Region:Asia
Author(s):Geetanshi
Product Code:KRAD4772
Pages:87
Published On:December 2025
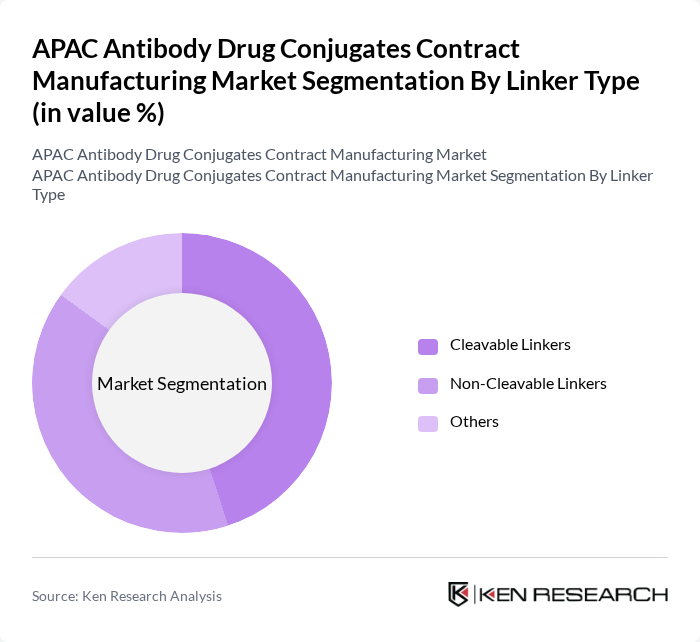
By Linker Type:The market is segmented into Cleavable Linkers, Non-Cleavable Linkers, and Others. Cleavable linkers are gaining traction due to their ability to release the drug in a controlled and tumor?selective manner, enhancing therapeutic efficacy and reducing systemic toxicity. Non?cleavable linkers, while highly stable in circulation, are also significant as they provide a consistent and durable release profile once internalized and degraded in target cells. The "Others" category includes various innovative linker technologies, such as enzymatically cleavable and site?specific conjugation linkers, that are emerging in the market to improve payload stability, homogeneity, and therapeutic index.
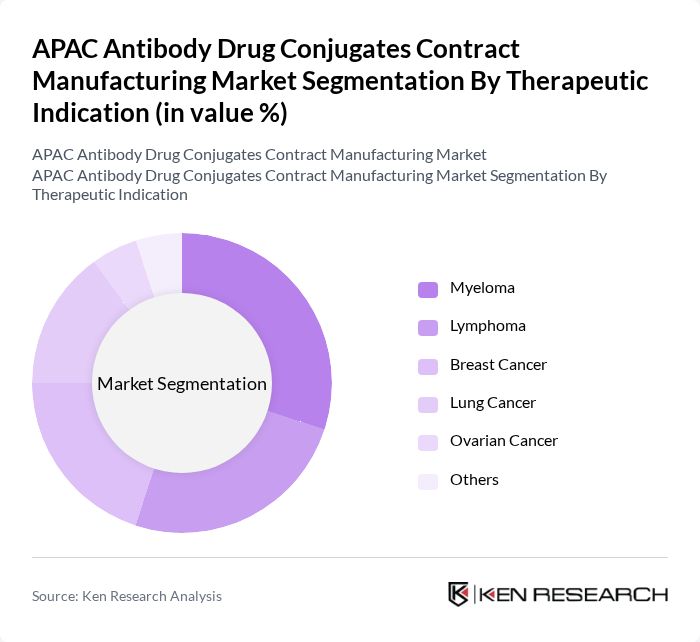
By Therapeutic Indication:The market is categorized into Myeloma, Lymphoma, Breast Cancer, Lung Cancer, Ovarian Cancer, and Others. Myeloma and Lymphoma are leading indications due to the high incidence of hematologic malignancies in Asia Pacific and the established clinical effectiveness of antibody drug conjugates in these cancers. Breast and Lung cancers also represent significant segments, driven by the high disease burden in major APAC countries and the increasing number of ADCs and other targeted therapies being developed and clinically evaluated for these indications.
The APAC Antibody Drug Conjugates Contract Manufacturing Market is characterized by a dynamic mix of regional and international players. Leading participants such as WuXi Biologics (Cayman) Inc., Lonza Group Ltd., Samsung BioLogics, Recipharm, Catalent Inc., Sartorius AG, Piramal Healthcare, AbbVie Inc., Merck KGaA, AstraZeneca, Daiichi Sankyo, Takeda Pharmaceutical Company Limited, Pfizer Inc., Amgen Inc., Sterling Check Corp. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the APAC antibody drug conjugates contract manufacturing market appears promising, driven by ongoing innovations and increasing investments in biopharmaceuticals. As the demand for personalized medicine continues to rise, manufacturers are expected to adopt advanced technologies, including AI and automation, to enhance production efficiency. Additionally, collaborations between pharmaceutical companies and contract manufacturers are likely to increase, fostering the development of novel ADCs that address unmet medical needs in the region.
| Segment | Sub-Segments |
|---|---|
| By Linker Type | Cleavable Linkers Non-Cleavable Linkers Others |
| By Therapeutic Indication | Myeloma Lymphoma Breast Cancer Lung Cancer Ovarian Cancer Others |
| By ADC Generation | First Generation ADCs Second Generation ADCs Third Generation ADCs |
| By Manufacturing Process | Batch Production Continuous Production Others |
| By Geography | China Japan India South Korea Taiwan Others |
| By End-User | Pharmaceutical Companies Biotech Firms Contract Development and Manufacturing Organizations (CDMOs) Contract Manufacturing Organizations (CMOs) Research Institutions |
| By Service Type | Development Services Commercial Manufacturing Fill-Finish Services |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Drug Development | 120 | Clinical Researchers, Oncology Specialists |
| Contract Manufacturing for ADCs | 85 | CMO Executives, Production Managers |
| Regulatory Compliance in ADC Manufacturing | 55 | Regulatory Affairs Managers, Quality Control Officers |
| Market Access and Pricing Strategies | 65 | Market Access Managers, Pricing Analysts |
| Clinical Trial Management for ADCs | 95 | Clinical Trial Managers, Biostatisticians |
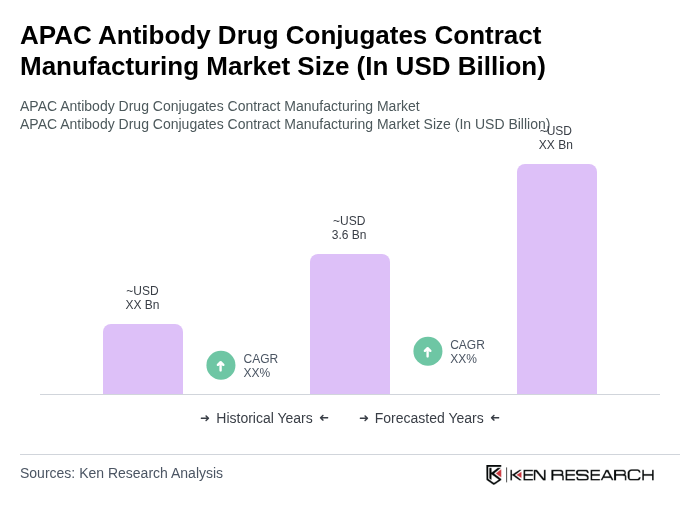
The APAC Antibody Drug Conjugates Contract Manufacturing Market is valued at approximately USD 3.6 billion, reflecting significant growth driven by the increasing prevalence of cancer and the demand for targeted therapies that enhance treatment efficacy while minimizing side effects.