Region:Middle East
Author(s):Geetanshi
Product Code:KRAA6136
Pages:80
Published On:January 2026
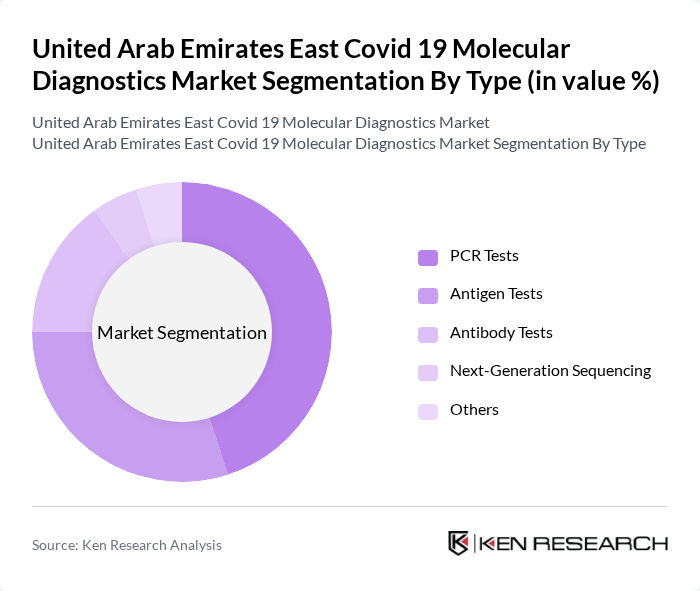
By Type:The market is segmented into various types of diagnostic tests, including PCR Tests, Antigen Tests, Antibody Tests, Next-Generation Sequencing, and Others. Among these, PCR Tests have emerged as the dominant segment due to their high accuracy and reliability in detecting Covid-19 infections. The increasing preference for PCR testing in both clinical and travel settings has significantly contributed to its market leadership. Antigen Tests are also gaining traction due to their rapid results, making them popular for mass screening in public places.
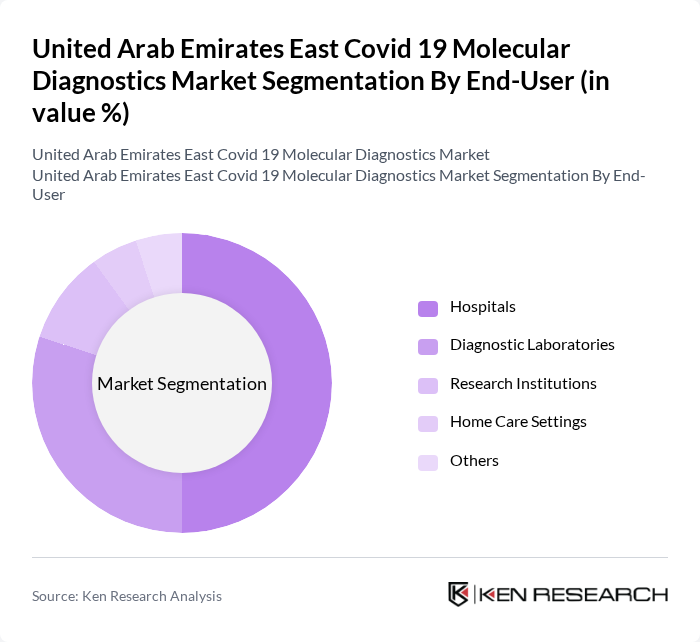
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Research Institutions, Home Care Settings, and Others. Hospitals are the leading end-user segment, driven by the high volume of Covid-19 testing required for patient management and treatment. The demand for diagnostic services in hospitals has surged, leading to increased investments in laboratory infrastructure and testing capabilities. Diagnostic Laboratories also play a crucial role, particularly in providing specialized testing services and supporting hospitals with high-throughput testing.
The United Arab Emirates East Covid 19 Molecular Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Thermo Fisher Scientific, Bio-Rad Laboratories, Cepheid, Hologic, Qiagen, BGI Genomics, GenMark Diagnostics, PerkinElmer, Agilent Technologies, Luminex Corporation, Mylab Discovery Solutions, MedGenome contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Covid-19 molecular diagnostics market in the UAE appears promising, driven by ongoing technological advancements and a commitment to public health. As the government continues to invest in healthcare infrastructure, the integration of AI and telehealth services is expected to enhance diagnostic capabilities. Additionally, the focus on preventive healthcare will likely lead to increased demand for innovative testing solutions, ensuring that the market remains resilient and adaptive to future health challenges.
| Segment | Sub-Segments |
|---|---|
| By Type | PCR Tests Antigen Tests Antibody Tests Next-Generation Sequencing Others |
| By End-User | Hospitals Diagnostic Laboratories Research Institutions Home Care Settings Others |
| By Region | Abu Dhabi Dubai Sharjah Ajman Others |
| By Technology | Real-Time PCR Digital PCR Microarray Technology Others |
| By Application | Clinical Diagnostics Research Applications Drug Development Others |
| By Investment Source | Private Investments Government Funding International Grants Others |
| By Policy Support | Health Insurance Coverage Subsidies for Testing Tax Incentives for R&D Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Public Health Laboratories | 100 | Laboratory Directors, Chief Pathologists |
| Private Diagnostic Centers | 80 | Operations Managers, Quality Assurance Officers |
| Healthcare Providers | 120 | Physicians, Medical Directors |
| Government Health Agencies | 60 | Policy Makers, Epidemiologists |
| Research Institutions | 50 | Research Scientists, Lab Technicians |
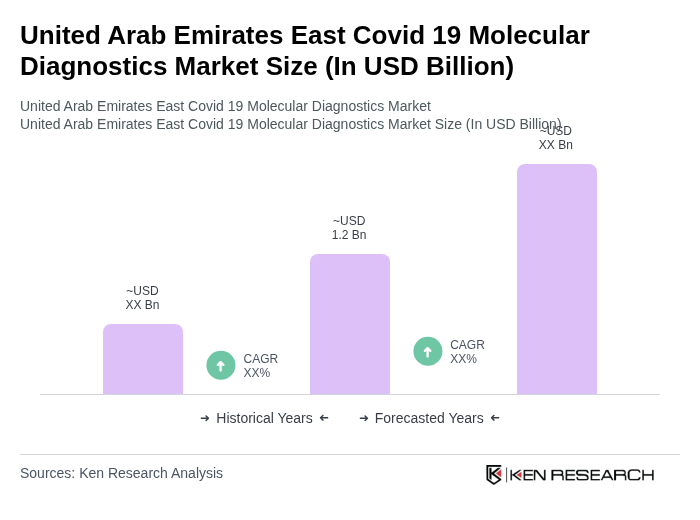
The United Arab Emirates East Covid-19 Molecular Diagnostics Market is valued at approximately USD 1.2 billion, reflecting significant growth driven by the demand for accurate and rapid testing solutions amid the ongoing pandemic challenges.