Region:Middle East
Author(s):Geetanshi
Product Code:KRAA0489
Pages:80
Published On:December 2025
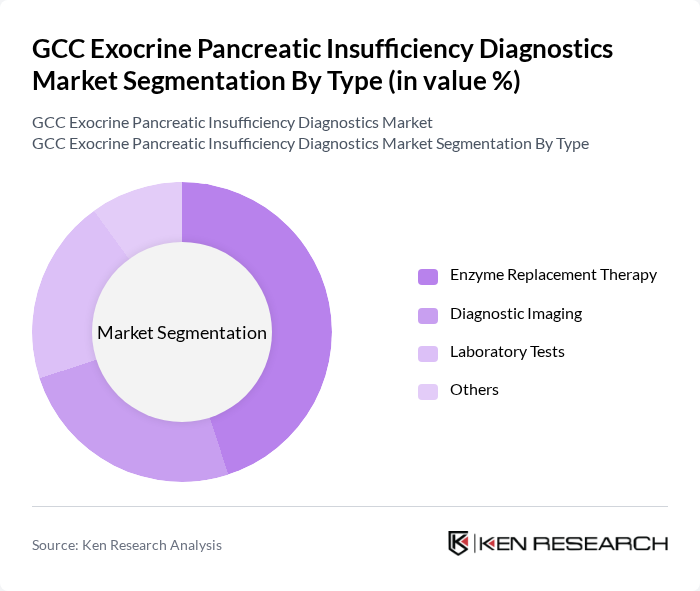
By Type:The diagnostics market is segmented into various types, including Enzyme Replacement Therapy, Diagnostic Imaging, Laboratory Tests, and Others. Among these, Enzyme Replacement Therapy is the leading sub-segment due to its critical role in managing exocrine pancreatic insufficiency, particularly in patients with chronic pancreatitis and cystic fibrosis. The increasing awareness of the importance of timely diagnosis and treatment options has further propelled the demand for this segment.
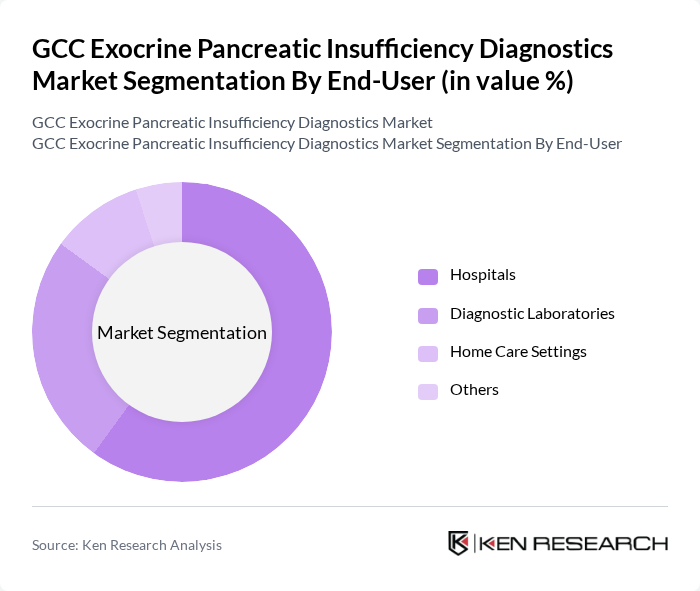
By End-User:The market is also segmented by end-users, including Hospitals, Diagnostic Laboratories, Home Care Settings, and Others. Hospitals are the dominant end-user segment, primarily due to their comprehensive facilities and resources for diagnosing and treating exocrine pancreatic insufficiency. The increasing number of hospital admissions for related conditions and the availability of advanced diagnostic tools in these settings contribute significantly to this segment's growth.
The GCC Exocrine Pancreatic Insufficiency Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as ChiRhoClin, Certest Biotec, ScheBo Biotech AG, ALPCO Diagnostics, Immundiagnostik AG, and Labcorp contribute to innovation, geographic expansion, and service delivery in this space.
The future of the GCC exocrine pancreatic insufficiency diagnostics market appears promising, driven by advancements in digital health technologies and a growing emphasis on personalized medicine. The integration of AI and telehealth solutions is expected to enhance diagnostic accuracy and accessibility, particularly in remote areas. Additionally, the expansion of healthcare infrastructure, including the projected need for 12,317 new hospital beds in future, will facilitate the deployment of advanced diagnostic services, including those for EPI, thereby improving patient care and outcomes.
| Segment | Sub-Segments |
|---|---|
| By Type | Enzyme Replacement Therapy Diagnostic Imaging Laboratory Tests Others |
| By End-User | Hospitals Diagnostic Laboratories Home Care Settings Others |
| By Age Group | Pediatric Adult Geriatric Others |
| By Gender | Male Female Others |
| By Diagnostic Method | Non-Invasive Tests Invasive Tests Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Others |
| By Region | Saudi Arabia UAE Qatar Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gastroenterology Clinics | 100 | Gastroenterologists, Clinic Managers |
| Diagnostic Laboratories | 80 | Laboratory Technicians, Quality Control Managers |
| Patient Advocacy Groups | 60 | Patient Representatives, Healthcare Advocates |
| Healthcare Policy Makers | 50 | Health Ministry Officials, Policy Analysts |
| Pharmaceutical Companies | 70 | Product Managers, Market Access Specialists |
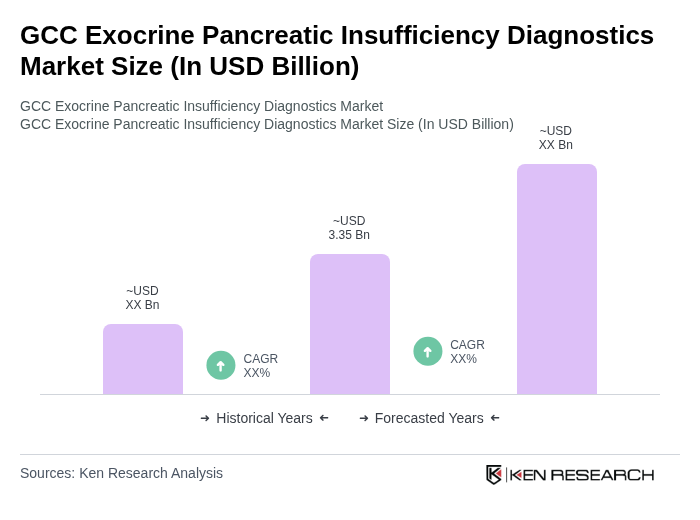
The GCC Exocrine Pancreatic Insufficiency Diagnostics Market is valued at approximately USD 3.35 billion, reflecting a significant growth driven by the increasing prevalence of chronic pancreatitis, cystic fibrosis, and diabetes among patient populations in the region.