Region:North America
Author(s):Rebecca
Product Code:KRAB5999
Pages:84
Published On:October 2025
By Type:The cold chain market is segmented into various types, including Refrigerated Transport, Temperature-Controlled Warehousing, Packaging Solutions, Monitoring Systems, Logistics Management Services, and Others. Each of these segments plays a crucial role in ensuring the safe and effective delivery of temperature-sensitive biologics and specialty drugs.
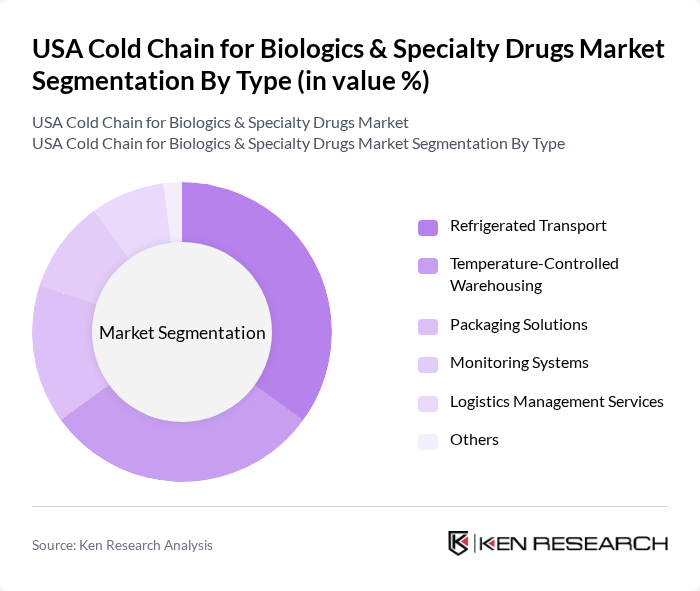
The Refrigerated Transport segment is currently dominating the market due to the increasing need for efficient and reliable transportation of temperature-sensitive products. This segment is essential for maintaining the cold chain from manufacturers to end-users, ensuring that biologics and specialty drugs remain within the required temperature ranges. The rise in e-commerce and direct-to-consumer delivery models has further amplified the demand for refrigerated transport solutions, making it a critical component of the cold chain logistics ecosystem.
By End-User:The market is segmented by end-users, including Pharmaceutical Companies, Biotechnology Firms, Research Institutions, Hospitals and Clinics, and Others. Each of these end-users has specific requirements for cold chain logistics, driven by the nature of the products they handle.
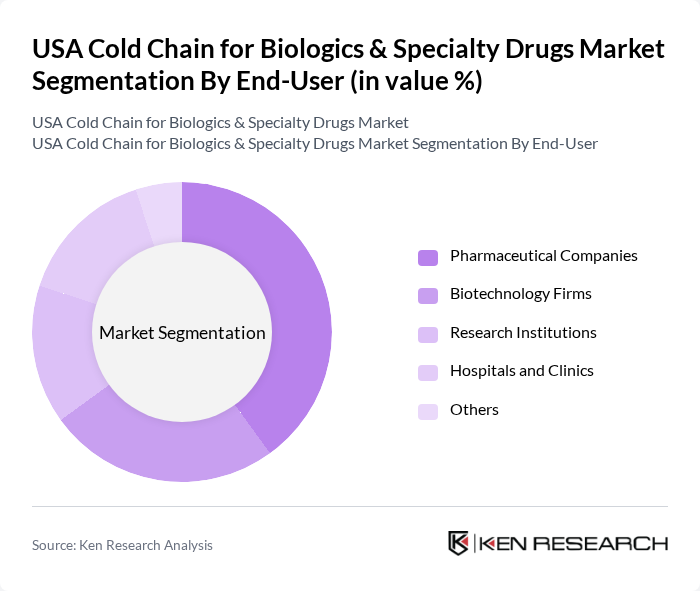
Pharmaceutical Companies are the leading end-users in the cold chain market, primarily due to their extensive product portfolios that include a wide range of biologics and specialty drugs. These companies require robust cold chain solutions to ensure compliance with regulatory standards and maintain product efficacy. The increasing focus on research and development in the pharmaceutical sector further drives the demand for cold chain logistics, as new therapies and vaccines are developed and brought to market.
The USA Cold Chain for Biologics & Specialty Drugs Market is characterized by a dynamic mix of regional and international players. Leading participants such as AmerisourceBergen Corporation, Cardinal Health, Inc., DHL Supply Chain, FedEx Corporation, UPS Healthcare, Thermo Fisher Scientific Inc., McKesson Corporation, Lineage Logistics, Cold Chain Technologies, BioLife Solutions, Inc., Pelican BioThermal, Envirotainer, Cryoport, Inc., Inmark Packaging, Softbox Systems contribute to innovation, geographic expansion, and service delivery in this space.
The future of the cold chain logistics market for biologics and specialty drugs in the U.S. appears promising, driven by technological innovations and increasing healthcare demands. As the market adapts to the rise of personalized medicine and e-commerce, companies are likely to invest in advanced monitoring systems and sustainable practices. Additionally, the integration of AI for predictive analytics will enhance operational efficiency, ensuring that products are delivered safely and effectively, thereby meeting the evolving needs of healthcare providers and patients alike.
| Segment | Sub-Segments |
|---|---|
| By Type | Refrigerated Transport Temperature-Controlled Warehousing Packaging Solutions Monitoring Systems Logistics Management Services Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Research Institutions Hospitals and Clinics Others |
| By Distribution Mode | Direct Distribution Third-Party Logistics E-commerce Platforms Others |
| By Packaging Type | Insulated Containers Refrigerated Pallets Temperature-Controlled Boxes Others |
| By Service Type | Transportation Services Storage Services Monitoring Services Others |
| By Temperature Range | Controlled Room Temperature Refrigerated (2-8°C) Frozen (-20°C and below) Others |
| By Application | Vaccines Gene Therapies Monoclonal Antibodies Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biologics Distribution Logistics | 100 | Supply Chain Managers, Logistics Coordinators |
| Specialty Drug Cold Chain Management | 80 | Pharmacy Directors, Operations Managers |
| Temperature-Controlled Storage Solutions | 70 | Warehouse Managers, Facility Operations Heads |
| Regulatory Compliance in Cold Chain | 60 | Quality Assurance Managers, Compliance Officers |
| Healthcare Provider Perspectives on Cold Chain | 90 | Healthcare Administrators, Pharmacists |
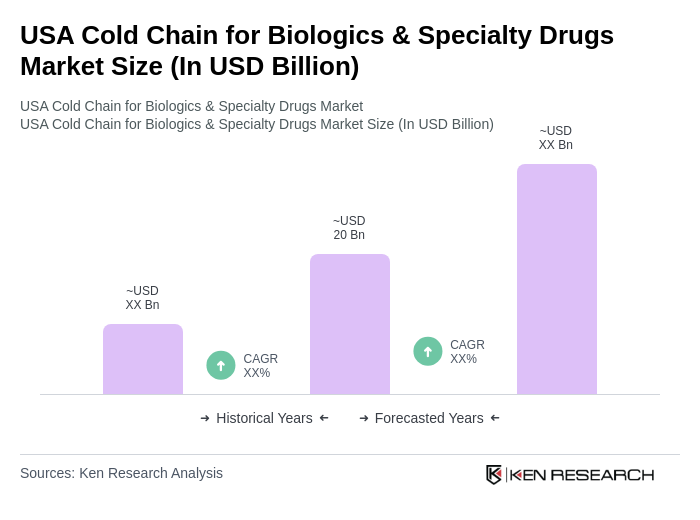
The USA Cold Chain for Biologics & Specialty Drugs Market is valued at approximately USD 20 billion, driven by the increasing demand for biologics and specialty drugs that require stringent temperature control during storage and transportation.