About the Report
Base Year 2024US Fusion Biopsy Market Overview
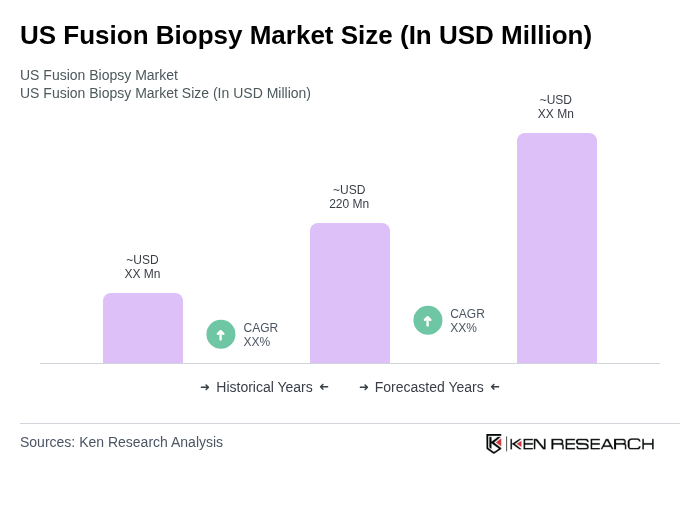
- The US Fusion Biopsy Market is valued at USD 220 million, based on recent analysis. Growth is driven by the increasing prevalence of prostate cancer, the shift toward precision oncology and early detection protocols, and ongoing technological advancements such as AI-enhanced image-fusion capabilities and enhanced MRI integration.
- Key players in this market include Koninklijke Philips N.V., GE HealthCare (including BK Medical), and KOELIS. These companies play a significant role in driving growth and maintaining high standards in MRI-ultrasound fusion systems and expanded clinical adoption. The United States remains the dominant region, fueled by robust imaging infrastructure, high screening volumes, and strong academic-institution medical centers supporting fusion biopsy deployment.
- In 2024, the Centers for Medicare & Medicaid Services (CMS) and the American Medical Association (AMA) established a new Category III code (0898T) and national payment rate for Avenda Health’s Unfold AI mapping technology, which enhances image-guided fusion biopsy interpretation and supports adoption through a clear reimbursement framework.
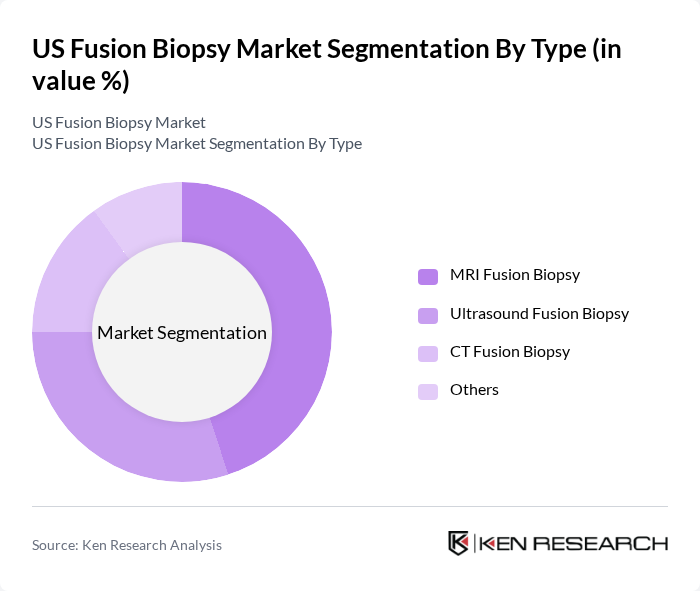
US Fusion Biopsy Market Segmentation
By Type:The market is segmented into various types of fusion biopsy techniques, including MRI Fusion Biopsy, Ultrasound Fusion Biopsy, CT Fusion Biopsy, and Others. Among these, MRI Fusion Biopsy is currently the leading segment due to its high accuracy and effectiveness in detecting prostate cancer. The increasing adoption of MRI technology in clinical settings and the growing preference for non-invasive procedures are driving the demand for MRI Fusion Biopsy.
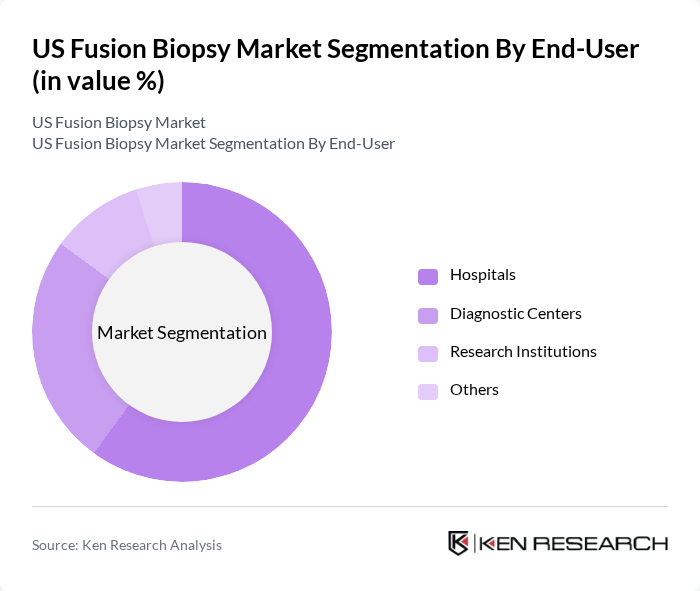
By End-User:The end-user segmentation includes Hospitals, Diagnostic Centers, Research Institutions, and Others. Hospitals dominate this segment due to their advanced facilities and higher patient volumes. The increasing number of hospitals adopting fusion biopsy technologies for prostate cancer diagnosis is a significant factor contributing to this dominance.
US Fusion Biopsy Market Competitive Landscape
The US Fusion Biopsy Market is characterized by a dynamic mix of regional and international players. Leading participants such as Koninklijke Philips N.V., GE HealthCare, and KOELIS contribute to innovation, geographic expansion, and service delivery in this space.
US Fusion Biopsy Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Prostate Cancer:The American Cancer Society estimates that in future, approximately 288,300 new cases of prostate cancer will be diagnosed in the United States. This rising incidence drives the demand for advanced diagnostic techniques, including fusion biopsy, which combines MRI and ultrasound imaging to enhance detection accuracy. As healthcare providers seek effective solutions to manage this growing patient population, the fusion biopsy market is positioned for significant growth, supported by the increasing need for precise cancer diagnostics.
- Advancements in Imaging Technology:The integration of cutting-edge imaging technologies, such as high-resolution MRI and real-time ultrasound, has revolutionized the biopsy process. In future, the global medical imaging market is projected to reach $45 billion, with significant investments directed towards enhancing imaging capabilities. These advancements facilitate more accurate targeting of suspicious lesions, thereby improving biopsy outcomes. As healthcare facilities adopt these technologies, the demand for fusion biopsy systems is expected to rise, further propelling market growth.
- Rising Demand for Minimally Invasive Procedures:Minimally invasive procedures are increasingly preferred due to their associated benefits, including reduced recovery times and lower complication rates. According to the National Institutes of Health, minimally invasive surgeries have seen a 20% increase in adoption over the past five years. Fusion biopsy, being less invasive than traditional methods, aligns with this trend, attracting both patients and healthcare providers. This shift towards less invasive diagnostic techniques is a key driver for the fusion biopsy market's expansion.
Market Challenges
- High Cost of Fusion Biopsy Systems:The initial investment required for fusion biopsy systems can be substantial, often exceeding $100,000 per unit. This high cost poses a significant barrier for many healthcare facilities, particularly smaller practices and outpatient centers. As a result, the adoption of fusion biopsy technology may be limited, hindering market growth. Financial constraints can also affect the availability of these advanced systems in underserved regions, further complicating access to necessary diagnostic procedures.
- Limited Reimbursement Policies:Reimbursement for fusion biopsy procedures remains inconsistent across various insurance providers. In future, only 60% of private insurers are expected to cover fusion biopsy, creating financial uncertainty for healthcare providers. This lack of comprehensive reimbursement policies can deter facilities from investing in fusion biopsy technologies, as they may not recover the costs associated with these advanced procedures. Consequently, this challenge may slow the market's growth and limit patient access to innovative diagnostic options.
US Fusion Biopsy Market Future Outlook
The US fusion biopsy market is poised for transformative growth, driven by technological advancements and an increasing focus on personalized medicine. As healthcare providers continue to embrace innovative diagnostic solutions, the integration of artificial intelligence and telemedicine into biopsy procedures is expected to enhance accuracy and patient engagement. Furthermore, the expansion of outpatient surgical centers will facilitate greater access to fusion biopsy, ultimately improving patient outcomes and driving market expansion in the coming years.
Market Opportunities
- Development of AI-Assisted Biopsy Technologies:The emergence of AI-assisted technologies presents a significant opportunity for the fusion biopsy market. By leveraging machine learning algorithms, these technologies can enhance image analysis and lesion detection, potentially increasing diagnostic accuracy. As AI integration becomes more prevalent, healthcare providers may adopt these systems to improve patient outcomes and streamline biopsy procedures, driving market growth.
- Expansion of Outpatient Surgical Centers:The growth of outpatient surgical centers is creating new opportunities for fusion biopsy procedures. In future, the number of outpatient surgical centers in the US is projected to reach 6,000, providing increased access to advanced diagnostic services. This trend aligns with the rising demand for minimally invasive procedures, positioning fusion biopsy as a preferred option in these settings, thereby enhancing market potential.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | MRI Fusion Biopsy Ultrasound Fusion Biopsy CT Fusion Biopsy Others |
| By End-User | Hospitals Diagnostic Centers Research Institutions Others |
| By Procedure Type | Transrectal Biopsy Transperineal Biopsy Others |
| By Imaging Technology | MRI Ultrasound CT Others |
| By Region | Northeast Midwest South West |
| By Patient Demographics | Age Group (Under 50, 50-70, Over 70) Gender (Male, Female) Others |
| By Clinical Setting | Inpatient Outpatient Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Food and Drug Administration, Centers for Medicare & Medicaid Services)
Manufacturers and Producers of Biopsy Equipment
Healthcare Providers and Hospitals
Medical Device Distributors
Health Insurance Companies
Industry Associations (e.g., American Urological Association, Radiological Society of North America)
Pharmaceutical Companies involved in Oncology
Players Mentioned in the Report:
Philips Healthcare
Siemens Healthineers
GE Healthcare
Hitachi Medical Systems
Canon Medical Systems
Medtronic
Boston Scientific
Hologic
Becton Dickinson
Varian Medical Systems
iCAD
Focal Therapeutics
CMR Surgical
Endomag
UroNav
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. US Fusion Biopsy Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 US Fusion Biopsy Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. US Fusion Biopsy Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of prostate cancer
3.1.2 Advancements in imaging technology
3.1.3 Rising demand for minimally invasive procedures
3.1.4 Growing awareness and adoption of fusion biopsy techniques
3.2 Market Challenges
3.2.1 High cost of fusion biopsy systems
3.2.2 Limited reimbursement policies
3.2.3 Technical complexities in procedure
3.2.4 Shortage of trained professionals
3.3 Market Opportunities
3.3.1 Expansion of outpatient surgical centers
3.3.2 Development of AI-assisted biopsy technologies
3.3.3 Increasing investment in healthcare infrastructure
3.3.4 Collaborations between technology firms and healthcare providers
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Integration of telemedicine in biopsy procedures
3.4.3 Rise in patient-centric healthcare models
3.4.4 Adoption of robotic-assisted biopsy systems
3.5 Government Regulation
3.5.1 FDA guidelines on medical devices
3.5.2 Medicare reimbursement policies
3.5.3 State-level healthcare regulations
3.5.4 Compliance with HIPAA standards
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. US Fusion Biopsy Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. US Fusion Biopsy Market Segmentation
8.1 By Type
8.1.1 MRI Fusion Biopsy
8.1.2 Ultrasound Fusion Biopsy
8.1.3 CT Fusion Biopsy
8.1.4 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Diagnostic Centers
8.2.3 Research Institutions
8.2.4 Others
8.3 By Procedure Type
8.3.1 Transrectal Biopsy
8.3.2 Transperineal Biopsy
8.3.3 Others
8.4 By Imaging Technology
8.4.1 MRI
8.4.2 Ultrasound
8.4.3 CT
8.4.4 Others
8.5 By Region
8.5.1 Northeast
8.5.2 Midwest
8.5.3 South
8.5.4 West
8.6 By Patient Demographics
8.6.1 Age Group (Under 50, 50-70, Over 70)
8.6.2 Gender (Male, Female)
8.6.3 Others
8.7 By Clinical Setting
8.7.1 Inpatient
8.7.2 Outpatient
8.7.3 Others
9. US Fusion Biopsy Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Product Innovation Rate
9.2.8 Sales Conversion Rate
9.2.9 Average Order Value
9.2.10 Customer Satisfaction Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Philips Healthcare
9.5.2 Siemens Healthineers
9.5.3 GE Healthcare
9.5.4 Hitachi Medical Systems
9.5.5 Canon Medical Systems
9.5.6 Medtronic
9.5.7 Boston Scientific
9.5.8 Hologic
9.5.9 Becton Dickinson
9.5.10 Varian Medical Systems
9.5.11 iCAD
9.5.12 Focal Therapeutics
9.5.13 CMR Surgical
9.5.14 Endomag
9.5.15 UroNav
10. US Fusion Biopsy Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Vendor Selection Criteria
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends
10.2.2 Funding Sources
10.2.3 Project Prioritization
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Operational Challenges
10.3.2 Financial Constraints
10.3.3 Technological Limitations
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training Requirements
10.4.2 Infrastructure Readiness
10.4.3 Acceptance of New Technologies
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics
10.5.2 Case Studies
10.5.3 Future Use Cases
10.5.4 Others
11. US Fusion Biopsy Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
1.3 Value Proposition Analysis
1.4 Competitive Landscape Overview
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Audience Segmentation
2.4 Communication Strategies
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 Online Distribution Channels
3.4 Partnership Opportunities
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
4.4 Customer Willingness to Pay
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments Analysis
5.3 Emerging Trends Identification
5.4 Future Demand Projections
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Feedback Mechanisms
6.4 Relationship Management Strategies
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Customer-Centric Innovations
7.4 Competitive Differentiation
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
8.4 Market Research Activities
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging Strategies
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
12.2 Risk Mitigation Strategies
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from healthcare organizations and industry associations
- Review of published articles in medical journals focusing on fusion biopsy techniques
- Examination of regulatory documents and guidelines from the FDA regarding biopsy procedures
Primary Research
- Interviews with urologists and radiologists specializing in prostate cancer diagnosis
- Surveys with hospital administrators regarding equipment procurement and usage
- Field interviews with medical device sales representatives to gather insights on market trends
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and secondary data sources
- Triangulation of market data from clinical studies, sales reports, and expert opinions
- Sanity checks through feedback from a panel of healthcare professionals
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on national healthcare expenditure on oncology
- Segmentation of the market by geographic regions and healthcare facilities
- Incorporation of trends in prostate cancer incidence rates and screening practices
Bottom-up Modeling
- Collection of data on the number of fusion biopsy procedures performed annually
- Estimation of average costs associated with fusion biopsy equipment and procedures
- Analysis of market share data from leading medical device manufacturers
Forecasting & Scenario Analysis
- Multi-variable regression analysis incorporating factors such as technological advancements and demographic shifts
- Scenario modeling based on potential changes in healthcare policies and reimbursement rates
- Development of baseline, optimistic, and pessimistic market growth projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Urology Clinics | 100 | Urologists, Clinic Managers |
| Hospitals with Oncology Departments | 80 | Oncologists, Hospital Administrators |
| Radiology Centers | 70 | Radiologists, Imaging Technologists |
| Medical Device Distributors | 60 | Sales Representatives, Product Managers |
| Healthcare Policy Makers | 50 | Health Economists, Policy Analysts |
Frequently Asked Questions
What is the current value of the US Fusion Biopsy Market?
The US Fusion Biopsy Market is valued at approximately USD 220 million, driven by the rising prevalence of prostate cancer and advancements in imaging technologies, particularly AI-enhanced image-fusion capabilities.