Region:Middle East
Author(s):Shubham
Product Code:KRAD0826
Pages:97
Published On:December 2025
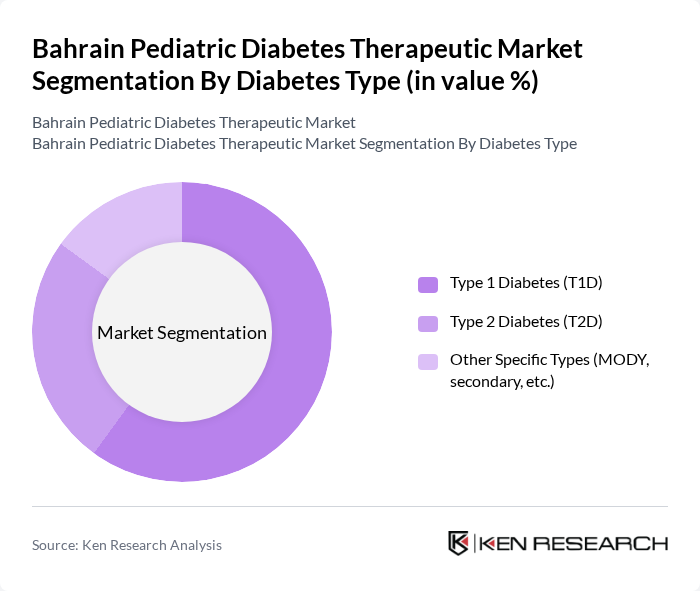
By Diabetes Type:The pediatric diabetes therapeutic market is segmented into Type 1 Diabetes (T1D), Type 2 Diabetes (T2D), and Other Specific Types (such as MODY and secondary diabetes). Type 1 Diabetes is the most prevalent among children, driving the demand for insulin therapies and continuous glucose monitoring solutions. The increasing awareness and diagnosis of diabetes in children have led to a growing market for all types, but T1D remains the dominant segment due to its higher incidence rates.
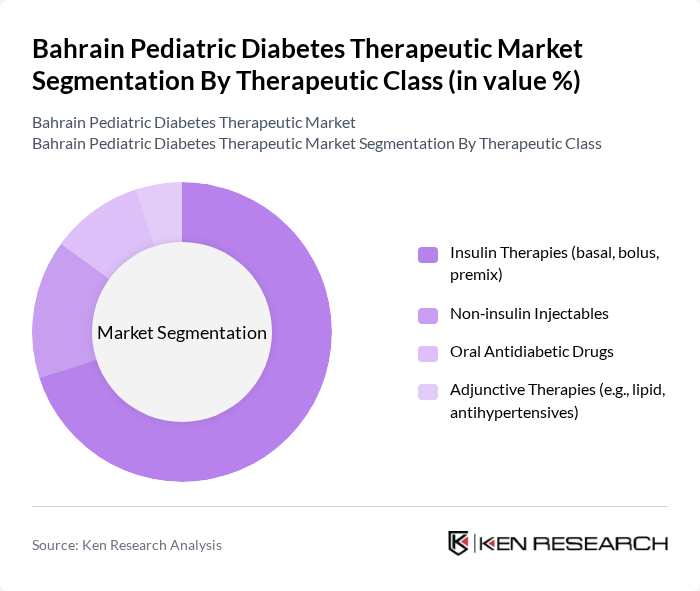
By Therapeutic Class:The market is further segmented into Insulin Therapies (including basal, bolus, and premix), Non-insulin Injectables, Oral Antidiabetic Drugs, and Adjunctive Therapies (such as lipid and antihypertensives). Insulin Therapies dominate the market due to the essential role of insulin in managing Type 1 Diabetes, which is prevalent in the pediatric population. The increasing adoption of advanced insulin delivery systems and the introduction of new formulations are also contributing to the growth of this segment.
The Bahrain Pediatric Diabetes Therapeutic Market is characterized by a dynamic mix of regional and international players. Leading participants such as Novo Nordisk A/S, Sanofi S.A., Eli Lilly and Company, F. Hoffmann-La Roche Ltd (Roche Diabetes Care), Medtronic plc, Abbott Laboratories, Bayer AG (Bayer HealthCare), Dexcom, Inc., Insulet Corporation, Ascensia Diabetes Care Holdings AG, Ypsomed AG, Boehringer Ingelheim International GmbH, Merck & Co., Inc., Johnson & Johnson (including LifeScan/Animas legacy), ACON Laboratories, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Bahrain Pediatric Diabetes Therapeutic Market appears promising, driven by advancements in technology and government initiatives. The introduction of AI-enabled early detection systems and standardized clinical pathways will enhance diabetes management. Additionally, the National Early Screening Programme for Non-Communicable Diseases aims to engage over 150,000 residents, improving early detection capabilities for pediatric diabetes. These developments indicate a proactive approach to managing pediatric diabetes in Bahrain, fostering a supportive environment for innovative solutions.
| Segment | Sub-Segments |
|---|---|
| By Diabetes Type | Type 1 Diabetes (T1D) Type 2 Diabetes (T2D) Other Specific Types (MODY, secondary, etc.) |
| By Therapeutic Class | Insulin Therapies (basal, bolus, premix) Non?insulin Injectables Oral Antidiabetic Drugs Adjunctive Therapies (e.g., lipid, antihypertensives) |
| By Delivery / Device Type | Insulin Pumps Insulin Pens and Syringes Continuous Glucose Monitoring (CGM) Self-Monitoring Blood Glucose (SMBG) Devices Connected Digital Therapeutics & Apps |
| By Age Group | –5 Years –12 Years –18 Years |
| By End-User | Public Hospitals Private Hospitals Pediatric & Endocrinology Clinics Homecare Settings |
| By Distribution Channel | Hospital Pharmacies Retail / Community Pharmacies Online Pharmacies & E-commerce |
| By Payer Type | Government & Public Insurance Private Insurance Out-of-Pocket / Self-Pay |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pediatric Endocrinology Clinics | 45 | Pediatric Endocrinologists, Diabetes Educators |
| Diabetes Support Groups | 80 | Parents of Children with Diabetes, Caregivers |
| Healthcare Policy Makers | 50 | Health Ministry Officials, Public Health Experts |
| Pharmaceutical Representatives | 40 | Sales Managers, Product Specialists |
| Diabetes Management Programs | 60 | Program Coordinators, Healthcare Administrators |
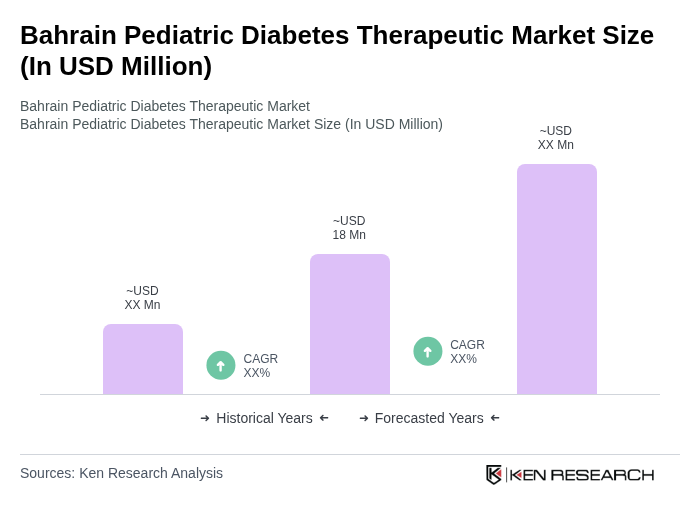
The Bahrain Pediatric Diabetes Therapeutic Market is valued at approximately USD 18 million, driven by the rising prevalence of pediatric diabetes, increased healthcare spending, and the adoption of advanced management technologies such as insulin pumps and continuous glucose monitoring systems.