Region:North America
Author(s):Geetanshi
Product Code:KRAA4537
Pages:99
Published On:September 2025
 Market.png)
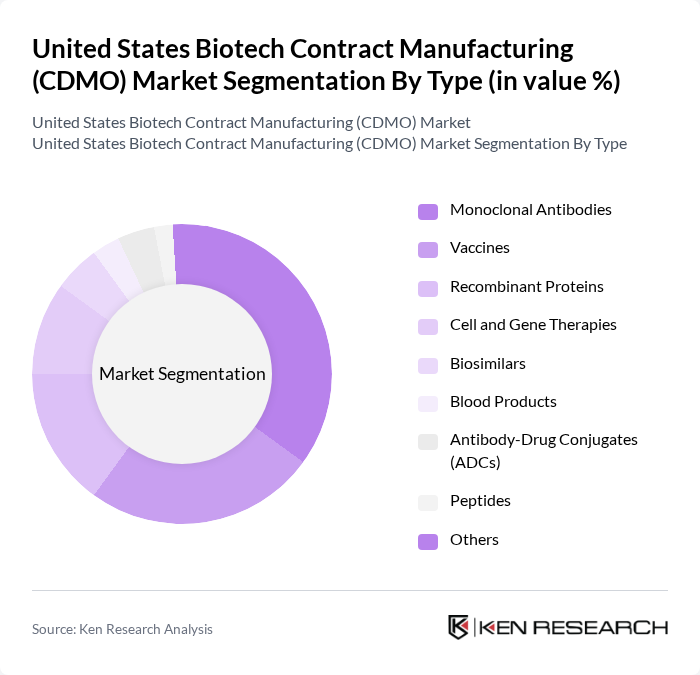
By Type:The market is segmented into various types, including Monoclonal Antibodies, Vaccines, Recombinant Proteins, Cell and Gene Therapies, Biosimilars, Blood Products, Antibody-Drug Conjugates (ADCs), Peptides, and Others. Among these,Monoclonal Antibodiesare leading the market due to their widespread application in therapeutic treatments, particularly for oncology and autoimmune diseases, and their effectiveness in targeting specific disease pathways. The increasing investment in research and development for innovative therapies, including next-generation biologics and biosimilars, further propels this segment's growth .
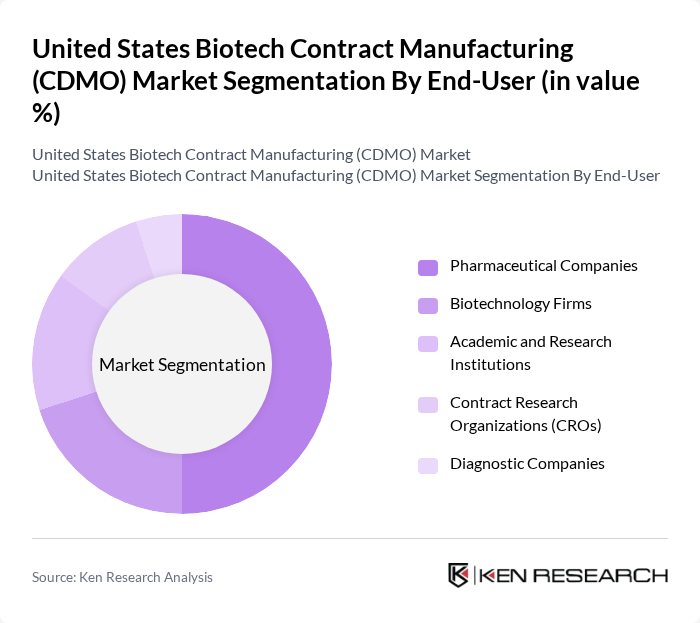
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Academic and Research Institutions, Contract Research Organizations (CROs), and Diagnostic Companies.Pharmaceutical Companiesdominate this segment as they increasingly rely on CDMOs for the production of complex biologics and to streamline their manufacturing processes. The trend of outsourcing is driven by the need for cost efficiency, access to advanced manufacturing technologies, and the ability to rapidly scale production in response to market demands .
The United States Biotech Contract Manufacturing (CDMO) Market is characterized by a dynamic mix of regional and international players. Leading participants such as Lonza Group AG, Catalent, Inc., Thermo Fisher Scientific Inc. (Patheon), Fujifilm Diosynth Biotechnologies, Samsung Biologics, WuXi AppTec, Avid Bioservices, Inc., KBI Biopharma, Inc., Emergent BioSolutions Inc., AGC Biologics, AbbVie Inc., Amgen Inc., Genentech, Inc. (Roche), Biogen Inc., Celerion contribute to innovation, geographic expansion, and service delivery in this space.
The future of the U.S. biotech contract manufacturing market appears promising, driven by ongoing innovations and a growing focus on personalized medicine. As the demand for biologics continues to rise, CDMOs will play a pivotal role in facilitating the development and production of complex therapies. Additionally, the integration of advanced technologies such as AI and automation will enhance operational efficiencies, enabling CDMOs to meet the evolving needs of biopharmaceutical companies while ensuring compliance with stringent regulations.
| Segment | Sub-Segments |
|---|---|
| By Type | Monoclonal Antibodies Vaccines Recombinant Proteins Cell and Gene Therapies Biosimilars Blood Products Antibody-Drug Conjugates (ADCs) Peptides Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic and Research Institutions Contract Research Organizations (CROs) Diagnostic Companies |
| By Application | Therapeutics Diagnostics Research and Development Clinical Trials |
| By Service Type | Process Development Manufacturing (Clinical & Commercial) Analytical & Quality Control Packaging and Labeling Fill-Finish Services |
| By Scale of Operation | Small Scale Medium Scale Large Scale |
| By Distribution Channel | Direct Sales Online Sales Distributors |
| By Region | Northeast Midwest South West |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biotech Firms Utilizing CDMO Services | 120 | R&D Directors, Procurement Managers |
| Pharmaceutical Companies Engaged in Outsourcing | 90 | Project Managers, Supply Chain Executives |
| Quality Assurance in Contract Manufacturing | 60 | Quality Control Managers, Compliance Officers |
| Regulatory Affairs in Biotech | 50 | Regulatory Affairs Specialists, Legal Advisors |
| Market Trends and Insights | 70 | Market Analysts, Business Development Managers |
The United States Biotech Contract Manufacturing (CDMO) Market is valued at approximately USD 22 billion, driven by the increasing demand for biopharmaceuticals, advancements in biotechnology, and the rising prevalence of chronic diseases such as cancer and diabetes.