Region:North America
Author(s):Rebecca
Product Code:KRAC9638
Pages:97
Published On:November 2025
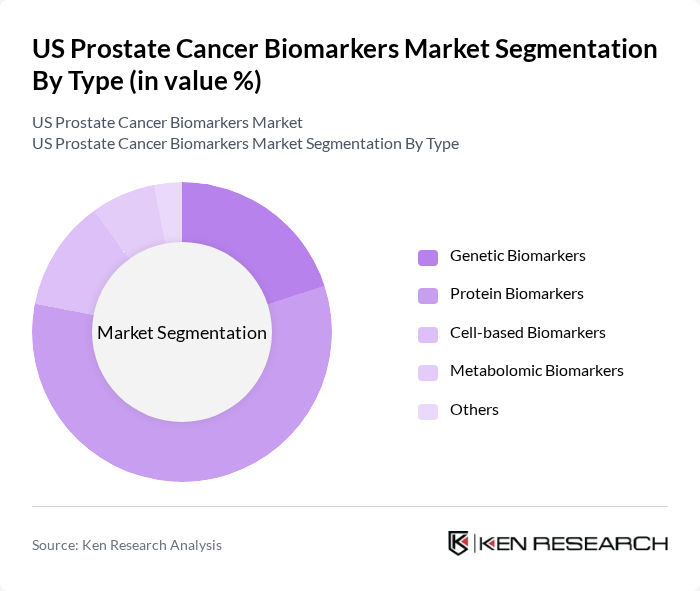
By Type:The market is segmented into various types of biomarkers, including Genetic Biomarkers, Protein Biomarkers, Cell-based Biomarkers, Metabolomic Biomarkers, and Others. Among these, Protein Biomarkers, particularly PSA, dominate the market due to their established role in screening and monitoring. Genetic Biomarkers are gaining traction for identifying hereditary prostate cancer risks and guiding treatment decisions. Cell-based and Metabolomic Biomarkers are emerging as innovative tools for personalized therapy, with metabolomic approaches showing rapid growth due to their ability to profile disease-related metabolic changes.
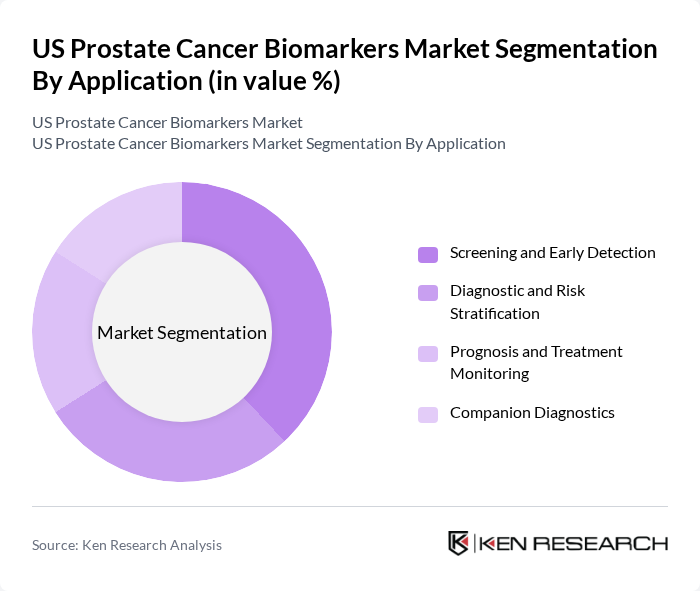
By Application:The applications of prostate cancer biomarkers include Screening and Early Detection, Diagnostic and Risk Stratification, Prognosis and Treatment Monitoring, and Companion Diagnostics. Screening and Early Detection applications are particularly significant as they help identify prostate cancer at an early stage, leading to better treatment outcomes. The increasing focus on personalized medicine is driving the demand for Companion Diagnostics, which tailor treatment based on individual biomarker profiles. Diagnostic and Risk Stratification applications are also expanding, as clinicians seek to optimize therapy selection and monitor disease progression more precisely.
The US Prostate Cancer Biomarkers Market is characterized by a dynamic mix of regional and international players. Leading participants such as Myriad Genetics, Exact Sciences, Veracyte, BioReference Laboratories, Foundation Medicine, Caris Life Sciences, Siemens Healthineers, Abbott Laboratories, Roche Diagnostics, Illumina, LabCorp, Quest Diagnostics, OPKO Health, MDxHealth, and Hologic contribute to innovation, geographic expansion, and service delivery in this space.
The future of the US prostate cancer biomarkers market appears promising, driven by technological advancements and a growing emphasis on personalized medicine. As the healthcare landscape shifts towards more tailored treatment approaches, the integration of artificial intelligence in biomarker analysis is expected to enhance diagnostic accuracy. Additionally, the increasing demand for non-invasive testing methods will likely spur innovation, leading to the development of more efficient and patient-friendly diagnostic solutions in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Genetic Biomarkers Protein Biomarkers Cell-based Biomarkers Metabolomic Biomarkers Others |
| By Application | Screening and Early Detection Diagnostic and Risk Stratification Prognosis and Treatment Monitoring Companion Diagnostics |
| By End-User | Hospitals & Diagnostic Laboratories Academic & Research Institutes Biopharmaceutical Companies Others |
| By Biomarker Class | PSA (Prostate-Specific Antigen) PCA3 (Prostate Cancer Antigen 3) Kscore Others |
| By Sample Type | Blood Samples Urine Samples Tissue Samples Others |
| By Region | Northeast Midwest South West |
| By Policy Support | Government Grants Tax Incentives Research Funding Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncologist Insights | 100 | Medical Oncologists, Urologists |
| Laboratory Perspectives | 60 | Laboratory Managers, Pathologists |
| Patient Awareness Surveys | 120 | Prostate Cancer Patients, Caregivers |
| Healthcare Policy Analysis | 50 | Healthcare Administrators, Policy Makers |
| Biomarker Manufacturer Feedback | 40 | Product Managers, Sales Executives |
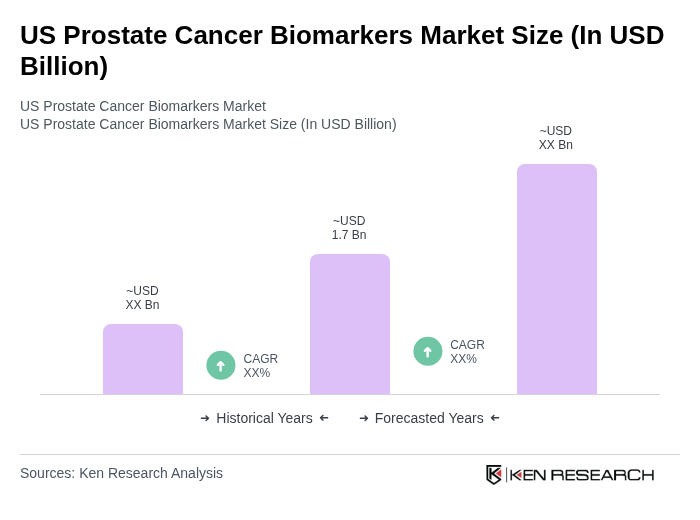
The US Prostate Cancer Biomarkers Market is valued at approximately USD 1.7 billion, driven by factors such as the increasing prevalence of prostate cancer and advancements in biomarker technology, including artificial intelligence integration and liquid biopsy innovations.