Region:North America
Author(s):Shubham
Product Code:KRAD2508
Pages:95
Published On:January 2026
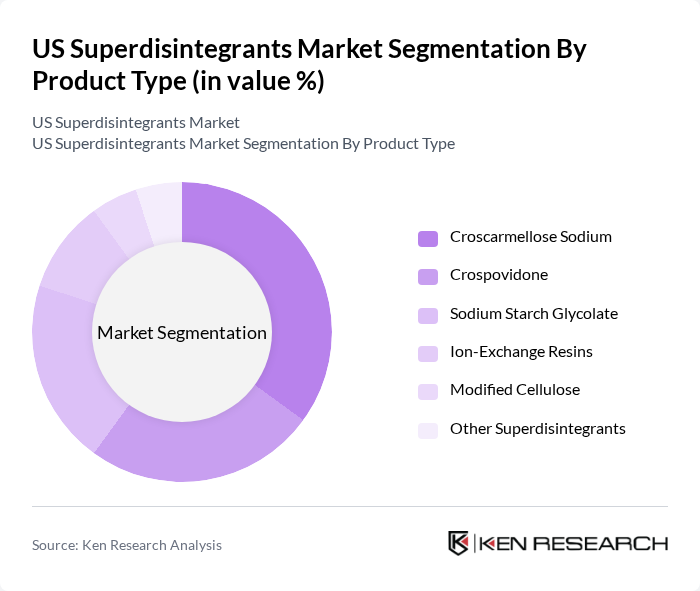
By Product Type:The product type segmentation includes various superdisintegrants that are essential for enhancing the disintegration of tablets and capsules. The leading subsegment is Croscarmellose Sodium, which holds the largest share in global usage and is widely adopted in both direct compression and wet granulation because of its strong swelling and wicking properties, enabling rapid tablet disintegration even at low concentrations. Crospovidone and Sodium Starch Glycolate also hold significant market shares due to their proven effectiveness across immediate-release formulations and their broad pharmacopeial acceptance. Ion-Exchange Resins and Modified Cellulose are gaining traction as they offer unique benefits, such as modulating disintegration in challenging formulations and improving mouthfeel or stability in certain oral dosage forms, while Other Superdisintegrants cater to niche applications, including specialized fast-dissolving and pediatric formulations.
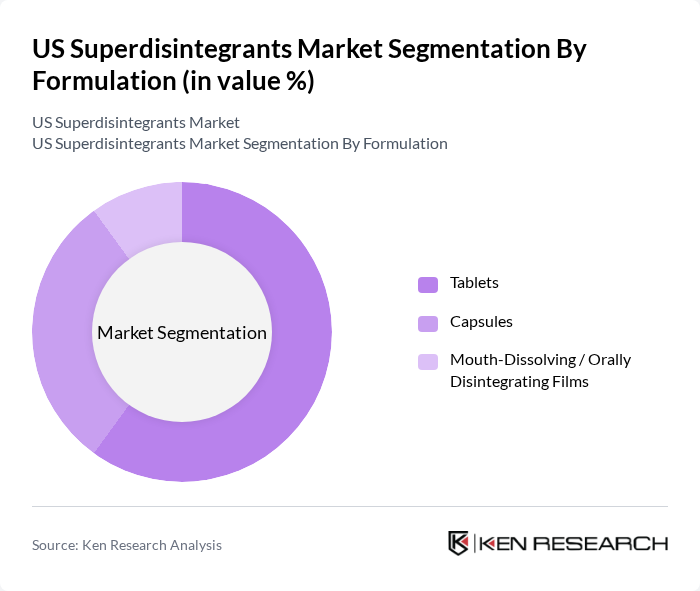
By Formulation:The formulation segmentation highlights the various forms in which superdisintegrants are utilized, including tablets, capsules, and mouth-dissolving or orally disintegrating films. Tablets dominate this segment due to their widespread use, cost-effectiveness, and the high share of tablets and capsules in global superdisintegrant consumption, reflecting strong preference for solid oral dosage forms in both branded and generic markets. Capsules are also popular, particularly for dietary supplements, high-value APIs, and products requiring flexible fill combinations. Mouth-dissolving and orally disintegrating films are emerging as convenient alternatives for pediatric, geriatric, and dysphagic populations, aligning with the broader trend toward patient-centric dosage forms and improved adherence.
The US Superdisintegrants Market is characterized by a dynamic mix of regional and international players. Leading participants such as Ashland Global Holdings Inc., BASF SE, Evonik Industries AG, JRS Pharma GmbH & Co. KG, FMC Corporation, Colorcon, Inc., DuPont de Nemours, Inc., Merck KGaA, Croda International Plc, Ingredion Incorporated, Roquette Frères, Sigma-Aldrich (Merck Group), HPM Chemicals & Fertilizers Ltd., Sentaur Chemicals Inc., Polyplastics Co., Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
The US superdisintegrants market is poised for significant growth, driven by the increasing demand for efficient drug formulations and the rise of generic drug production. As technological advancements continue to reshape drug delivery systems, the integration of innovative superdisintegrants will become essential. Additionally, the focus on personalized medicine will further propel the need for tailored formulations, creating a dynamic landscape for superdisintegrants in the coming years. The market is expected to adapt to these trends, fostering new opportunities for growth and development.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Croscarmellose Sodium Crospovidone Sodium Starch Glycolate Ion-Exchange Resins Modified Cellulose Other Superdisintegrants |
| By Formulation | Tablets Capsules Mouth-Dissolving / Orally Disintegrating Films |
| By Therapeutic Area | Gastrointestinal Diseases Neurological Diseases Oncology Infectious Diseases Cardiovascular Diseases Hematological Diseases Inflammatory Diseases Other Therapeutic Areas |
| By Customer Type | Originator / Branded Pharmaceutical Companies Generic Drug Manufacturers Contract Development and Manufacturing Organizations (CDMOs/CMOs) Research and Academic Institutions Other Industrial Users |
| By Superdisintegrant Source | Synthetic Superdisintegrants Natural Superdisintegrants Co-Processed Superdisintegrants |
| By US Region | Northeast Midwest South West |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Formulation Development | 120 | Formulation Scientists, R&D Managers |
| Excipient Procurement Strategies | 90 | Procurement Managers, Supply Chain Analysts |
| Market Trends in Drug Delivery | 80 | Market Analysts, Product Managers |
| Regulatory Compliance in Excipients | 60 | Regulatory Affairs Specialists, Quality Assurance Managers |
| Innovations in Superdisintegrants | 70 | Research Scientists, Industry Experts |
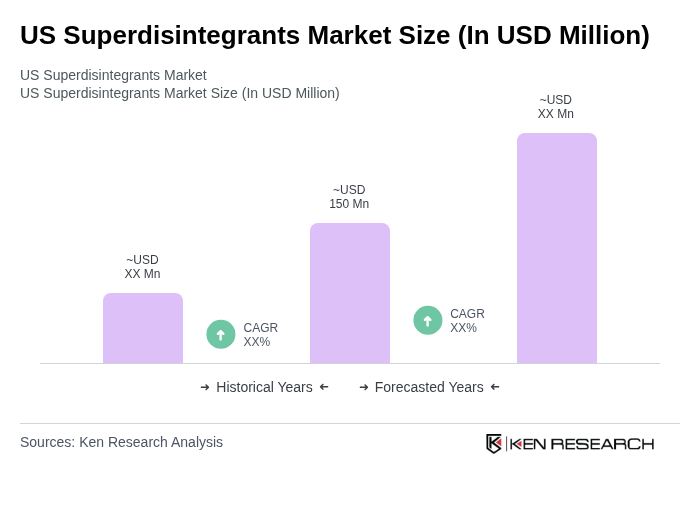
The US Superdisintegrants Market is valued at approximately USD 150 million, driven by the increasing demand for efficient oral solid drug formulations and the growing prevalence of chronic diseases.