Region:Middle East
Author(s):Dev
Product Code:KRAA3913
Pages:85
Published On:January 2026
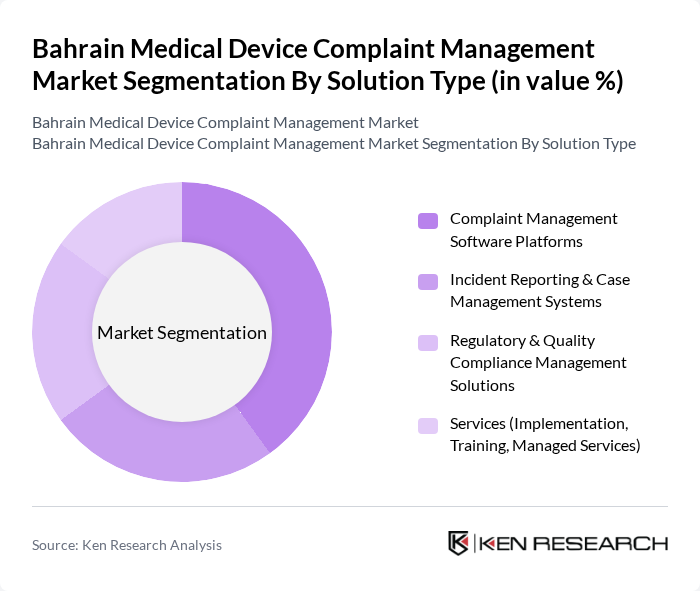
By Solution Type:The solution type segmentation includes various subsegments such as Complaint Management Software Platforms, Incident Reporting & Case Management Systems, Regulatory & Quality Compliance Management Solutions, and Services (Implementation, Training, Managed Services). Among these, Complaint Management Software Platforms are leading the market due to their ability to centralize device?related feedback, automate workflows, support audit trails, and integrate with quality management and regulatory reporting systems, which is increasingly important as providers adopt more connected and data?driven medical technologies. The increasing adoption of digital solutions in healthcare, including electronic medical records, telemedicine, and remote monitoring, is driving the need for interoperable complaint management tools that can capture real?time incident data and support faster corrective and preventive actions.
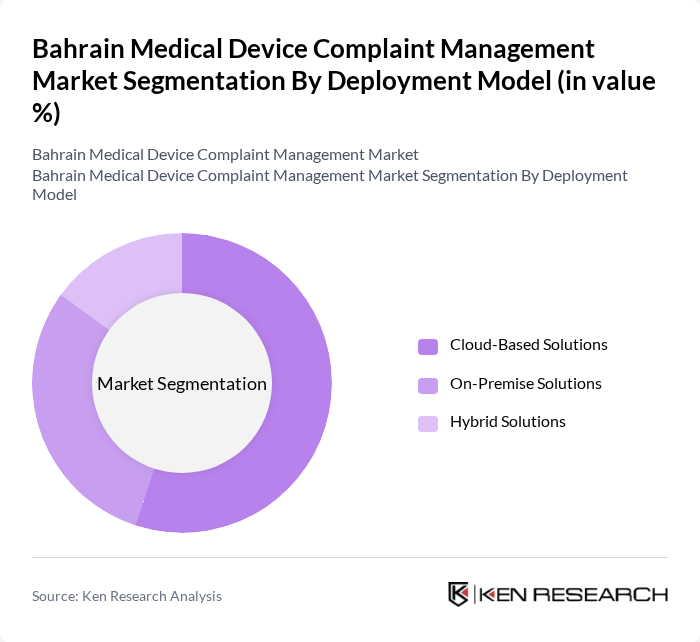
By Deployment Model:The deployment model segmentation includes Cloud-Based Solutions, On-Premise Solutions, and Hybrid Solutions. Cloud-Based Solutions are dominating this segment due to their scalability, subscription?based cost models, ease of remote access, and ability to support multi?site hospital networks and distributed device inventories, which aligns with broader cloud adoption trends in healthcare IT across the region. The growing trend of telehealth, remote device monitoring, and the need for real?time data access for safety signal detection and regulatory reporting are further enhancing the adoption of cloud-based complaint management systems, while on?premise and hybrid models remain relevant for institutions with specific data residency or integration requirements.
The Bahrain Medical Device Complaint Management Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Johnson & Johnson MedTech, Siemens Healthineers AG, GE HealthCare Technologies Inc., Philips Healthcare (Royal Philips), Abbott Laboratories, Boston Scientific Corporation, Stryker Corporation, Becton, Dickinson and Company (BD), Zimmer Biomet Holdings, Inc., 3M Health Care (3M Company), Olympus Corporation, Baxter International Inc., Hologic, Inc., Terumo Corporation contribute to innovation, geographic expansion, and service delivery in this space, leveraging their global post?market surveillance, vigilance, and quality systems to support complaint handling for devices deployed in Bahraini healthcare facilities.
The future of the Bahrain medical device complaint management market is poised for transformation, driven by technological advancements and regulatory changes. As healthcare providers increasingly adopt digital solutions, the integration of artificial intelligence and data analytics will enhance complaint resolution processes. Furthermore, collaboration with regulatory bodies is expected to streamline compliance efforts, ensuring that patient safety remains at the forefront. These developments will create a more responsive and efficient complaint management landscape, ultimately benefiting both healthcare providers and patients.
| Segment | Sub-Segments |
|---|---|
| By Solution Type | Complaint Management Software Platforms Incident Reporting & Case Management Systems Regulatory & Quality Compliance Management Solutions Services (Implementation, Training, Managed Services) |
| By Deployment Model | Cloud-Based Solutions On-Premise Solutions Hybrid Solutions |
| By End-User | Medical Device Manufacturers Hospitals & Health Systems Clinics & Diagnostic Centers Distributors & Importers Regulatory Bodies & Notified Entities |
| By Complaint Type | Product Quality & Performance Issues Adverse Events & Safety Incidents Usability & Human Factors Issues Service & Support-Related Complaints Others |
| By Device Category | Diagnostic Devices & Imaging Systems Therapeutic & Surgical Devices Patient Monitoring Devices Single-Use & Consumable Devices Others |
| By Organization Size | Large Enterprises Small & Medium Enterprises (SMEs) |
| By Workflow | Intake & Triage Investigation & Root Cause Analysis Corrective & Preventive Actions (CAPA) Regulatory Reporting & Documentation |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Complaint Management | 60 | Quality Assurance Managers, Clinical Risk Managers |
| Device Manufacturer Feedback | 50 | Regulatory Affairs Specialists, Product Managers |
| Patient Experience Surveys | 120 | Patients, Caregivers |
| Healthcare Provider Insights | 70 | Doctors, Nurses, Medical Technologists |
| Regulatory Compliance Interviews | 40 | Compliance Officers, Legal Advisors |
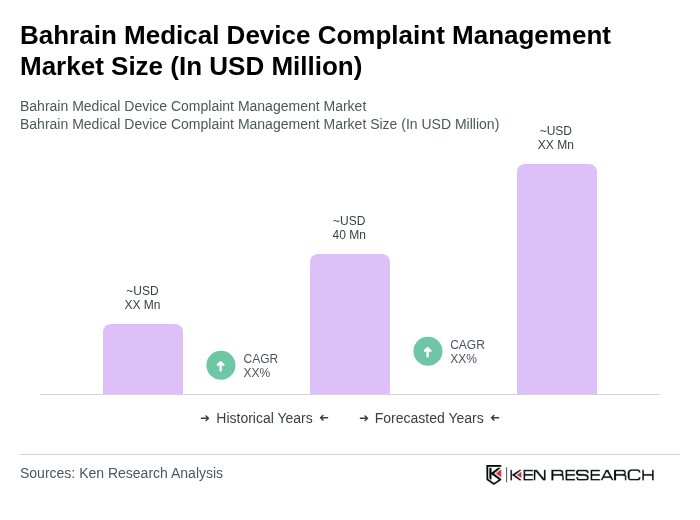
The Bahrain Medical Device Complaint Management Market is valued at approximately USD 40 million, reflecting the growth and demand for effective complaint management systems within the broader medical devices market, which is around USD 400 million.