Region:Middle East
Author(s):Shubham
Product Code:KRAA8754
Pages:91
Published On:November 2025
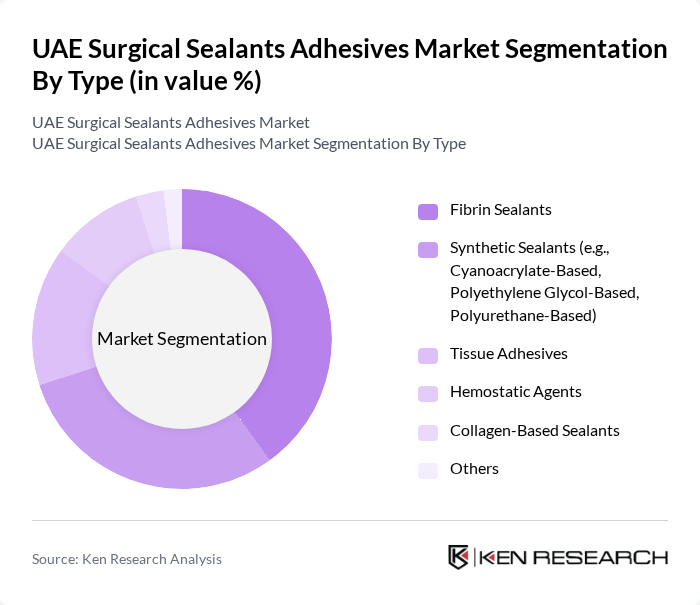
By Type:The market is segmented into various types of surgical sealants and adhesives, including fibrin sealants, synthetic sealants, tissue adhesives, hemostatic agents, collagen-based sealants, and others. Among these, fibrin sealants are gaining traction due to their biocompatibility and effectiveness in promoting wound healing. Synthetic sealants, particularly cyanoacrylate-based products, are also popular due to their ease of use and rapid bonding capabilities. The increasing preference for minimally invasive surgeries and the adoption of blended formulations that combine natural and synthetic polymers are driving demand for these products .
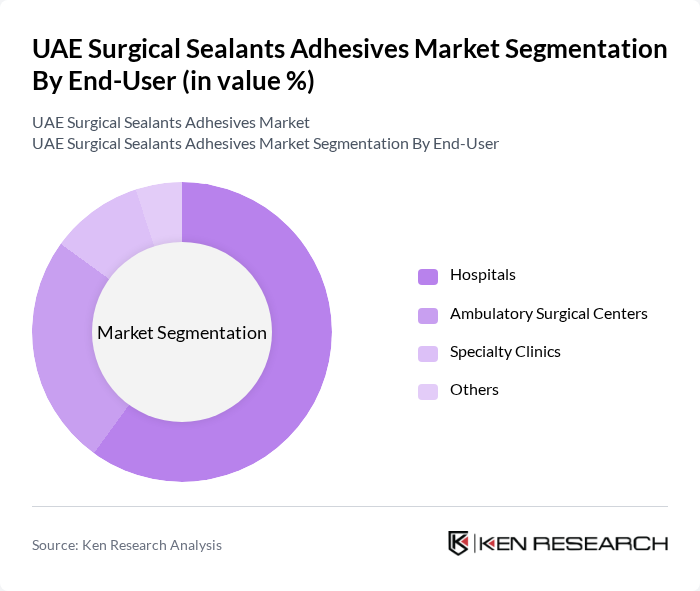
By End-User:The end-user segmentation includes hospitals, ambulatory surgical centers, specialty clinics, and others. Hospitals are the primary end-users of surgical sealants and adhesives, driven by the increasing number of surgical procedures performed in these facilities. Ambulatory surgical centers are also witnessing growth due to the rising trend of outpatient surgeries, which require effective sealing solutions to minimize complications and enhance recovery times. Specialty clinics are expanding their adoption of advanced sealant technologies for cosmetic, reconstructive, and minimally invasive procedures .
The UAE Surgical Sealants Adhesives Market is characterized by a dynamic mix of regional and international players. Leading participants such as Johnson & Johnson (Ethicon, Inc.), Medtronic plc, B. Braun Melsungen AG, Baxter International Inc., 3M Company, CryoLife, Inc. (now Artivion, Inc.), Integra LifeSciences Holdings Corporation, Advanced Medical Solutions Group plc, Stryker Corporation, Cohera Medical, Inc. (now part of Ethicon), Tissue Regenix Group plc, Sealantis Ltd. (an AMS company), Adhezion Biomedical, LLC, Medline Industries, LP, Hemostasis, LLC contribute to innovation, geographic expansion, and service delivery in this space.
The future of the UAE surgical sealants adhesives market appears promising, driven by ongoing advancements in medical technology and an increasing focus on patient-centered care. As healthcare infrastructure expands, the demand for innovative surgical solutions will likely rise. Additionally, the integration of digital technologies in surgical procedures is expected to enhance the efficiency and effectiveness of sealants, further propelling market growth. Stakeholders must remain agile to capitalize on these evolving trends and consumer preferences.
| Segment | Sub-Segments |
|---|---|
| By Type | Fibrin Sealants Synthetic Sealants (e.g., Cyanoacrylate-Based, Polyethylene Glycol-Based, Polyurethane-Based) Tissue Adhesives Hemostatic Agents Collagen-Based Sealants Others |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Clinics Others |
| By Application | Cardiovascular Surgery Orthopedic Surgery Neurological Surgery General Surgery Cosmetic and Reconstructive Surgery Burns & Skin Grafting Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | Abu Dhabi Dubai Sharjah Others |
| By Material Type | Natural Polymers (e.g., Fibrin, Collagen, Gelatin) Synthetic Polymers (e.g., Cyanoacrylate, Polyethylene Glycol) Others |
| By Regulatory Approval Status | CE Marked FDA Approved UAE Ministry of Health Approved Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| General Surgery Adhesives | 45 | Surgeons, Operating Room Managers |
| Orthopedic Sealants | 38 | Orthopedic Surgeons, Medical Device Buyers |
| Cardiovascular Adhesives | 42 | Cardiologists, Hospital Procurement Officers |
| Wound Closure Products | 40 | Wound Care Specialists, Clinical Directors |
| Regulatory and Compliance Insights | 35 | Regulatory Affairs Managers, Quality Assurance Officers |
The UAE Surgical Sealants Adhesives Market is valued at approximately USD 15 million, reflecting a five-year historical analysis. This growth is driven by factors such as the increasing prevalence of chronic diseases and advancements in surgical techniques.