About the Report
Base Year 2024Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market Overview
- The Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market is valued at USD 1.0 billion, based on a five-year historical analysis and triangulation of the Australia glass syringes market (around USD 245 million) with broader prefilled syringes, primary packaging, and pharmaceutical packaging equipment spending in the country. This growth is primarily driven by the increasing demand for biologics and biosimilars, alongside the rising prevalence of chronic and lifestyle-related diseases such as diabetes, cancer, and autoimmune disorders that require injectable therapies. The market is also supported by advancements in aseptic filling, automation, and inspection technologies that enhance the safety, sterility assurance, and efficiency of drug delivery systems and compliant packaging lines.
- Key cities such as Sydney and Melbourne dominate the market due to their robust healthcare infrastructure, large hospital networks, and concentration of pharmaceutical, biotechnology, and contract manufacturing organizations, including facilities clustered in New South Wales and Victoria. These urban centers are hubs for innovation and research, attracting investments in healthcare, biopharmaceutical R&D, and advanced manufacturing, which further propels the demand for glass prefilled syringes, vials, and associated high?performance packaging equipment.
- The regulatory environment for injectable medicines and their packaging in Australia is governed by the Therapeutic Goods Administration (TGA) under the Therapeutic Goods Act 1989 and associated Therapeutic Goods (Manufacturing Principles) Determination 2020, which adopts the PIC/S Guide to Good Manufacturing Practice for Medicinal Products. These instruments require sponsors and manufacturers of sterile injectable products to implement controls such as tamper-evident features, container–closure integrity testing, validated sterilisation, and traceable labelling for finished dosage forms, thereby driving the demand for advanced, compliant packaging solutions for glass prefilled syringes and vials in the market.

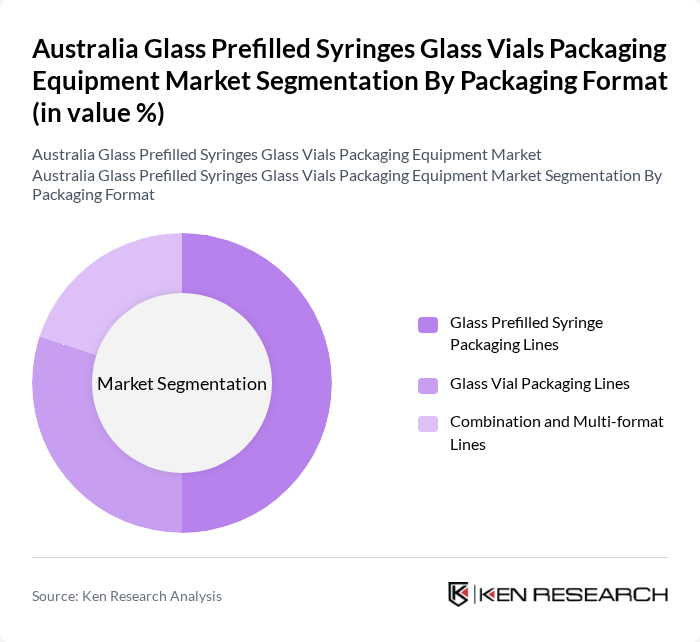
Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market Segmentation
By Equipment Type:The equipment type segmentation includes various categories essential for the packaging process. The dominant sub-segment is Filling and Stoppering Equipment, which is crucial for ensuring accurate dosage, minimizing particulate contamination, and maintaining sterility during the aseptic filling of glass prefilled syringes and vials. This segment is driven by the increasing production of injectable drugs and biologics, the shift toward ready?to?use containers, and the need for high?throughput, fully automated filling solutions that comply with global GMP and inspection standards. Other notable segments include Washing and Sterilization Equipment, which ensures container cleanliness and depyrogenation before filling, and Capping and Sealing Equipment, which is vital for container?closure integrity, tamper evidence, and protection of sensitive formulations throughout their shelf life.

By Packaging Format:The packaging format segmentation includes Glass Prefilled Syringe Packaging Lines, Glass Vial Packaging Lines, and Combination and Multi-format Lines. The Glass Prefilled Syringe Packaging Lines segment is leading, aligned with the broader trend that glass remains the preferred material for prefilled syringes due to its superior barrier properties and compatibility with biologics, as well as the growing preference among healthcare providers and patients for unit?dose, ready?to?inject formats that offer convenience and reduce the risk of dosing errors. The demand for Glass Vial Packaging Lines is also significant, driven by their extensive use in vaccines, oncology drugs, and hospital pharmacy preparations, while Combination and Multi?format Lines are gaining importance as manufacturers seek flexibility to handle different container types and sizes on a single automated platform.
Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market Competitive Landscape
The Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market is characterized by a dynamic mix of regional and international players. Leading participants such as Schott AG, Gerresheimer AG, Stevanato Group S.p.A (including Ompi), West Pharmaceutical Services, Inc., Nipro Corporation, Becton, Dickinson and Company, AptarGroup, Inc., SGD Pharma, Syntegon Technology GmbH, IMA Group, Romaco Group, Bausch+Ströbel Maschinenfabrik Ilshofen GmbH+Co. KG, Optima packaging group GmbH, Marchesini Group S.p.A., Dara Pharmaceutical Packaging contribute to innovation, geographic expansion, and service delivery in this space.
Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market Industry Analysis
Growth Drivers
- Increasing Demand for Biologics:The Australian biologics market is projected to reach AUD 10 billion in future, driven by the rising prevalence of chronic diseases. This surge in demand for biologics necessitates advanced packaging solutions, particularly glass prefilled syringes and vials, which ensure product integrity and safety. The Australian government’s investment in healthcare innovation, estimated at AUD 1.5 billion in future, further supports this growth, emphasizing the need for reliable packaging technologies in the biologics sector.
- Advancements in Packaging Technology:The packaging equipment sector is witnessing significant technological advancements, with investments in automation and smart packaging solutions expected to exceed AUD 500 million in future. These innovations enhance efficiency and reduce human error, crucial for maintaining the sterility of glass prefilled syringes and vials. Additionally, the integration of IoT technologies in packaging is projected to improve traceability and compliance, aligning with the increasing regulatory demands in the pharmaceutical industry.
- Rising Healthcare Expenditure:Australia’s healthcare expenditure is anticipated to reach AUD 200 billion in future, reflecting a 5% annual growth rate. This increase is driven by an aging population and a growing focus on preventive care. As healthcare spending rises, pharmaceutical companies are investing more in packaging solutions that enhance patient safety and convenience, such as glass prefilled syringes, which are preferred for their ease of use and reduced risk of contamination, thereby driving market growth.
Market Challenges
- High Production Costs:The production costs for glass prefilled syringes and vials are significantly high, with estimates reaching AUD 0.50 per unit in future. This is primarily due to the expensive raw materials and the intricate manufacturing processes involved. As a result, smaller manufacturers may struggle to compete with larger firms that benefit from economies of scale, potentially limiting market entry and innovation in the sector.
- Stringent Regulatory Requirements:The Australian Therapeutic Goods Administration (TGA) imposes rigorous regulations on medical devices, including packaging solutions. Compliance with these regulations often requires extensive testing and validation, which can cost upwards of AUD 1 million per product line. This regulatory burden can deter new entrants and slow down the introduction of innovative packaging solutions, posing a significant challenge to market growth.
Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market Future Outlook
The future of the Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market appears promising, driven by ongoing innovations and a shift towards sustainable practices. As the demand for biologics continues to rise, manufacturers are likely to invest in advanced packaging technologies that enhance safety and efficiency. Additionally, the increasing focus on personalized medicine will further propel the need for tailored packaging solutions, ensuring that the market remains dynamic and responsive to evolving healthcare needs.
Market Opportunities
- Expansion in Emerging Markets:The growing healthcare infrastructure in emerging markets presents significant opportunities for Australian manufacturers. With an expected increase in healthcare spending in these regions, estimated at AUD 50 billion in future, there is a rising demand for reliable packaging solutions, including glass prefilled syringes and vials, which can cater to diverse healthcare needs.
- Innovations in Sustainable Packaging:The shift towards eco-friendly packaging solutions is gaining momentum, with the Australian government aiming for a 70% recycling rate in future. This trend opens avenues for manufacturers to develop sustainable glass packaging options, appealing to environmentally conscious consumers and aligning with regulatory expectations, thereby enhancing market competitiveness.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Equipment Type | Washing and Sterilization Equipment Filling and Stoppering Equipment Capping and Sealing Equipment Inspection and Leak Detection Equipment Labelling and Coding Equipment Secondary and Tertiary Packaging Equipment |
| By Packaging Format | Glass Prefilled Syringe Packaging Lines Glass Vial Packaging Lines Combination and Multi-format Lines |
| By Automation Level | Manual and Semi-automatic Equipment Fully Automatic Equipment Robotics and High-throughput Systems |
| By Filling Technology | Volumetric and Peristaltic Filling Systems Aseptic and Barrier Isolator Systems Ready-to-use (RTU) and Nested Container Systems |
| By End-User | Pharmaceutical Manufacturers Biotechnology and Biologics Manufacturers Contract Development and Manufacturing Organizations (CDMOs/CMOs) Hospital and Compounding Pharmacies Others |
| By Application | Vaccines and Immunization Products Biologics and Biosimilars Oncology and Specialty Injectables Autoimmune and Chronic Disease Therapies Others |
| By Region | New South Wales Victoria Queensland Western Australia South Australia Rest of Australia |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Therapeutic Goods Administration, Australian Competition and Consumer Commission)
Manufacturers and Producers of Glass Prefilled Syringes and Vials
Pharmaceutical Companies
Biotechnology Firms
Packaging Equipment Suppliers
Logistics and Supply Chain Companies
Quality Assurance and Compliance Organizations
Players Mentioned in the Report:
Schott AG
Gerresheimer AG
Stevanato Group S.p.A (including Ompi)
West Pharmaceutical Services, Inc.
Nipro Corporation
Becton, Dickinson and Company
AptarGroup, Inc.
SGD Pharma
Syntegon Technology GmbH
IMA Group
Romaco Group
Bausch+Strobel Maschinenfabrik Ilshofen GmbH+Co. KG
Optima packaging group GmbH
Marchesini Group S.p.A.
Dara Pharmaceutical Packaging
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for biologics
3.1.2 Advancements in packaging technology
3.1.3 Rising healthcare expenditure
3.1.4 Growing focus on patient safety and convenience
3.2 Market Challenges
3.2.1 High production costs
3.2.2 Stringent regulatory requirements
3.2.3 Competition from alternative packaging solutions
3.2.4 Supply chain disruptions
3.3 Market Opportunities
3.3.1 Expansion in emerging markets
3.3.2 Innovations in sustainable packaging
3.3.3 Collaborations with pharmaceutical companies
3.3.4 Increasing adoption of automated packaging solutions
3.4 Market Trends
3.4.1 Shift towards eco-friendly materials
3.4.2 Growth of personalized medicine
3.4.3 Integration of smart packaging technologies
3.4.4 Rise in demand for prefilled syringes
3.5 Government Regulation
3.5.1 TGA regulations on medical devices
3.5.2 Compliance with ISO standards
3.5.3 Guidelines for packaging materials
3.5.4 Environmental regulations on waste management
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market Segmentation
8.1 By Equipment Type
8.1.1 Washing and Sterilization Equipment
8.1.2 Filling and Stoppering Equipment
8.1.3 Capping and Sealing Equipment
8.1.4 Inspection and Leak Detection Equipment
8.1.5 Labelling and Coding Equipment
8.1.6 Secondary and Tertiary Packaging Equipment
8.2 By Packaging Format
8.2.1 Glass Prefilled Syringe Packaging Lines
8.2.2 Glass Vial Packaging Lines
8.2.3 Combination and Multi-format Lines
8.3 By Automation Level
8.3.1 Manual and Semi-automatic Equipment
8.3.2 Fully Automatic Equipment
8.3.3 Robotics and High-throughput Systems
8.4 By Filling Technology
8.4.1 Volumetric and Peristaltic Filling Systems
8.4.2 Aseptic and Barrier Isolator Systems
8.4.3 Ready-to-use (RTU) and Nested Container Systems
8.5 By End-User
8.5.1 Pharmaceutical Manufacturers
8.5.2 Biotechnology and Biologics Manufacturers
8.5.3 Contract Development and Manufacturing Organizations (CDMOs/CMOs)
8.5.4 Hospital and Compounding Pharmacies
8.5.5 Others
8.6 By Application
8.6.1 Vaccines and Immunization Products
8.6.2 Biologics and Biosimilars
8.6.3 Oncology and Specialty Injectables
8.6.4 Autoimmune and Chronic Disease Therapies
8.6.5 Others
8.7 By Region
8.7.1 New South Wales
8.7.2 Victoria
8.7.3 Queensland
8.7.4 Western Australia
8.7.5 South Australia
8.7.6 Rest of Australia
9. Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Australia Market Revenue (USD Million)
9.2.4 3-year Revenue CAGR in Australia (%)
9.2.5 EBITDA Margin (%)
9.2.6 Return on Invested Capital (ROIC, %)
9.2.7 Capex Intensity (% of Revenue)
9.2.8 Market Share in Glass Prefilled Syringe Lines (%)
9.2.9 Market Share in Glass Vial Packaging Lines (%)
9.2.10 Installed Base in Australia (Number of Lines)
9.2.11 Order Backlog / Book-to-Bill Ratio
9.2.12 Average Selling Price per Line (USD)
9.2.13 On-time Delivery Rate (%)
9.2.14 Line OEE at Customer Sites (Average %)
9.2.15 R&D Intensity (% of Revenue)
9.2.16 New Product Launches (Last 3 Years)
9.2.17 Geographic Coverage within Australia (States/Regions Served)
9.2.18 Key Strategic Partnerships and Reference Customers
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Schott AG
9.5.2 Gerresheimer AG
9.5.3 Stevanato Group S.p.A (including Ompi)
9.5.4 West Pharmaceutical Services, Inc.
9.5.5 Nipro Corporation
9.5.6 Becton, Dickinson and Company
9.5.7 AptarGroup, Inc.
9.5.8 SGD Pharma
9.5.9 Syntegon Technology GmbH
9.5.10 IMA Group
9.5.11 Romaco Group
9.5.12 Bausch+Ströbel Maschinenfabrik Ilshofen GmbH+Co. KG
9.5.13 Optima packaging group GmbH
9.5.14 Marchesini Group S.p.A.
9.5.15 Dara Pharmaceutical Packaging
10. Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Government Health Departments
10.1.2 Pharmaceutical Regulatory Bodies
10.1.3 Public Health Organizations
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Healthcare Infrastructure
10.2.2 Spending on Medical Equipment
10.2.3 Budget Allocation for Research and Development
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Supply Chain Inefficiencies
10.3.2 Quality Assurance Challenges
10.3.3 Regulatory Compliance Issues
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of New Technologies
10.4.2 Training and Support Needs
10.4.3 Financial Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Cost Savings
10.5.2 Evaluation of Performance Metrics
10.5.3 Identification of New Use Cases
10.5.4 Others
11. Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Solutions
9.2 Export Entry Strategy
9.2.1 Target Countries Identification
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnerships Evaluation
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability Strategies
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup Activities
15.1.2 Market Entry Steps
15.1.3 Growth Acceleration Strategies
15.1.4 Scale & Stabilize Actions
15.2 Key Activities and Milestones
15.2.1 Milestone Identification
15.2.2 Activity Scheduling
Research Methodology
Phase 1: Approach1
Desk Research
- Industry reports from Australian packaging associations and regulatory bodies
- Market analysis publications focusing on glass prefilled syringes and vials
- Statistical data from government health departments regarding pharmaceutical packaging trends
Primary Research
- Interviews with key stakeholders in pharmaceutical companies utilizing glass packaging
- Surveys with packaging equipment manufacturers and suppliers in Australia
- Field interviews with quality assurance managers in healthcare facilities
Validation & Triangulation
- Cross-validation of findings through multiple industry reports and expert opinions
- Triangulation of data from primary interviews and secondary research sources
- Sanity checks conducted through expert panel discussions and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Analysis of total pharmaceutical market size in Australia to estimate glass packaging share
- Segmentation of market by application areas such as vaccines, biologics, and injectables
- Incorporation of trends in healthcare spending and regulatory impacts on packaging
Bottom-up Modeling
- Volume estimates based on production capacities of leading glass vial manufacturers
- Cost analysis derived from pricing models of glass prefilled syringes and vials
- Estimation of market size based on projected growth rates in the pharmaceutical sector
Forecasting & Scenario Analysis
- Multi-variable forecasting using historical growth rates and emerging market trends
- Scenario analysis based on potential regulatory changes and technological advancements
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 100 | Production Managers, Quality Control Officers |
| Healthcare Providers | 80 | Pharmacists, Hospital Administrators |
| Packaging Equipment Suppliers | 60 | Sales Directors, Technical Support Managers |
| Regulatory Bodies | 40 | Compliance Officers, Policy Makers |
| Research Institutions | 40 | Research Scientists, Market Analysts |
Frequently Asked Questions
What is the current value of the Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market?
The Australia Glass Prefilled Syringes Glass Vials Packaging Equipment Market is valued at approximately USD 1.0 billion, reflecting a comprehensive analysis of the glass syringes market and broader pharmaceutical packaging expenditures in the country.