Region:North America
Author(s):Rebecca
Product Code:KRAD5022
Pages:96
Published On:December 2025
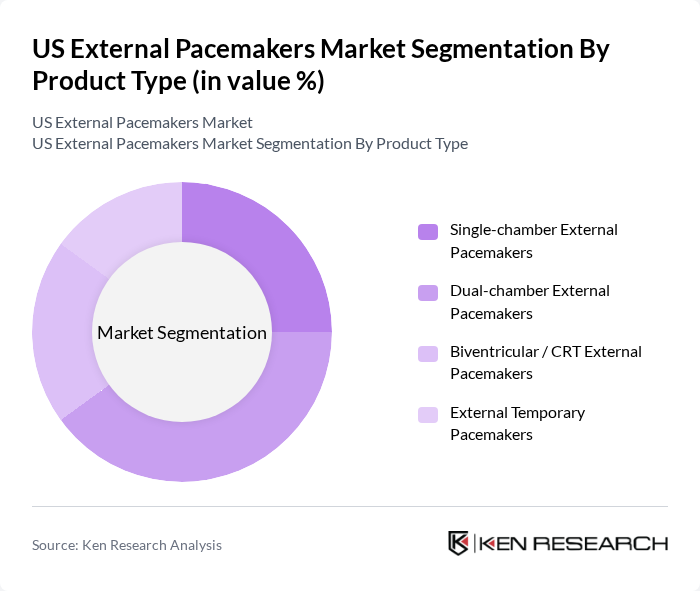
By Product Type:The product type segmentation includes various categories of external pacemakers, each designed to cater to specific patient needs and clinical scenarios. The subsegments are Single-chamber External Pacemakers, Dual-chamber External Pacemakers, Biventricular / CRT External Pacemakers, and External Temporary Pacemakers. Dual-chamber External Pacemakers account for a prominent share of usage as they better mimic physiological atrioventricular synchrony and are widely used for managing advanced atrioventricular block and other complex rhythm disorders in intensive care and perioperative settings. Increasing awareness of the hemodynamic benefits of dual-chamber pacing, coupled with improvements in programmability, safety features, and compatibility with monitoring systems, has supported their strong adoption in US hospitals.
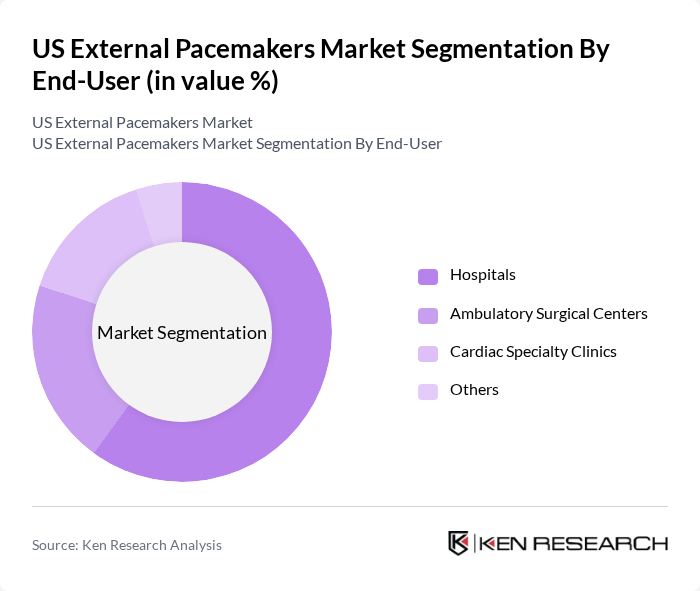
By End-User:The end-user segmentation encompasses various healthcare settings where external pacemakers are utilized, including Hospitals, Ambulatory Surgical Centers, Cardiac Specialty Clinics, and Others. Hospitals dominate this segment due to their comprehensive cardiac care facilities, intensive and coronary care units, and the capability to manage high-acuity cases requiring temporary pacing after cardiac surgery, acute myocardial infarction, or catheter-based interventions. The increasing number of cardiovascular procedures, such as open-heart surgery, structural heart interventions, and complex electrophysiology procedures performed in hospital settings, along with the presence of multidisciplinary cardiac teams, has solidified hospitals as the primary end-user of external pacemakers.
The US External Pacemakers Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Boston Scientific Corporation, Abbott Laboratories (Abbott), BIOTRONIK SE & Co. KG, OSYPKA MEDICAL GmbH, OSCOR Inc. (a Teleflex company), Pacetronix Ltd., Lepu Medical Technology (Beijing) Co., Ltd., Avery Biomedical Devices, Inc., ZOLL Medical Corporation, MicroPort Scientific Corporation, Edwards Lifesciences Corporation, LivaNova PLC, Philips Healthcare (Philips Medical Systems), GE HealthCare Technologies Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The US external pacemakers market is poised for significant transformation, driven by technological innovations and demographic shifts. As the prevalence of cardiac diseases continues to rise, healthcare providers are increasingly adopting advanced pacemaker technologies that enhance patient care. Furthermore, the integration of digital health solutions is expected to streamline monitoring and improve patient outcomes. These trends indicate a robust future for the market, with ongoing investments in research and development likely to yield new opportunities for growth and improved patient management strategies.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Single-chamber External Pacemakers Dual-chamber External Pacemakers Biventricular / CRT External Pacemakers External Temporary Pacemakers |
| By End-User | Hospitals Ambulatory Surgical Centers Cardiac Specialty Clinics Others |
| By Clinical Application | Bradycardia and Heart Block Atrial Fibrillation and Other Arrhythmias Management Acute Myocardial Infarction and Post?cardiac Surgery Support Others |
| By Distribution Channel | Direct Sales to Hospitals and IDNs Medical Device Distributors Group Purchasing Organizations (GPOs) Online / E?procurement Portals |
| By US Region | Northeast Midwest South West |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients Others |
| By Technology Feature | Conventional External Pacemakers External Pacemakers with Remote Monitoring / Telemetry MRI?compatible External Pacemakers Battery and Lead?less Innovation Platforms |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiology Clinics | 100 | Cardiologists, Clinic Administrators |
| Hospitals with Electrophysiology Departments | 90 | Procurement Officers, Department Heads |
| Medical Device Distributors | 60 | Sales Managers, Product Specialists |
| Biomedical Engineering Firms | 50 | Biomedical Engineers, R&D Managers |
| Health Insurance Providers | 70 | Policy Analysts, Claims Managers |
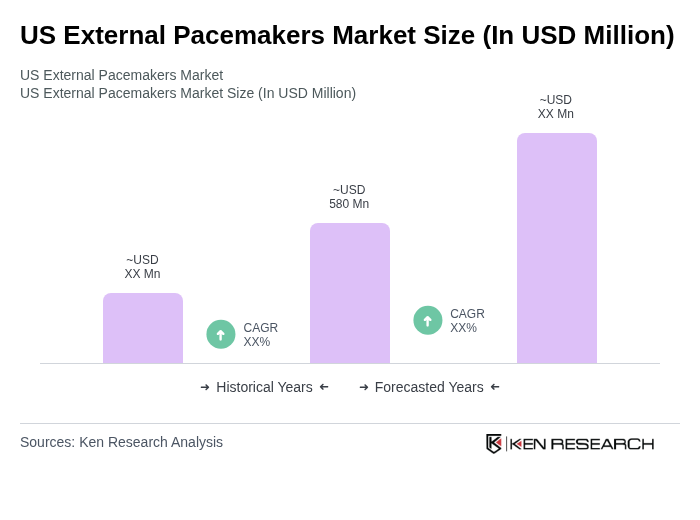
The US External Pacemakers Market is valued at approximately USD 580 million, reflecting a significant share of the broader US pacemakers segment. This valuation is based on a five-year historical analysis of national revenue and the increasing demand for external devices in North America.