About the Report
Base Year 2024UAE Neurology Clinical Trials Market Overview
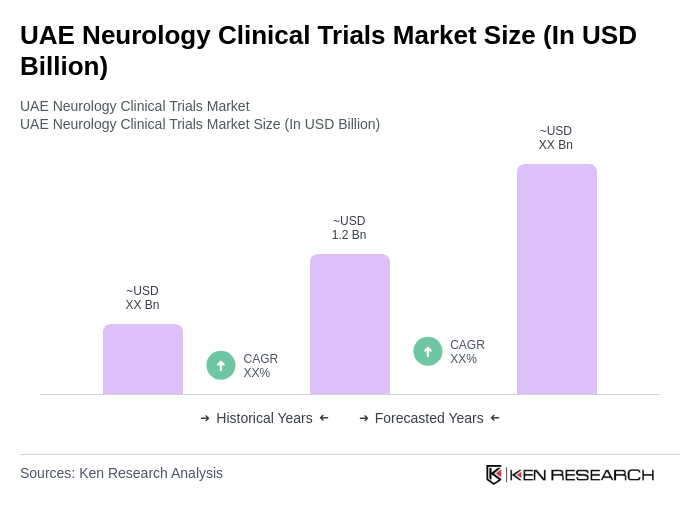
- The UAE Neurology Clinical Trials Market is valued at USD 1.2 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of neurological disorders, advancements in clinical research methodologies, and a growing focus on personalized medicine. The rising demand for innovative therapies and the establishment of robust healthcare infrastructure further contribute to the market's expansion. The UAE clinical research sector demonstrates strong performance, with pharmaceutical companies significantly investing in studies, particularly Phase III and Phase IV trials alongside observational studies. Enhanced tertiary care capacity, improved imaging and EEG capabilities, and streamlined approval processes ranging from 30 to 90 days have strengthened the market position.
- Key cities dominating the market include Dubai and Abu Dhabi, which are recognized for their advanced healthcare facilities and research institutions. The presence of international pharmaceutical companies and a supportive regulatory environment in these cities enhance their attractiveness for conducting clinical trials, making them pivotal hubs for neurology research in the region. Dubai and Abu Dhabi host specialized neuroscience units with robust Institutional Review Board and ethics processes, facilitating feasibility assessments through improved data interoperability. Growing global-local partnerships, site accreditation programs, and enhanced Good Clinical Practice compliance have accelerated trial initiation and site utilization across these metropolitan centers.
- The Health Authority Abu Dhabi's medical research section promotes world-class research that improves health and quality of life while ensuring ethical conduct of research and protection of human subjects, issued by the Health Authority Abu Dhabi (HAAD) in 2013. This framework encompasses comprehensive oversight of clinical trial approvals, ethical review requirements, informed consent protocols, and patient safety monitoring standards. The regulatory structure mandates institutional review board approval for all interventional studies, establishes clear timelines for study authorization, and enforces adherence to international research ethics guidelines, thereby fostering a conducive environment for research and development in the neurology sector.
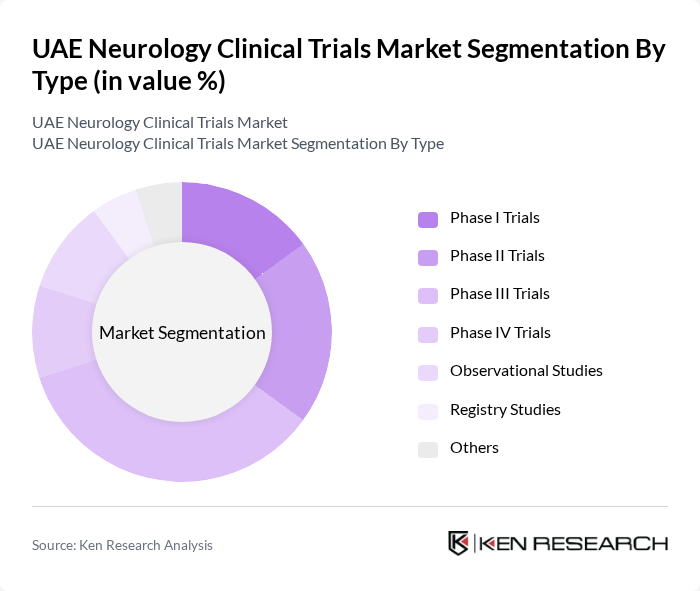
UAE Neurology Clinical Trials Market Segmentation
By Type:The market is segmented into various types of clinical trials, including Phase I Trials, Phase II Trials, Phase III Trials, Phase IV Trials, Observational Studies, Registry Studies, and Others. Each of these trial phases plays a crucial role in the drug development process, with Phase III Trials being particularly significant due to their focus on efficacy and safety in larger populations. Phase III trials dominate the UAE market, accounting for the largest segment with substantial revenue share, reflecting the country's capacity to conduct large-scale efficacy studies. The UAE has witnessed increased adoption of decentralized and hybrid trial models utilizing eConsent, home-based assessments, wearables, and imaging biomarkers, resulting in shorter cycle times and improved patient retention rates.
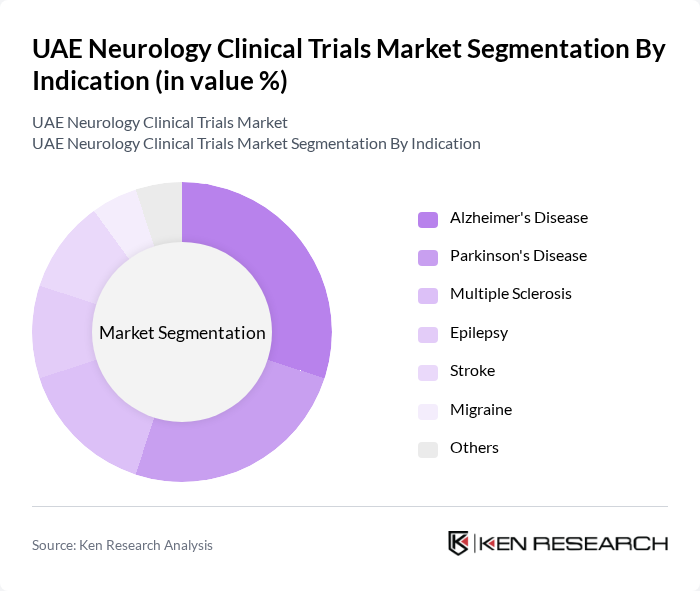
By Indication:The market is also segmented by indications, including Alzheimer's Disease, Parkinson's Disease, Multiple Sclerosis, Epilepsy, Stroke, Migraine, and Others. Alzheimer's Disease and Parkinson's Disease are particularly prominent due to their high prevalence and the urgent need for effective treatments, driving significant research efforts in these areas. Alzheimer's Disease holds particular importance in the UAE, with the country ranked second globally for the highest percentage of people suffering from dementia according to a Lancet study published in 2022, significantly boosting demand for dementia research and clinical trials in neurological disorders. Regional activity predominantly focuses on Phase II trials for neurodegeneration and pain, while Phase III programs have emerged for multiple sclerosis, epilepsy, and migraine, with registries for device-assisted stroke and movement disorders also prominent.
UAE Neurology Clinical Trials Market Competitive Landscape
The UAE Neurology Clinical Trials Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abu Dhabi Clinical Trials Center, Dubai Health Authority, Gulf Clinical Research, Al Ain Hospital, Emirates Clinical Research Institute, Medcare Hospitals, Cleveland Clinic Abu Dhabi, Sheikh Khalifa Medical City, Dubai Science Park, Abu Dhabi University, University of Sharjah, RAK Hospital, Mediclinic City Hospital, Aster DM Healthcare, NMC Healthcare contribute to innovation, geographic expansion, and service delivery in this space.
UAE Neurology Clinical Trials Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Neurological Disorders:The UAE has witnessed a significant rise in neurological disorders, with an estimated 1.6 million individuals affected by conditions such as epilepsy, multiple sclerosis, and Alzheimer's disease. According to the World Health Organization, the prevalence of dementia in the UAE is projected to reach 1.6% of the population in future, highlighting the urgent need for clinical trials to develop effective treatments and interventions.
- Rising Investment in Research and Development:The UAE government allocated approximately AED 1.5 billion (USD 408 million) for health research and innovation in future. This investment aims to enhance the country's capabilities in clinical research, particularly in neurology. The establishment of research centers and partnerships with universities is expected to foster innovation and attract global pharmaceutical companies to conduct trials in the region.
- Advancements in Technology and Methodologies:The integration of advanced technologies, such as artificial intelligence and machine learning, is revolutionizing clinical trials in the UAE. In future, it is estimated that 35% of neurology trials will utilize AI for patient recruitment and data analysis, significantly improving efficiency and accuracy. This technological shift is expected to enhance trial outcomes and attract more sponsors to the UAE market.
Market Challenges
- Regulatory Hurdles and Compliance Issues:Navigating the regulatory landscape in the UAE can be complex, with stringent requirements for clinical trial approvals. In future, the average time for obtaining necessary approvals is projected to be around 6-12 months, which can delay trial initiation. These regulatory challenges may deter some international sponsors from conducting trials in the region, impacting overall market growth.
- High Costs Associated with Clinical Trials:The financial burden of conducting clinical trials in the UAE is significant, with costs averaging AED 2.1 million (USD 572,000) per trial. This high expenditure can limit the number of trials initiated, particularly for smaller biotech firms. Additionally, the need for specialized facilities and trained personnel further escalates costs, posing a challenge for market entry and sustainability.
UAE Neurology Clinical Trials Market Future Outlook
The future of the UAE neurology clinical trials market appears promising, driven by increasing investments in healthcare innovation and a growing focus on patient-centric trial designs. As the prevalence of neurological disorders continues to rise, the demand for effective treatments will propel research initiatives. Furthermore, the integration of telemedicine and decentralized trial methodologies is expected to enhance patient recruitment and retention, ultimately leading to more successful trial outcomes and a robust clinical research environment.
Market Opportunities
- Expansion of Telemedicine and Remote Trials:The rise of telemedicine in the UAE presents a unique opportunity for conducting remote clinical trials. In future, it is anticipated that 30% of neurology trials will incorporate telehealth solutions, allowing for broader patient access and improved data collection, thus enhancing trial efficiency and reducing costs.
- Collaborations with International Research Organizations:The UAE's strategic location and investment in healthcare make it an attractive hub for international collaborations. In future, partnerships with global research organizations are expected to increase by 25%, facilitating knowledge exchange and access to advanced clinical trial methodologies, ultimately benefiting the local neurology research landscape.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Phase I Trials Phase II Trials Phase III Trials Phase IV Trials Observational Studies Registry Studies Others |
| By Indication | Alzheimer's Disease Parkinson's Disease Multiple Sclerosis Epilepsy Stroke Migraine Others |
| By Phase | Early Phase Trials Late Phase Trials Post-Marketing Studies Others |
| By Geography | Abu Dhabi Dubai Sharjah Ajman Ras Al Khaimah Fujairah Others |
| By Sponsor Type | Pharmaceutical Companies Biotechnology Firms Academic Institutions Government Agencies Contract Research Organizations (CROs) Others |
| By Patient Demographics | Age Group Gender Socioeconomic Status Others |
| By Funding Source | Public Funding Private Funding Non-Profit Organizations Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health and Prevention, Dubai Health Authority)
Pharmaceutical Companies
Biotechnology Firms
Clinical Research Organizations (CROs)
Medical Device Manufacturers
Health Insurance Providers
Patient Advocacy Groups
Players Mentioned in the Report:
Abu Dhabi Clinical Trials Center
Dubai Health Authority
Gulf Clinical Research
Al Ain Hospital
Emirates Clinical Research Institute
Medcare Hospitals
Cleveland Clinic Abu Dhabi
Sheikh Khalifa Medical City
Dubai Science Park
Abu Dhabi University
University of Sharjah
RAK Hospital
Mediclinic City Hospital
Aster DM Healthcare
NMC Healthcare
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. UAE Neurology Clinical Trials Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 UAE Neurology Clinical Trials Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. UAE Neurology Clinical Trials Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of neurological disorders
3.1.2 Rising investment in research and development
3.1.3 Government support for clinical research
3.1.4 Advancements in technology and methodologies
3.2 Market Challenges
3.2.1 Regulatory hurdles and compliance issues
3.2.2 High costs associated with clinical trials
3.2.3 Limited patient recruitment and retention
3.2.4 Competition from other regions
3.3 Market Opportunities
3.3.1 Expansion of telemedicine and remote trials
3.3.2 Collaborations with international research organizations
3.3.3 Development of personalized medicine approaches
3.3.4 Growing demand for innovative therapies
3.4 Market Trends
3.4.1 Increased focus on patient-centric trial designs
3.4.2 Utilization of artificial intelligence in data analysis
3.4.3 Shift towards decentralized clinical trials
3.4.4 Emphasis on real-world evidence in trial outcomes
3.5 Government Regulation
3.5.1 Clinical trial registration requirements
3.5.2 Ethical guidelines for human subject research
3.5.3 Data protection and privacy regulations
3.5.4 Approval processes for investigational drugs
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. UAE Neurology Clinical Trials Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. UAE Neurology Clinical Trials Market Segmentation
8.1 By Type
8.1.1 Phase I Trials
8.1.2 Phase II Trials
8.1.3 Phase III Trials
8.1.4 Phase IV Trials
8.1.5 Observational Studies
8.1.6 Registry Studies
8.1.7 Others
8.2 By Indication
8.2.1 Alzheimer's Disease
8.2.2 Parkinson's Disease
8.2.3 Multiple Sclerosis
8.2.4 Epilepsy
8.2.5 Stroke
8.2.6 Migraine
8.2.7 Others
8.3 By Phase
8.3.1 Early Phase Trials
8.3.2 Late Phase Trials
8.3.3 Post-Marketing Studies
8.3.4 Others
8.4 By Geography
8.4.1 Abu Dhabi
8.4.2 Dubai
8.4.3 Sharjah
8.4.4 Ajman
8.4.5 Ras Al Khaimah
8.4.6 Fujairah
8.4.7 Others
8.5 By Sponsor Type
8.5.1 Pharmaceutical Companies
8.5.2 Biotechnology Firms
8.5.3 Academic Institutions
8.5.4 Government Agencies
8.5.5 Contract Research Organizations (CROs)
8.5.6 Others
8.6 By Patient Demographics
8.6.1 Age Group
8.6.2 Gender
8.6.3 Socioeconomic Status
8.6.4 Others
8.7 By Funding Source
8.7.1 Public Funding
8.7.2 Private Funding
8.7.3 Non-Profit Organizations
8.7.4 Others
9. UAE Neurology Clinical Trials Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Clinical Trial Success Rate
9.2.6 Patient Recruitment Rate
9.2.7 Pricing Strategy
9.2.8 Customer Satisfaction Score
9.2.9 Regulatory Compliance Rate
9.2.10 Innovation Index
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Abu Dhabi Clinical Trials Center
9.5.2 Dubai Health Authority
9.5.3 Gulf Clinical Research
9.5.4 Al Ain Hospital
9.5.5 Emirates Clinical Research Institute
9.5.6 Medcare Hospitals
9.5.7 Cleveland Clinic Abu Dhabi
9.5.8 Sheikh Khalifa Medical City
9.5.9 Dubai Science Park
9.5.10 Abu Dhabi University
9.5.11 University of Sharjah
9.5.12 RAK Hospital
9.5.13 Mediclinic City Hospital
9.5.14 Aster DM Healthcare
9.5.15 NMC Healthcare
10. UAE Neurology Clinical Trials Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health and Prevention
10.1.2 Ministry of Education
10.1.3 Ministry of Community Development
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Clinical Research Facilities
10.2.2 Funding for Neurology Research Initiatives
10.2.3 Partnerships with Private Sector
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Healthcare Providers
10.3.2 Research Institutions
10.3.3 Pharmaceutical Companies
10.3.4 Patients
10.4 User Readiness for Adoption
10.4.1 Awareness of Clinical Trials
10.4.2 Accessibility of Trial Information
10.4.3 Willingness to Participate
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Evaluation of Trial Outcomes
10.5.2 Long-term Patient Follow-up
10.5.3 Expansion into New Indications
10.5.4 Others
11. UAE Neurology Clinical Trials Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Model Structuring
1.4 Key Partnerships and Alliances
1.5 Customer Segmentation
1.6 Cost Structure Analysis
1.7 Competitive Advantage Assessment
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Audience Identification
2.4 Communication Strategy
2.5 Digital Marketing Approaches
2.6 Event Participation and Sponsorships
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 Online Distribution Channels
3.4 Partnerships with Healthcare Providers
3.5 Logistics and Supply Chain Management
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
4.4 Customer Willingness to Pay
5. Unmet Demand & Latent Needs
5.1 Category Gaps Identification
5.2 Consumer Segments Analysis
5.3 Emerging Trends and Preferences
5.4 Future Needs Forecasting
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Feedback Mechanisms
6.4 Community Engagement Initiatives
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Unique Selling Points
7.4 Customer-Centric Innovations
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
8.4 Training and Development
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published clinical trial registries and databases specific to neurology in the UAE
- Review of government health reports and publications from the UAE Ministry of Health and Prevention
- Examination of academic journals and articles focusing on neurology clinical trials conducted in the region
Primary Research
- Interviews with neurologists and clinical research coordinators at leading hospitals and research institutions
- Surveys targeting pharmaceutical companies involved in neurology drug development
- Focus groups with patients participating in neurology clinical trials to gather insights on their experiences
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including clinical trial outcomes and expert opinions
- Triangulation of data from healthcare providers, regulatory bodies, and industry reports
- Sanity checks conducted through expert panel reviews comprising neurologists and clinical researchers
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total market size based on national healthcare expenditure and allocation to neurology
- Segmentation of the market by types of neurological disorders and corresponding clinical trial phases
- Incorporation of trends in healthcare policy and funding for neurological research in the UAE
Bottom-up Modeling
- Collection of data on the number of active neurology clinical trials and their respective budgets
- Estimation of costs associated with conducting trials, including patient recruitment and site management
- Volume x cost analysis based on historical data from completed trials in the UAE
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as population aging and prevalence of neurological disorders
- Scenario modeling based on potential changes in regulatory frameworks and funding availability
- Development of baseline, optimistic, and pessimistic forecasts for the neurology clinical trials market through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Neurology Clinical Trial Coordinators | 45 | Clinical Research Coordinators, Trial Managers |
| Neurologists in UAE Hospitals | 40 | Consultant Neurologists, Attending Physicians |
| Pharmaceutical Companies Conducting Trials | 40 | Clinical Development Managers, Regulatory Affairs Specialists |
| Patients Participating in Trials | 50 | Trial Participants, Patient Advocacy Group Members |
| Healthcare Policy Makers | 40 | Health Economists, Policy Analysts |
Frequently Asked Questions
What is the current value of the UAE Neurology Clinical Trials Market?
The UAE Neurology Clinical Trials Market is valued at approximately USD 1.2 billion, reflecting significant growth driven by the rising prevalence of neurological disorders and advancements in clinical research methodologies.