About the Report
Base Year 2024Japan Glass Prefilled Syringes Glass Vials Packaging Equipment Market Overview
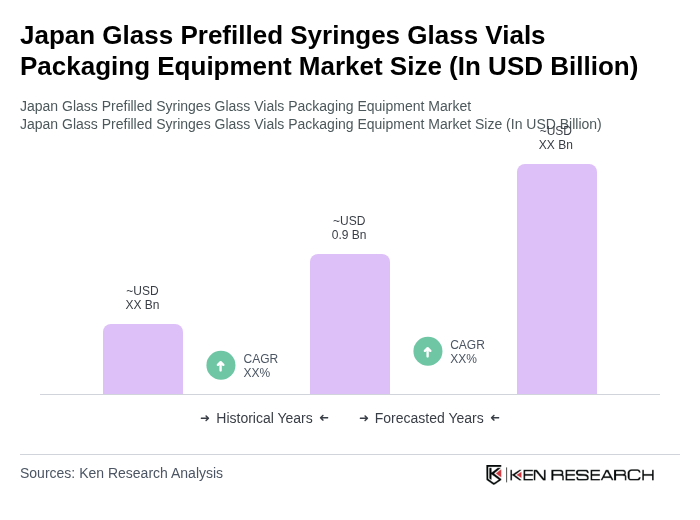
- The Japan Glass Prefilled Syringes Glass Vials Packaging Equipment Market is valued at USD 0.9 billion, based on a five-year historical analysis and triangulation with the broader Japan syringes and prefilled syringes markets. This growth is primarily driven by the increasing demand for biologics and biosimilars, which require advanced container-closure and fill–finish packaging solutions to ensure product integrity and safety. The rise in chronic diseases such as diabetes, rheumatoid arthritis, and oncology indications, along with an ageing population and the expansion of home- and self-administration of injectable therapies, has further propelled the market.
- Key regions dominating this market include Kanto, Kansai, and Chubu, in line with the regional distribution of pharmaceutical manufacturing and healthcare demand in Japan. Kanto, home to Tokyo, is a major hub for pharmaceutical companies, CDMOs, and research institutions, fostering innovation and demand for advanced glass container and packaging equipment solutions. Kansai and Chubu also contribute significantly due to their established medical device and pharmaceutical manufacturing bases, proximity to key logistics networks, and presence of major players in Osaka, Kyoto, and Nagoya.
- In 2023, the Japanese government strengthened regulations and quality expectations for pharmaceutical packaging, particularly for injectable products including prefilled syringes and vials, by enforcing the Ministerial Ordinance on Standards for Manufacturing Control and Quality Control for Drugs and Quasi-drugs (MHLW Ordinance No. 179, issued by the Ministry of Health, Labour and Welfare, 2004, as amended) together with the revised GMP-related provisions under the Pharmaceuticals and Medical Devices Act (PMD Act). These instruments require marketing authorization holders and manufacturers to maintain validated aseptic processing, container-closure integrity testing, in-process controls, and documented quality risk management for injectable packaging operations, thereby driving sustained investment in advanced inspection, filling, capping, leak-detection, and serialization equipment.
Japan Glass Prefilled Syringes Glass Vials Packaging Equipment Market Segmentation
By Equipment Type:The equipment type segmentation includes various categories essential for the packaging of glass prefilled syringes and vials. The subsegments are Prefilled syringe packaging equipment, Vial filling equipment, Capping and sealing equipment, Inspection and leak-detection equipment, Labelling and serialization equipment, and Secondary and tertiary packaging equipment. Each of these equipment types supports critical fill–finish and final packaging steps to ensure sterility, container-closure integrity, correct labelling, and full traceability of injectable products, in line with Japanese GMP and global regulatory expectations.
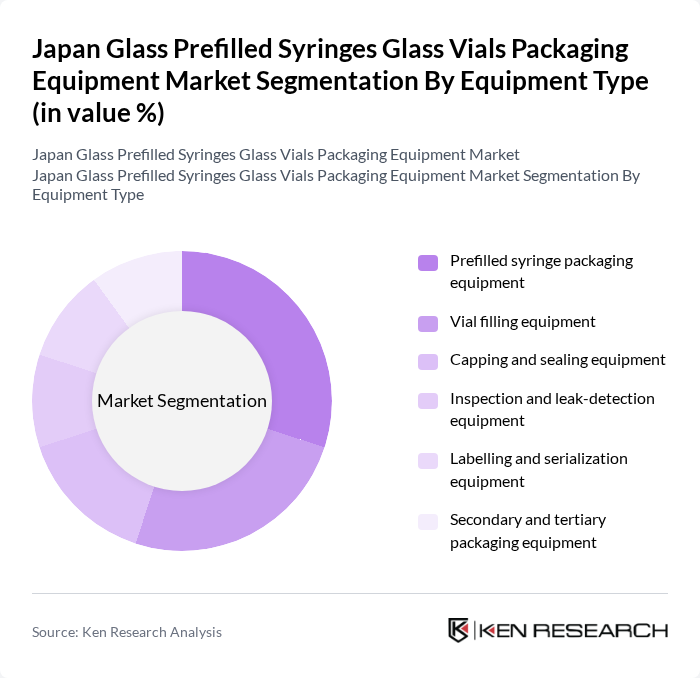
The prefilled syringe packaging equipment segment is currently dominating the market due to the increasing preference for prefilled syringes among healthcare providers and patients, supported by strong growth of the Japan prefilled syringes market. This trend is driven by the convenience of use, reduced risk of contamination, and improved dosing accuracy associated with ready-to-use, pre-measured injectable formats. Additionally, the rise in self-administration of medications and home-based care, particularly in chronic disease management and long-term biologic therapy, has further fueled the demand for high-speed, highly automated prefilled syringe filling, stoppering, and packaging lines. As a result, manufacturers are focusing on enhancing the efficiency, flexibility (e.g., quick changeover between formats), and reliability of prefilled syringe packaging solutions, including integration of inline inspection and data-driven process control.
By Automation Level:The automation level segmentation includes Manual and semi-automatic equipment and Fully automatic equipment. This classification reflects the varying degrees of automation in packaging processes, which can significantly impact production efficiency, throughput, and compliance with aseptic processing requirements.

The fully automatic equipment segment is leading the market due to the growing demand for high-speed production and efficiency in packaging operations, particularly for large-scale biologics and vaccine fill–finish in Japan. Manufacturers are increasingly investing in automation to reduce labor costs, minimize human error, and enhance production capacity while meeting stringent Japanese GMP and international regulatory standards for aseptic processing, in-line inspection, and electronic batch documentation. The trend towards fully automated systems is also driven by the need for advanced data integrity, real-time monitoring, and integration of serialization and aggregation capabilities to support global supply chain and track-and-trace requirements.
Japan Glass Prefilled Syringes Glass Vials Packaging Equipment Market Competitive Landscape
The Japan Glass Prefilled Syringes Glass Vials Packaging Equipment Market is characterized by a dynamic mix of regional and international players. Leading participants such as Gerresheimer AG, Schott Pharma AG & Co. KGaA, West Pharmaceutical Services, Inc., Becton, Dickinson and Company (BD), Nipro Corporation, Stevanato Group S.p.A (Ompi), AptarGroup, Inc. (Aptar Pharma), SMC Ltd., Stelis Biopharma, Vetter Pharma-Fertigung GmbH & Co. KG, Terumo Corporation, Catalent, Inc., Recipharm AB, H&T Presspart, and other notable Japan-focused packaging equipment OEMs contribute to innovation, geographic expansion, and service delivery in this space, including development of advanced glass containers, elastomer components, and integrated fill–finish and inspection solutions for the Japanese market.
Japan Glass Prefilled Syringes Glass Vials Packaging Equipment Market Industry Analysis
Growth Drivers
- Increasing Demand for Biologics:The demand for biologics in Japan is projected to reach ¥1.5 trillion in future, driven by the rise in chronic diseases and an aging population. This surge necessitates advanced packaging solutions, particularly glass prefilled syringes and vials, which ensure drug stability and safety. The Japanese government’s support for biopharmaceutical innovation further fuels this growth, as it aims to enhance healthcare outcomes and reduce the burden of disease on the healthcare system.
- Advancements in Packaging Technology:The Japanese packaging industry is witnessing significant technological advancements, with investments exceeding ¥200 billion in future. Innovations such as smart packaging and enhanced barrier properties are improving the safety and efficacy of biologics. These advancements are crucial as they address the need for tamper-proof and user-friendly packaging, which is essential for maintaining the integrity of sensitive biologics during storage and transportation, thereby driving market growth.
- Rising Healthcare Expenditure:Japan's healthcare expenditure is expected to reach ¥45 trillion in future, reflecting a 5% increase from the previous period. This rise is attributed to the government’s commitment to improving healthcare services and supporting innovative treatments. Increased spending on healthcare directly correlates with the demand for advanced packaging solutions, as pharmaceutical companies seek to ensure the safe delivery of medications, particularly biologics, through reliable packaging methods like glass prefilled syringes and vials.
Market Challenges
- High Manufacturing Costs:The production of glass prefilled syringes and vials involves significant capital investment, with manufacturing costs averaging around ¥30 billion annually for major players. These high costs are primarily due to the stringent quality control measures and advanced technology required to ensure product safety and efficacy. As a result, smaller manufacturers may struggle to compete, limiting market entry and innovation, which poses a challenge to overall market growth.
- Stringent Regulatory Requirements:The Japanese pharmaceutical industry is governed by strict regulations, with compliance costs estimated at ¥10 billion annually for manufacturers. These regulations, enforced by the Pharmaceuticals and Medical Devices Agency (PMDA), ensure product safety and efficacy but can delay product launches and increase operational costs. Navigating these regulatory landscapes poses a significant challenge for companies looking to innovate and expand their product offerings in the glass packaging market.
Japan Glass Prefilled Syringes Glass Vials Packaging Equipment Market Future Outlook
The future of the Japan glass prefilled syringes and glass vials packaging equipment market appears promising, driven by technological advancements and increasing healthcare investments. As the demand for biologics continues to rise, manufacturers are likely to focus on developing innovative packaging solutions that enhance drug delivery and patient safety. Additionally, the integration of automation in manufacturing processes is expected to streamline production, reduce costs, and improve efficiency, positioning the market for sustained growth in the coming years.
Market Opportunities
- Expansion in Emerging Markets:The growing healthcare infrastructure in emerging markets presents significant opportunities for Japanese manufacturers. With an estimated market potential of ¥500 billion in future, companies can leverage their expertise in glass packaging to meet the rising demand for safe and effective drug delivery systems in these regions, enhancing their global footprint.
- Innovations in Sustainable Packaging:The shift towards eco-friendly packaging solutions is gaining momentum, with the sustainable packaging market projected to reach ¥200 billion in future. Japanese manufacturers can capitalize on this trend by developing recyclable and biodegradable glass packaging options, aligning with global sustainability goals and attracting environmentally conscious consumers, thereby enhancing their market competitiveness.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Equipment Type | Prefilled syringe packaging equipment Vial filling equipment Capping and sealing equipment Inspection and leak-detection equipment Labelling and serialization equipment Secondary and tertiary packaging equipment |
| By Automation Level | Manual and semi-automatic equipment Fully automatic equipment |
| By Packaging Operation | Filling and dosing Stoppering and capping Washing and sterilization Visual inspection and quality control Labelling, printing, and serialization Cartoning and case packing |
| By Application | Vaccines Biologics and biosimilars Small-molecule injectables Oncology Diabetes and autoimmune therapies Others |
| By End-User | Pharmaceutical manufacturers Biopharmaceutical companies Contract development and manufacturing organizations (CDMOs/CMOs) Hospital and institutional compounding pharmacies Others |
| By Line Configuration | Standalone equipment Integrated turnkey lines |
| By Region | Kanto Kansai Chubu Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Pharmaceuticals and Medical Devices Agency, Ministry of Health, Labour and Welfare)
Manufacturers and Producers of Glass Prefilled Syringes and Vials
Packaging Equipment Suppliers
Pharmaceutical Companies
Biotechnology Firms
Quality Control and Assurance Organizations
Logistics and Supply Chain Management Companies
Players Mentioned in the Report:
Gerresheimer AG
Schott Pharma AG & Co. KGaA
West Pharmaceutical Services, Inc.
Becton, Dickinson and Company (BD)
Nipro Corporation
Stevanato Group S.p.A (Ompi)
AptarGroup, Inc. (Aptar Pharma)
SMC Ltd.
Stelis Biopharma
Vetter Pharma-Fertigung GmbH & Co. KG
Terumo Corporation
Catalent, Inc.
Recipharm AB
H&T Presspart
Other notable Japan-focused packaging equipment OEMs
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Japan Glass Prefilled Syringes Glass Vials Packaging Equipment Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Japan Glass Prefilled Syringes Glass Vials Packaging Equipment Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Japan Glass Prefilled Syringes Glass Vials Packaging Equipment Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for biologics
3.1.2 Advancements in packaging technology
3.1.3 Rising healthcare expenditure
3.1.4 Growing focus on patient safety
3.2 Market Challenges
3.2.1 High manufacturing costs
3.2.2 Stringent regulatory requirements
3.2.3 Competition from alternative packaging solutions
3.2.4 Supply chain disruptions
3.3 Market Opportunities
3.3.1 Expansion in emerging markets
3.3.2 Innovations in sustainable packaging
3.3.3 Collaborations with pharmaceutical companies
3.3.4 Increasing adoption of automation in manufacturing
3.4 Market Trends
3.4.1 Shift towards eco-friendly materials
3.4.2 Growth of personalized medicine
3.4.3 Integration of smart packaging technologies
3.4.4 Rising demand for prefilled syringes
3.5 Government Regulation
3.5.1 Compliance with GMP standards
3.5.2 Regulations on glass quality and safety
3.5.3 Guidelines for labeling and packaging
3.5.4 Environmental regulations on waste management
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Japan Glass Prefilled Syringes Glass Vials Packaging Equipment Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Japan Glass Prefilled Syringes Glass Vials Packaging Equipment Market Segmentation
8.1 By Equipment Type
8.1.1 Prefilled syringe packaging equipment
8.1.2 Vial filling equipment
8.1.3 Capping and sealing equipment
8.1.4 Inspection and leak-detection equipment
8.1.5 Labelling and serialization equipment
8.1.6 Secondary and tertiary packaging equipment
8.2 By Automation Level
8.2.1 Manual and semi-automatic equipment
8.2.2 Fully automatic equipment
8.3 By Packaging Operation
8.3.1 Filling and dosing
8.3.2 Stoppering and capping
8.3.3 Washing and sterilization
8.3.4 Visual inspection and quality control
8.3.5 Labelling, printing, and serialization
8.3.6 Cartoning and case packing
8.4 By Application
8.4.1 Vaccines
8.4.2 Biologics and biosimilars
8.4.3 Small-molecule injectables
8.4.4 Oncology
8.4.5 Diabetes and autoimmune therapies
8.4.6 Others
8.5 By End-User
8.5.1 Pharmaceutical manufacturers
8.5.2 Biopharmaceutical companies
8.5.3 Contract development and manufacturing organizations (CDMOs/CMOs)
8.5.4 Hospital and institutional compounding pharmacies
8.5.5 Others
8.6 By Line Configuration
8.6.1 Standalone equipment
8.6.2 Integrated turnkey lines
8.7 By Region
8.7.1 Kanto
8.7.2 Kansai
8.7.3 Chubu
8.7.4 Others
9. Japan Glass Prefilled Syringes Glass Vials Packaging Equipment Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company name
9.2.2 Group size (large, medium, or small as per industry convention)
9.2.3 Japan glass prefilled syringes & glass vials packaging equipment revenue (latest year)
9.2.4 3-year revenue CAGR in Japan packaging equipment
9.2.5 Installed base of lines in Japan (number of lines / key sites)
9.2.6 Market share in Japan (value and volume, %)
9.2.7 Order backlog / book-to-bill ratio
9.2.8 Average selling price per line / per module
9.2.9 R&D intensity (% of revenue)
9.2.10 New product launches and upgrades (last 3 years)
9.2.11 OEE (overall equipment effectiveness) benchmark on flagship lines
9.2.12 Service and maintenance revenue share (% of total)
9.2.13 Key Japanese pharma/biopharma accounts covered
9.2.14 Geographic footprint within Japan (plants, sales & service hubs)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Gerresheimer AG
9.5.2 Schott Pharma AG & Co. KGaA
9.5.3 West Pharmaceutical Services, Inc.
9.5.4 Becton, Dickinson and Company (BD)
9.5.5 Nipro Corporation
9.5.6 Stevanato Group S.p.A (Ompi)
9.5.7 AptarGroup, Inc. (Aptar Pharma)
9.5.8 SMC Ltd.
9.5.9 Stelis Biopharma
9.5.10 Vetter Pharma-Fertigung GmbH & Co. KG
9.5.11 Terumo Corporation
9.5.12 Catalent, Inc.
9.5.13 Recipharm AB
9.5.14 H&T Presspart
9.5.15 Other notable Japan-focused packaging equipment OEMs
10. Japan Glass Prefilled Syringes Glass Vials Packaging Equipment Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget allocation trends
10.1.2 Decision-making processes
10.1.3 Supplier selection criteria
10.1.4 Contract management practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment priorities
10.2.2 Spending patterns
10.2.3 Cost-saving initiatives
10.2.4 Future spending forecasts
10.3 Pain Point Analysis by End-User Category
10.3.1 Quality assurance challenges
10.3.2 Supply chain inefficiencies
10.3.3 Regulatory compliance issues
10.3.4 Cost management difficulties
10.4 User Readiness for Adoption
10.4.1 Training and support needs
10.4.2 Technology adoption barriers
10.4.3 Change management strategies
10.4.4 Feedback mechanisms
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance measurement metrics
10.5.2 Case studies of successful implementations
10.5.3 Scalability considerations
10.5.4 Future use case opportunities
11. Japan Glass Prefilled Syringes Glass Vials Packaging Equipment Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Customer segmentation analysis
1.5 Competitive landscape overview
1.6 Key partnerships identification
1.7 Risk assessment
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience definition
2.4 Communication strategies
2.5 Digital marketing initiatives
2.6 Trade show participation
2.7 Customer engagement tactics
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 E-commerce integration
3.4 Logistics optimization
3.5 Channel partner selection
3.6 Inventory management
3.7 Distribution network expansion
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands analysis
4.3 Competitor pricing strategies
4.4 Customer willingness to pay
4.5 Value-based pricing models
4.6 Discounting strategies
4.7 Price elasticity considerations
5. Unmet Demand & Latent Needs
5.1 Category gaps identification
5.2 Consumer segments analysis
5.3 Product development opportunities
5.4 Market entry strategies
5.5 Customer feedback incorporation
5.6 Trend analysis
5.7 Future demand forecasting
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
6.3 Customer feedback loops
6.4 Relationship management strategies
6.5 Community engagement initiatives
6.6 Customer education programs
6.7 Retention strategies
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Cost-benefit analysis
7.4 Unique selling propositions
7.5 Customer-centric approaches
7.6 Innovation in product offerings
7.7 Market differentiation strategies
8. Key Activities
8.1 Regulatory compliance
8.2 Branding initiatives
8.3 Distribution setup
8.4 Marketing campaigns
8.5 Training and development
8.6 Performance monitoring
8.7 Stakeholder engagement
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band analysis
9.1.3 Packaging strategies
9.2 Export Entry Strategy
9.2.1 Target countries identification
9.2.2 Compliance roadmap development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
10.5 Risk assessment
10.6 Market entry barriers
10.7 Strategic fit evaluation
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines for market entry
11.3 Budget allocation
11.4 Financial projections
11.5 Funding sources
11.6 Cost management strategies
11.7 Milestone tracking
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnership dynamics
12.3 Risk management strategies
12.4 Control mechanisms
12.5 Exit strategies
12.6 Long-term sustainability
12.7 Performance evaluation
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
13.3 Profit margin analysis
13.4 Revenue growth projections
13.5 Cost structure evaluation
13.6 Financial health assessment
13.7 Investment return analysis
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition targets
14.4 Strategic alliances
14.5 Supplier partnerships
14.6 Research collaborations
14.7 Community engagement partners
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity tracking
15.2.2 Milestone achievement
15.2.3 Performance reviews
15.2.4 Stakeholder updates
Research Methodology
Phase 1: Approach1
Desk Research
- Industry reports from Japanese pharmaceutical and packaging associations
- Market analysis publications focusing on glass prefilled syringes and vials
- Regulatory guidelines and updates from Japan's Ministry of Health, Labour and Welfare
Primary Research
- Interviews with R&D heads at leading pharmaceutical companies
- Surveys with packaging engineers specializing in glass solutions
- Field interviews with quality assurance managers in manufacturing plants
Validation & Triangulation
- Cross-validation of data from multiple industry sources and expert opinions
- Triangulation of market size estimates using sales data and production statistics
- Sanity checks through feedback from industry panels and focus groups
Phase 2: Market Size Estimation1
Top-down Assessment
- Analysis of total healthcare expenditure in Japan and its allocation to packaging
- Segmentation of the market by application areas such as vaccines and biologics
- Incorporation of trends in patient-centric drug delivery systems
Bottom-up Modeling
- Volume estimates based on production capacities of major glass vial manufacturers
- Cost analysis derived from supplier pricing and material costs
- Estimation of market share based on sales data from key players
Forecasting & Scenario Analysis
- Multi-variable forecasting using growth rates in the biopharmaceutical sector
- Scenario analysis based on potential regulatory changes and market disruptions
- Baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturing | 120 | Production Managers, Quality Control Supervisors |
| Biotechnology Firms | 90 | Research Scientists, Regulatory Affairs Specialists |
| Healthcare Providers | 80 | Pharmacists, Hospital Procurement Officers |
| Packaging Equipment Suppliers | 60 | Sales Managers, Technical Support Engineers |
| Regulatory Bodies | 50 | Compliance Officers, Policy Analysts |
Frequently Asked Questions
What is the current market value of the Japan Glass Prefilled Syringes Glass Vials Packaging Equipment Market?
The Japan Glass Prefilled Syringes Glass Vials Packaging Equipment Market is valued at approximately USD 0.9 billion, reflecting a significant growth driven by the increasing demand for biologics and biosimilars, as well as advancements in packaging technology.