Thermo Fisher Scientific, Merck KGaA, Charles River Laboratories, Lonza Group, Sanquin Diagnostiek B.V., Becton, Dickinson and Company, Abcam plc, Promega Corporation, QIAGEN N.V., Bio-Techne Corporation, F. Hoffmann-La Roche AG, Siemens Healthineers, Illumina, Inc., Agilent Technologies, Cell Technology Ltd. (CTL) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the UAE Monocyte Activation Test market appears promising, driven by ongoing advancements in healthcare technology and increasing government support for innovative diagnostic solutions. As the healthcare infrastructure expands, the integration of artificial intelligence and automation in testing processes is expected to enhance efficiency and accuracy. Furthermore, the growing emphasis on personalized medicine will likely create a robust demand for specialized tests, positioning the Monocyte Activation Test as a critical tool in managing chronic diseases effectively.
| Segment | Sub-Segments |
|---|---|
| By Type | PBMC-based tests Cell line-based tests In vivo tests Others |
| By End-User | Hospitals Diagnostic laboratories Pharmaceutical companies Biotechnology companies Research institutions Others |
| By Application | Drug development Clinical diagnostics Vaccine development Medical device testing Others |
| By Region | Abu Dhabi Dubai Sharjah Others |
| By Technology | Flow cytometry ELISA PCR High-throughput screening Others |
| By Investment Source | Private investments Government funding International grants Others |
| By Policy Support | Research grants Tax incentives Subsidies for testing Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Laboratories | 60 | Laboratory Managers, Quality Control Officers |
| Healthcare Providers | 50 | Physicians, Nurse Practitioners |
| Biotechnology Firms | 40 | Research Scientists, Product Development Managers |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Academic Institutions | 50 | Professors, Research Fellows in Immunology |
The UAE Monocyte Activation Test market is valued at approximately USD 1 million, reflecting its share in a global market exceeding USD 590 million. This valuation is based on a five-year historical analysis of market trends and growth factors.