About the Report
Base Year 2024United Arab Emirates Orphan Drugs Market Overview
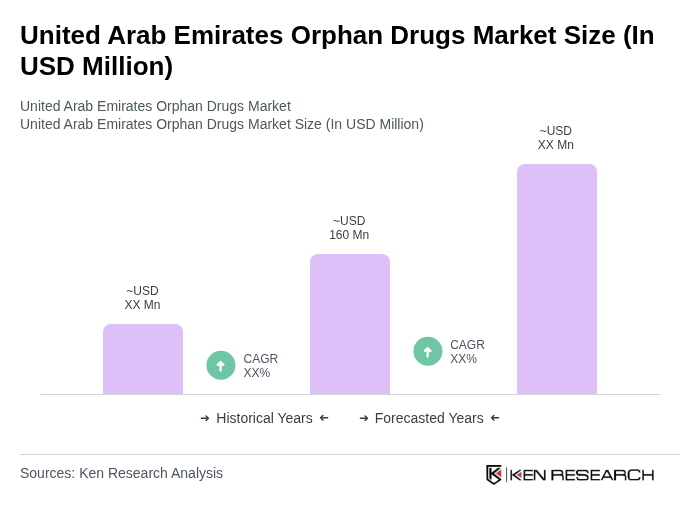
- The United Arab Emirates Orphan Drugs Market is valued at USD 160 million, based on a five-year historical analysis. This growth is primarily driven by increasing prevalence of rare diseases, advancements in biotechnology including gene and cell therapies, and supportive government policies aimed at enhancing healthcare access for patients with rare conditions.
- Key cities such as Dubai and Abu Dhabi dominate the market due to their advanced healthcare infrastructure, high disposable income, and a growing population that is increasingly aware of rare diseases. The presence of leading pharmaceutical companies and research institutions in these cities further strengthens their market position.
- The Federal Decree-Law No. 6 of 2022 on Health Technical Resources Regulation issued by the UAE Ministry of Health and Prevention governs orphan drugs, requiring registration with the Health Technical Resources Authority, compliance with international standards for rare disease treatments affecting fewer than 200,000 patients globally, and providing market exclusivity periods of up to seven years for approved products.
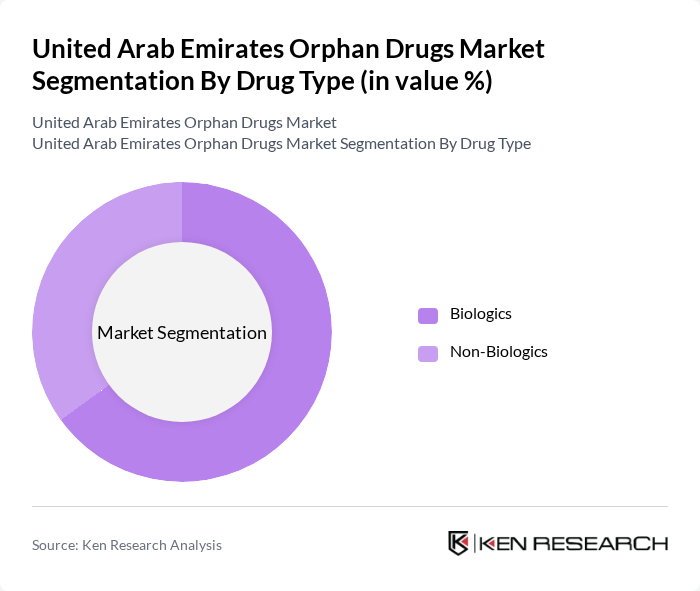
United Arab Emirates Orphan Drugs Market Segmentation
By Drug Type:The orphan drugs market can be segmented into two primary categories: Biologics and Non-Biologics. Biologics are derived from living organisms and are often used to treat complex diseases, while Non-Biologics include synthetic drugs that target specific conditions. The Biologics segment is currently leading the market due to the increasing demand for targeted therapies and personalized medicine.
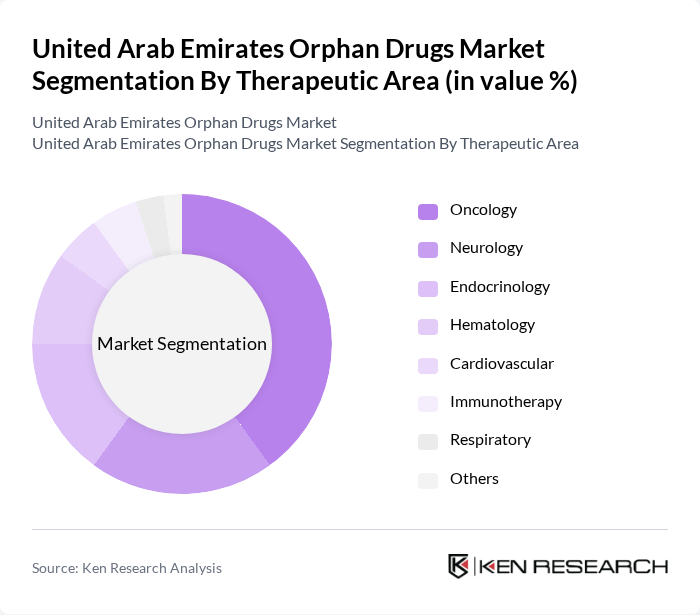
By Therapeutic Area:The orphan drugs market is further segmented by therapeutic areas, including Oncology, Neurology, Endocrinology, Hematology, Cardiovascular, Immunotherapy, Respiratory, and Others. Oncology is the leading therapeutic area, driven by the rising incidence of rare cancers and the development of innovative therapies that target specific cancer types.
United Arab Emirates Orphan Drugs Market Competitive Landscape
The United Arab Emirates Orphan Drugs Market is characterized by a dynamic mix of regional and international players. Leading participants such as Novartis, Sanofi, Pfizer, Roche, Amgen, GSK, Takeda, Alexion Pharmaceuticals, BioMarin Pharmaceutical, AbbVie, Merck & Co., Eli Lilly, Astellas Pharma, Sobi, Horizon Therapeutics contribute to innovation, geographic expansion, and service delivery in this space.
United Arab Emirates Orphan Drugs Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Rare Diseases:The United Arab Emirates has reported approximately 7,000 rare diseases affecting around 1 in 10,000 individuals. This growing prevalence is driving demand for orphan drugs, as healthcare providers seek effective treatments for these conditions. The UAE's population of approximately 9.5 million includes a significant number of patients with rare diseases, necessitating specialized medical interventions and increasing the urgency for orphan drug development and availability.
- Government Initiatives and Funding:The UAE government allocated AED 2.5 billion (approximately USD 680 million) to enhance healthcare services, including orphan drug research and development. Initiatives such as the UAE National Strategy for Rare Diseases aim to improve access to treatments. This funding supports clinical trials and incentivizes pharmaceutical companies to invest in orphan drug development, fostering a conducive environment for innovation in the healthcare sector.
- Advancements in Biotechnology:The UAE's biotechnology sector is rapidly evolving, with investments reaching AED 1.2 billion (around USD 327 million). Innovations in gene therapy and biomanufacturing are paving the way for new orphan drugs. The establishment of research hubs and partnerships with global biotech firms enhances the capacity for developing targeted therapies, addressing the unique needs of patients with rare diseases in the region.
Market Challenges
- High Cost of Drug Development:The average cost of developing an orphan drug can exceed USD 1.5 billion, which poses a significant barrier for companies in the UAE. This high financial burden often leads to limited investment in research and development, as firms weigh the risks against potential returns. Consequently, many promising therapies may not reach the market, leaving patients without viable treatment options for rare diseases.
- Regulatory Hurdles:The regulatory landscape for orphan drugs in the UAE can be complex, with lengthy approval processes that can take up to 18 months. This delay in market access can discourage pharmaceutical companies from pursuing orphan drug development. Additionally, the lack of clear guidelines for clinical trials and market authorization can create uncertainty, further complicating the path to bringing new therapies to patients in need.
United Arab Emirates Orphan Drugs Market Future Outlook
The future of the orphan drugs market in the UAE appears promising, driven by increasing collaboration between government entities and private sectors. Enhanced funding for research and development, coupled with a growing emphasis on personalized medicine, is expected to facilitate the introduction of innovative therapies in future. As healthcare infrastructure expands, the market is likely to witness a surge in the availability of orphan drugs, improving patient outcomes and fostering a more robust healthcare ecosystem.
Market Opportunities
- Expansion of Healthcare Infrastructure:The UAE is investing AED 3 billion (approximately USD 816 million) in healthcare infrastructure in future. This expansion will enhance access to orphan drugs, allowing more patients to receive necessary treatments. Improved facilities and services will also attract pharmaceutical companies to establish operations in the region, further stimulating market growth.
- Collaborations with Research Institutions:Partnerships between pharmaceutical companies and local research institutions are on the rise, with over 15 collaborations established. These alliances facilitate knowledge sharing and resource pooling, accelerating the development of orphan drugs. Such collaborations are crucial for addressing the unique challenges posed by rare diseases and enhancing the overall research landscape in the UAE.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Drug Type | Biologics Non-Biologics |
| By Therapeutic Area | Oncology Neurology Endocrinology Hematology Cardiovascular Immunotherapy Respiratory Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies |
| By Route of Administration | Oral Injectable Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health and Prevention, Emirates Health Services)
Pharmaceutical Manufacturers and Producers
Healthcare Providers and Hospitals
Distributors and Wholesalers
Patient Advocacy Groups
Health Insurance Companies
Biotechnology Firms
Players Mentioned in the Report:
Novartis
Sanofi
Pfizer
Roche
Amgen
GSK
Takeda
Alexion Pharmaceuticals
BioMarin Pharmaceutical
AbbVie
Merck & Co.
Eli Lilly
Astellas Pharma
Sobi
Horizon Therapeutics
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. United Arab Emirates Orphan Drugs Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 United Arab Emirates Orphan Drugs Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. United Arab Emirates Orphan Drugs Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of rare diseases
3.1.2 Government initiatives and funding
3.1.3 Advancements in biotechnology
3.1.4 Rising awareness among healthcare professionals
3.2 Market Challenges
3.2.1 High cost of drug development
3.2.2 Limited patient population
3.2.3 Regulatory hurdles
3.2.4 Market access issues
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Collaborations with research institutions
3.3.3 Increased investment in R&D
3.3.4 Growing demand for personalized medicine
3.4 Market Trends
3.4.1 Shift towards patient-centric approaches
3.4.2 Rise of digital health solutions
3.4.3 Focus on gene therapies
3.4.4 Integration of AI in drug discovery
3.5 Government Regulation
3.5.1 Orphan drug designation policies
3.5.2 Pricing and reimbursement frameworks
3.5.3 Clinical trial regulations
3.5.4 Market authorization processes
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. United Arab Emirates Orphan Drugs Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. United Arab Emirates Orphan Drugs Market Segmentation
8.1 By Drug Type
8.1.1 Biologics
8.1.2 Non-Biologics
8.2 By Therapeutic Area
8.2.1 Oncology
8.2.2 Neurology
8.2.3 Endocrinology
8.2.4 Hematology
8.2.5 Cardiovascular
8.2.6 Immunotherapy
8.2.7 Respiratory
8.2.8 Others
8.3 By Distribution Channel
8.3.1 Hospital Pharmacies
8.3.2 Retail Pharmacies
8.3.3 Online Pharmacies
8.4 By Route of Administration
8.4.1 Oral
8.4.2 Injectable
8.4.3 Others
9. United Arab Emirates Orphan Drugs Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Market Share (%)
9.2.3 Revenue Growth Rate (CAGR %)
9.2.4 R&D Spend as % of Revenue
9.2.5 Pipeline Strength (No. of Orphan Drugs in Development)
9.2.6 Regulatory Approvals (No. in UAE)
9.2.7 Market Penetration (UAE Coverage %)
9.2.8 EBITDA Margin (%)
9.2.9 Patient Reach (No. of Patients Treated)
9.2.10 Distribution Network Coverage
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Novartis
9.5.2 Sanofi
9.5.3 Pfizer
9.5.4 Roche
9.5.5 Amgen
9.5.6 GSK
9.5.7 Takeda
9.5.8 Alexion Pharmaceuticals
9.5.9 BioMarin Pharmaceutical
9.5.10 AbbVie
9.5.11 Merck & Co.
9.5.12 Eli Lilly
9.5.13 Astellas Pharma
9.5.14 Sobi
9.5.15 Horizon Therapeutics
10. United Arab Emirates Orphan Drugs Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health and Prevention
10.1.2 Ministry of Finance
10.1.3 Ministry of Education
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Research and Development Funding
10.2.3 Public-Private Partnerships
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Pharmacies
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training and Support Needs
10.4.3 Financial Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost Savings Analysis
10.5.2 Patient Outcomes Improvement
10.5.3 Market Expansion Opportunities
10.5.4 Others
11. United Arab Emirates Orphan Drugs Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from health authorities in the UAE
- Review of published studies on orphan drug prevalence and treatment options
- Examination of regulatory frameworks and policies affecting orphan drugs in the UAE
Primary Research
- Interviews with healthcare professionals specializing in rare diseases
- Surveys with pharmaceutical companies involved in orphan drug development
- Focus groups with patient advocacy organizations and caregivers
Validation & Triangulation
- Cross-validation of findings with data from international orphan drug registries
- Triangulation of insights from healthcare providers, patients, and industry experts
- Sanity checks through expert panel discussions and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total healthcare expenditure allocated to orphan drugs in the UAE
- Analysis of the demographic distribution of rare disease patients
- Incorporation of government health initiatives and funding for orphan drugs
Bottom-up Modeling
- Collection of sales data from leading pharmaceutical companies in the orphan drug sector
- Estimation of treatment costs based on drug pricing and patient population
- Volume x cost analysis for each orphan drug category
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering factors like disease prevalence and healthcare access
- Scenario modeling based on potential regulatory changes and market entry of new therapies
- Baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 120 | Oncologists, Geneticists, Rare Disease Specialists |
| Pharmaceutical Companies | 80 | Product Managers, R&D Directors |
| Patient Advocacy Groups | 60 | Advocacy Leaders, Patient Representatives |
| Regulatory Bodies | 40 | Policy Makers, Regulatory Affairs Managers |
| Healthcare Payers | 70 | Insurance Analysts, Health Economists |
Frequently Asked Questions
What is the current value of the United Arab Emirates Orphan Drugs Market?
The United Arab Emirates Orphan Drugs Market is valued at approximately USD 160 million, reflecting a significant growth driven by the increasing prevalence of rare diseases and advancements in biotechnology, alongside supportive government policies aimed at enhancing healthcare access.