USA Gastrointestinal GI Stents Market Overview
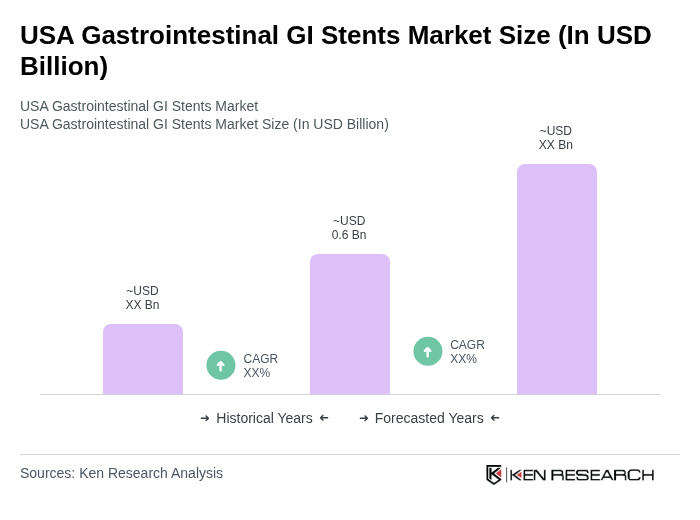
- The USA Gastrointestinal GI Stents Market is valued at USD 0.6 billion, based on a five?year historical analysis. Growth is driven by the rising prevalence of gastrointestinal cancers and lifestyle-related digestive disorders alongside increased healthcare expenditure and preference for minimally invasive endoscopic interventions. Advances such as drug-eluting, biodegradable and anti-migration stent designs further propel adoption, supported by improved clinical outcomes and procedural efficiency.
- Key players in this market include Boston Scientific, Cook Medical, Becton Dickinson, Taewoong Medical, Olympus and Merit Medical, which drive innovation through durable R&D pipelines and strategic expansion. Boston Scientific’s leadership is underscored by its WallFlex and AXIOS product lineup, while Cook Medical’s strength lies in minimally invasive designs, and other players contribute through visualization-integrated systems and regional manufacturing capabilities.
- In 2024, the U.S. Food and Drug Administration announced specific guidance for metal expandable biliary stents under the 510(k) pathway, detailing performance standards for safety and efficacy in biliary applications—pivotal for GI stent approvals and market entry.
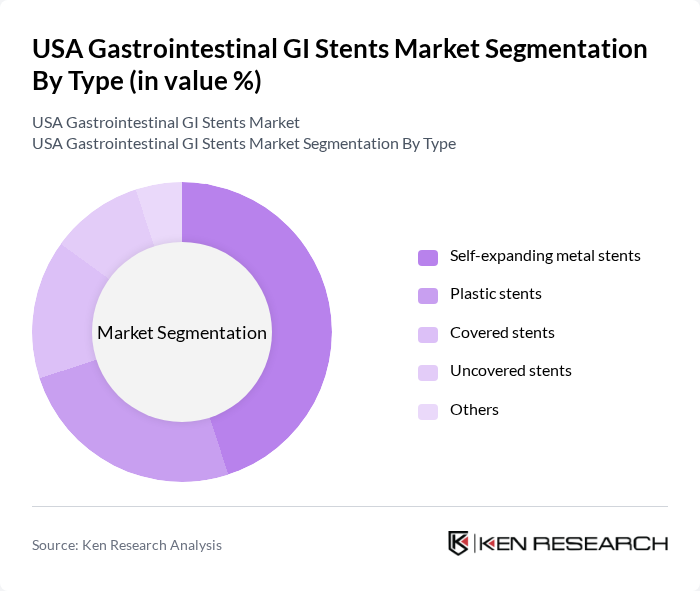
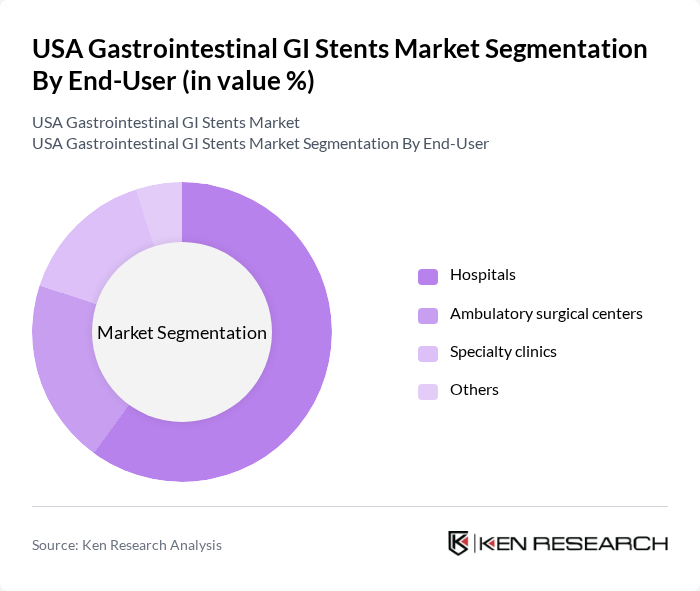
USA Gastrointestinal GI Stents Market Segmentation
By Type:The market is segmented into various types of stents, including self-expanding metal stents, plastic stents, covered stents, uncovered stents, and others. Among these, self-expanding metal stents dominate the market due to their superior flexibility and ability to adapt to the anatomy of the gastrointestinal tract, making them a preferred choice for many clinicians. The increasing adoption of these stents is also driven by advancements in design and materials, enhancing patient outcomes.
By End-User:The end-user segmentation includes hospitals, ambulatory surgical centers, specialty clinics, and others. Hospitals are the leading end-users, primarily due to their comprehensive facilities and access to advanced medical technologies. The increasing number of surgical procedures performed in hospitals, coupled with the growing patient population requiring gastrointestinal interventions, further solidifies their dominance in the market.
USA Gastrointestinal GI Stents Market Competitive Landscape
The USA Gastrointestinal GI Stents Market is characterized by a dynamic mix of regional and international players. Leading participants such as Boston Scientific Corporation, Medtronic plc, Cook Medical, Abbott Laboratories, B. Braun Melsungen AG, Olympus Corporation, ConMed Corporation, Merit Medical Systems, Inc., EndoChoice, Inc., Stryker Corporation, C. R. Bard, Inc., Halyard Health, Inc., Ethicon, Inc., Terumo Corporation, Medline Industries, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
USA Gastrointestinal GI Stents Market Industry Analysis
Growth Drivers
- Annual ERCP Procedures in the U.S.:Over 650,000 ERCP (endoscopic retrograde cholangiopancreatography) procedures are performed annually in the U.S. as of 2022. This high procedural volume directly supports the demand for gastrointestinal stents, particularly biliary stents. The increasing number of procedures correlates with a growing patient population suffering from gastrointestinal diseases, thereby driving the market for GI stents significantly ((https://www.fortunebusinessinsights.com/gastrointestinal-stent-market-114932?utm_source=openai)).
- North America Market Value (2024):The North American GI stents market was valued at USD 245.0 million in the future. This substantial market value reinforces the U.S. leadership in the GI stents sector, driven by advanced healthcare infrastructure and high adoption rates of innovative stent technologies. The robust healthcare system facilitates the availability and accessibility of GI stents, further propelling market growth ((https://www.fortunebusinessinsights.com/gastrointestinal-stent-market-114932?utm_source=openai)).
- U.S. Upper GI Stent Market Value (2024):The U.S. upper GI stent market is projected to reach USD 300 million in the future. This significant portion of the overall GI stents market highlights the focus on upper gastrointestinal interventions, which are increasingly performed due to rising incidences of related diseases. The demand for effective treatment options in this segment is expected to drive further innovations and market expansion ((https://www.linkedin.com/pulse/united-states-upper-gastrointestinal-stent-market-rkhbe/?utm_source=openai)).
Market Challenges
- High Cost of Stents per Unit:The price range for GI stents varies between USD 800 and USD 2,500 per unit. This elevated cost presents a significant barrier to broader adoption, particularly in cost-sensitive healthcare settings. The financial burden associated with purchasing stents can deter healthcare providers from utilizing these devices, thereby limiting market penetration and growth opportunities ((https://www.datamintelligence.com/research-report/gastrointestinal-stents-market?utm_source=openai)).
- High Procedure Cost:The cost of endoscopic stent placement procedures ranges from USD 3,000 to USD 10,000. These significant expenses, combined with variable reimbursement policies, create challenges for patients and healthcare providers alike. The high costs can limit access to necessary treatments, thereby impacting the overall growth of the GI stents market in the U.S. ((https://www.datamintelligence.com/research-report/gastrointestinal-stents-market?utm_source=openai)).
USA Gastrointestinal GI Stents Market Future Outlook
The future of the USA gastrointestinal GI stents market appears promising, driven by technological advancements and increasing healthcare accessibility. The adoption of smart and biodegradable stents is expected to enhance patient outcomes, while the expansion of ambulatory surgical centers (ASCs) will facilitate quicker access to GI stenting procedures. As the healthcare landscape evolves, these trends will likely foster innovation and improve the overall efficiency of gastrointestinal treatments, positioning the market for sustained growth in the coming years.
Market Opportunities
- ASC-Focused Procurement:The growth of ambulatory surgical centers (6,308 in the future) presents a unique opportunity for targeted procurement strategies. Manufacturers can tailor their product offerings to meet the specific needs of ASCs, where the adoption of advanced stents is on the rise, thereby enhancing market penetration and revenue potential ((https://www.fortunebusinessinsights.com/gastrointestinal-stent-market-114932?utm_source=openai)).
- North America Leadership:North America, with a market value of USD 245 million in the future, continues to lead the GI stents market. This dominance provides a solid foundation for innovation and specialization, allowing companies to capitalize on the region's advanced healthcare infrastructure and high demand for effective gastrointestinal treatments ((https://www.fortunebusinessinsights.com/gastrointestinal-stent-market-114932?utm_source=openai)).