Region:North America
Author(s):Geetanshi
Product Code:KRAD4789
Pages:100
Published On:December 2025
 Market.png)
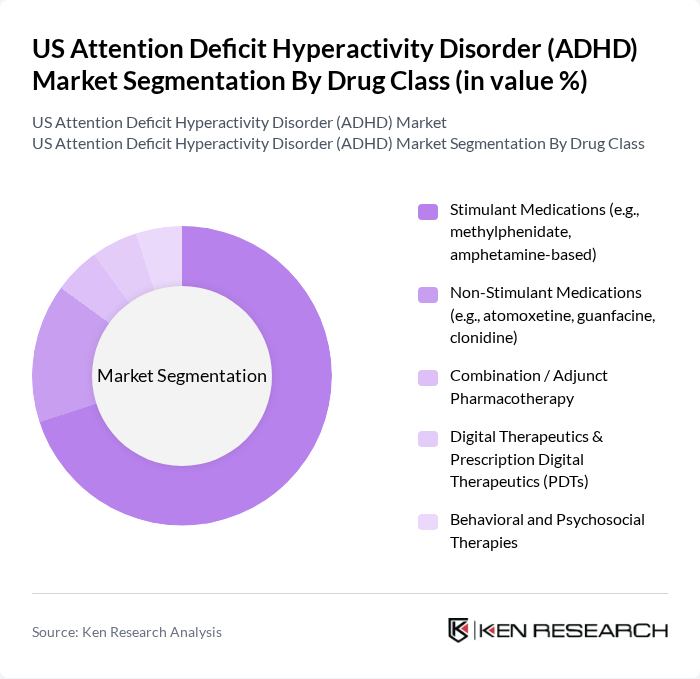
By Drug Class:The drug class segmentation includes various types of medications and interventions used to treat ADHD. The primary subsegments are Stimulant Medications, Non-Stimulant Medications, Combination/Adjunct Pharmacotherapy, Digital Therapeutics & Prescription Digital Therapeutics (PDTs), and Behavioral and Psychosocial Therapies. Stimulant medications, such as methylphenidate and amphetamine-based drugs, dominate the pharmacologic segment due to their well-documented efficacy, rapid onset of action, and long-standing inclusion in major clinical guidelines. Growing use of non-stimulants, digital therapeutics, and behavioral interventions reflects an increasing focus on individualized treatment plans and the management of side effects and comorbidities.
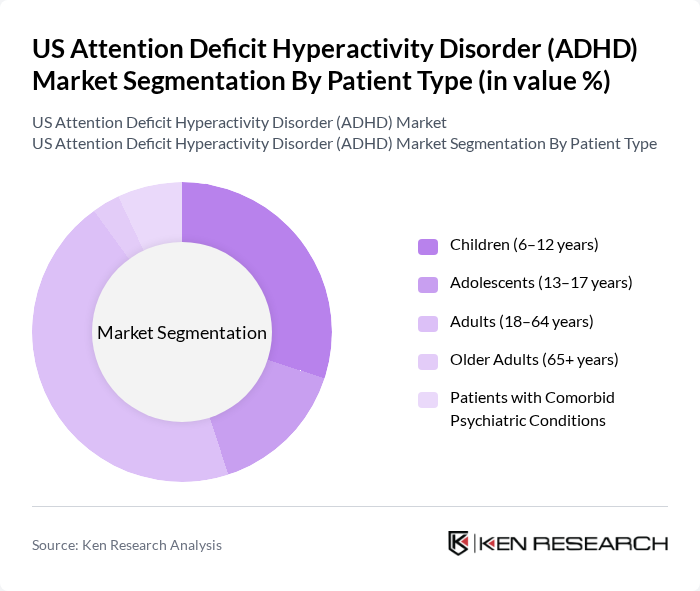
By Patient Type:The patient type segmentation categorizes individuals receiving ADHD treatment into distinct groups. These include Children (6–12 years), Adolescents (13–17 years), Adults (18–64 years), Older Adults (65+ years), and Patients with Comorbid Psychiatric Conditions. While ADHD historically has been most commonly diagnosed in school-aged children, recent epidemiological and prescribing data indicate that adults now account for a substantial and growing share of treated patients, driven by increased recognition of lifelong ADHD and expanded adult screening in primary care and psychiatry. Children and adolescents remain critical segments due to school-based identification, parental advocacy, and early treatment focusing on academic and behavioral outcomes.
The US Attention Deficit Hyperactivity Disorder (ADHD) Market is characterized by a dynamic mix of regional and international players. Leading participants such as Takeda Pharmaceutical Company Limited (including legacy Shire plc), Novartis AG, Johnson & Johnson (Janssen Pharmaceuticals), Pfizer Inc., Eli Lilly and Company, Teva Pharmaceutical Industries Ltd., Tris Pharma, Inc., Supernus Pharmaceuticals, Inc., Neos Therapeutics, Inc. (a subsidiary of Aytu BioPharma, Inc.), Ironshore Pharmaceuticals Inc. (a division of Highland Therapeutics Inc.), Akili, Inc., Corium, LLC, Medice Arzneimittel Pütter GmbH & Co. KG, Otsuka Pharmaceutical Co., Ltd., Lundbeck A/S contribute to innovation in extended-release formulations, non-stimulant options, and digital therapeutics, as well as geographic expansion and differentiated service delivery models across specialty clinics, telepsychiatry platforms, and integrated behavioral health networks.
The future of the ADHD market in the U.S. appears promising, driven by ongoing advancements in treatment methodologies and increasing integration of technology in healthcare. As telehealth services expand, more patients will gain access to specialized care, particularly in underserved areas. Furthermore, the growing emphasis on holistic treatment approaches, including behavioral therapies and community support, is expected to enhance patient outcomes and satisfaction, fostering a more inclusive environment for individuals with ADHD.
| Segment | Sub-Segments |
|---|---|
| By Drug Class | Stimulant Medications (e.g., methylphenidate, amphetamine-based) Non-Stimulant Medications (e.g., atomoxetine, guanfacine, clonidine) Combination / Adjunct Pharmacotherapy Digital Therapeutics & Prescription Digital Therapeutics (PDTs) Behavioral and Psychosocial Therapies |
| By Patient Type | Children (6–12 years) Adolescents (13–17 years) Adults (18–64 years) Older Adults (65+ years) Patients with Comorbid Psychiatric Conditions |
| By Distribution Channel | Retail Pharmacies Hospital Pharmacies Online & Mail-Order Pharmacies Specialty Clinics & ADHD Centers Others |
| By Care Setting | Outpatient & Office-Based Physician Practices Community Mental Health Centers Telehealth & Virtual Care Platforms School-Linked or School-Based Services Home-Based & Remote Monitoring Programs |
| By Formulation | Immediate-Release (IR) Formulations Extended-Release (ER) / Long-Acting Formulations Oral Solid Dosage Forms (tablets, capsules, chewables) Oral Liquids & Suspensions Others (e.g., transdermal systems) |
| By Payer Type | Private Commercial Insurance Medicaid & Other Public Programs Medicare Self-Pay / Out-of-Pocket Employer-Sponsored & Managed Care Plans |
| By Region (US) | Northeast Midwest South West U.S. Territories |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 150 | Pediatricians, Psychiatrists, Clinical Psychologists |
| Parents of ADHD Diagnosed Children | 150 | Parents, Guardians, Caregivers |
| Educators in Special Education | 100 | Special Education Teachers, School Counselors |
| Pharmaceutical Representatives | 80 | Sales Representatives, Product Managers |
| Policy Makers in Education and Health | 50 | Health Policy Analysts, Education Administrators |
The US Attention Deficit Hyperactivity Disorder (ADHD) market is valued at approximately USD 10.7 billion, reflecting significant growth driven by increased awareness, advancements in treatment options, and rising diagnosis rates among both children and adults.