About the Report
Base Year 2024Global Biobetters Market Overview
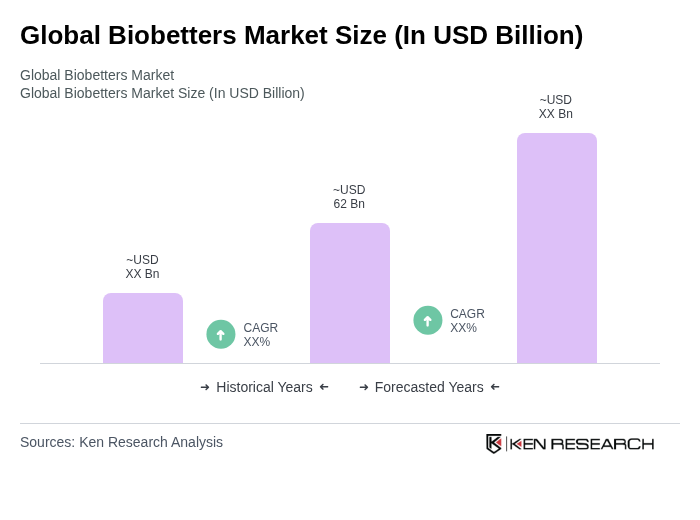
- The Global Biobetters Market is valued at USD 62 billion, based on recent analysis. Growth is driven by accelerating patent expirations of blockbuster biologics, increasing prevalence of chronic diseases such as diabetes and oncology, and technological advances in site-specific protein engineering that enhance efficacy, safety, and convenience. Advances in manufacturing and payer willingness to support differentiated biologics further bolster expansion.
- Key players in this market include Amgen, Novo Nordisk, Roche, Biogen, Merck & Co., and Sanofi, which drive innovation and commercialization through strategic alliances and proprietary engineering platforms. Market leadership is concentrated in North America, where advanced regulatory frameworks, payer support for clinically superior therapies, high R&D investment, and robust biotech infrastructure sustain dominance; Asia-Pacific is emerging rapidly due to regulatory reforms, rising healthcare spending, and expanding biomanufacturing capacity.
- In 2024, the U.S. government announced a joint regulatory plan under Executive Order 14081, which streamlines oversight of biotechnology products—including human biologics—by coordinating regulatory clarity, transparency, and efficiency across agencies.
Global Biobetters Market Segmentation
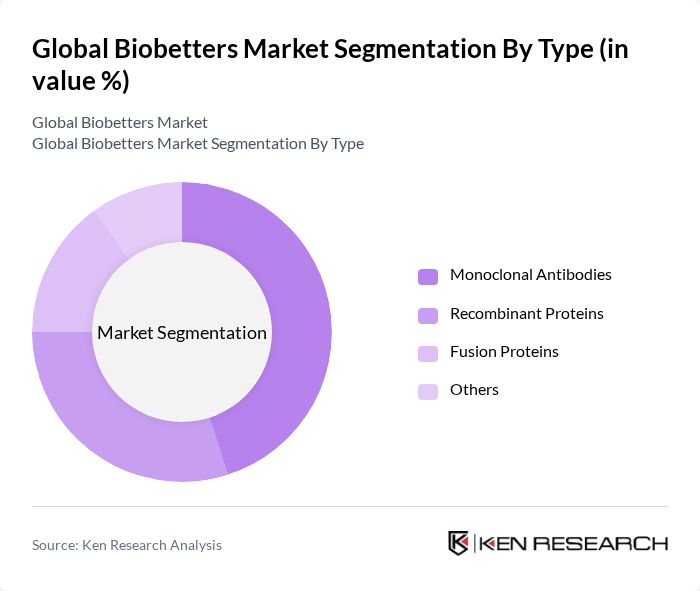
By Type:The biobetters market is segmented into four main types: Monoclonal Antibodies, Recombinant Proteins, Fusion Proteins, and Others. Monoclonal antibodies dominate the market due to their targeted therapeutic effects and increasing applications in oncology and autoimmune diseases. Recombinant proteins also hold a significant share, driven by advancements in genetic engineering and their use in various therapeutic areas. Fusion proteins are gaining traction due to their dual functionality, while the 'Others' category includes emerging biobetter products that are still in development.
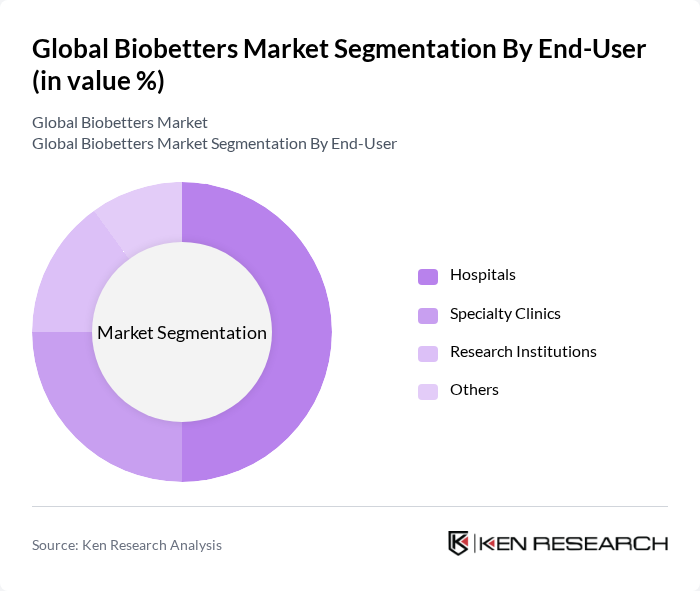
By End-User:The end-user segmentation includes Hospitals, Specialty Clinics, Research Institutions, and Others. Hospitals are the leading end-users, primarily due to their extensive patient base and the need for advanced therapeutic options. Specialty clinics are also significant, focusing on specific diseases and providing tailored treatments. Research institutions contribute to the market by developing new biobetter products, while the 'Others' category encompasses various healthcare settings that utilize biobetters.
Global Biobetters Market Competitive Landscape
The Global Biobetters Market is characterized by a dynamic mix of regional and international players. Leading participants such as Amgen Inc., AbbVie Inc., Roche Holding AG, Pfizer Inc., Johnson & Johnson, Merck & Co., Inc., Sanofi S.A., Novartis AG, GSK (GlaxoSmithKline) plc, Eli Lilly and Company, Biogen Inc., Regeneron Pharmaceuticals, Inc., Takeda Pharmaceutical Company Limited, Astellas Pharma Inc., Bayer AG contribute to innovation, geographic expansion, and service delivery in this space.
Global Biobetters Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The global burden of chronic diseases is escalating, with the World Health Organization reporting that non-communicable diseases accounted for 74% of all deaths in 2020. In future, it is projected that over 1.6 billion people will be living with chronic conditions, driving the demand for innovative treatments. This surge necessitates advanced therapeutic options, such as biobetters, which offer improved efficacy and safety profiles compared to existing therapies, thus propelling market growth.
- Advancements in Biotechnology:The biotechnology sector is experiencing rapid advancements, with global investments reaching approximately $350 billion in future. Innovations in genetic engineering, monoclonal antibodies, and protein engineering are enhancing the development of biobetters. These technologies enable the creation of more effective and targeted therapies, addressing unmet medical needs. As a result, the biobetters market is poised for significant growth, driven by the increasing availability of cutting-edge biotechnological solutions.
- Rising Demand for Personalized Medicine:The shift towards personalized medicine is reshaping the healthcare landscape, with the global personalized medicine market projected to reach $2.5 trillion in future. Patients increasingly seek tailored therapies that cater to their unique genetic profiles and health conditions. Biobetters, which can be customized to enhance therapeutic outcomes, are well-positioned to meet this demand. This trend is expected to significantly boost the adoption of biobetters in various therapeutic areas, including oncology and autoimmune diseases.
Market Challenges
- High Development Costs:The development of biobetters is associated with substantial financial investments, often exceeding $1.2 billion per product. This high cost is primarily due to extensive research and development phases, including preclinical and clinical trials. As a result, many companies face financial constraints, limiting their ability to innovate and bring new biobetter products to market. This challenge can hinder overall market growth and restrict the entry of new players into the biobetters sector.
- Stringent Regulatory Requirements:The biopharmaceutical industry is subject to rigorous regulatory scrutiny, with agencies like the FDA and EMA enforcing strict guidelines for biobetter approvals. In future, the average time for regulatory approval is estimated to be around 9 years, significantly delaying product launches. These stringent requirements can deter investment and slow down the pace of innovation in the biobetters market, posing a significant challenge for companies aiming to introduce new therapies.
Global Biobetters Market Future Outlook
The future of the biobetters market appears promising, driven by ongoing advancements in biotechnology and a growing emphasis on personalized medicine. As healthcare systems increasingly adopt patient-centric approaches, biobetters are likely to gain traction due to their enhanced efficacy and safety profiles. Furthermore, the integration of digital health technologies is expected to facilitate better patient monitoring and treatment adherence, further propelling market growth. Companies that leverage these trends will be well-positioned to capitalize on emerging opportunities in the biobetters landscape.
Market Opportunities
- Growing Investment in R&D:With global R&D spending projected to exceed $2.5 trillion in future, there is a significant opportunity for biobetter development. Increased funding will enable companies to explore innovative therapeutic solutions, enhancing their product pipelines and market competitiveness. This investment is crucial for advancing biobetter technologies and addressing unmet medical needs in various therapeutic areas.
- Expansion into Emerging Markets:Emerging markets, particularly in Asia and Africa, present substantial growth opportunities for biobetters. With rising healthcare expenditures projected to reach $1.8 trillion in these regions by future, companies can tap into new patient populations. This expansion can drive demand for advanced therapies, including biobetters, as healthcare systems strive to improve treatment outcomes and access to innovative medicines.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Monoclonal Antibodies Recombinant Proteins Fusion Proteins Others |
| By End-User | Hospitals Specialty Clinics Research Institutions Others |
| By Route of Administration | Subcutaneous Intravenous Intramuscular Others |
| By Therapeutic Area | Oncology Autoimmune Diseases Infectious Diseases Others |
| By Distribution Channel | Direct Sales Distributors Online Pharmacies Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Market Segment | Established Markets Emerging Markets Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, European Medicines Agency)
Biopharmaceutical Manufacturers
Contract Research Organizations (CROs)
Healthcare Providers and Hospitals
Pharmaceutical Supply Chain Companies
Health Insurance Companies
Biotechnology Industry Associations
Players Mentioned in the Report:
Amgen Inc.
AbbVie Inc.
Roche Holding AG
Pfizer Inc.
Johnson & Johnson
Merck & Co., Inc.
Sanofi S.A.
Novartis AG
GSK (GlaxoSmithKline) plc
Eli Lilly and Company
Biogen Inc.
Regeneron Pharmaceuticals, Inc.
Takeda Pharmaceutical Company Limited
Astellas Pharma Inc.
Bayer AG
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Biobetters Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Biobetters Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Biobetters Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of chronic diseases
3.1.2 Advancements in biotechnology
3.1.3 Rising demand for personalized medicine
3.1.4 Expanding healthcare expenditure
3.2 Market Challenges
3.2.1 High development costs
3.2.2 Stringent regulatory requirements
3.2.3 Market competition from biosimilars
3.2.4 Limited awareness among healthcare professionals
3.3 Market Opportunities
3.3.1 Growing investment in R&D
3.3.2 Expansion into emerging markets
3.3.3 Collaborations and partnerships
3.3.4 Technological innovations in drug delivery
3.4 Market Trends
3.4.1 Increasing focus on patient-centric approaches
3.4.2 Rise of digital health technologies
3.4.3 Shift towards value-based healthcare
3.4.4 Growing emphasis on sustainability in production
3.5 Government Regulation
3.5.1 Enhanced approval processes for biobetters
3.5.2 Guidelines for clinical trials
3.5.3 Pricing regulations for biopharmaceuticals
3.5.4 Policies promoting innovation in biotechnology
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Biobetters Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Biobetters Market Segmentation
8.1 By Type
8.1.1 Monoclonal Antibodies
8.1.2 Recombinant Proteins
8.1.3 Fusion Proteins
8.1.4 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Specialty Clinics
8.2.3 Research Institutions
8.2.4 Others
8.3 By Route of Administration
8.3.1 Subcutaneous
8.3.2 Intravenous
8.3.3 Intramuscular
8.3.4 Others
8.4 By Therapeutic Area
8.4.1 Oncology
8.4.2 Autoimmune Diseases
8.4.3 Infectious Diseases
8.4.4 Others
8.5 By Distribution Channel
8.5.1 Direct Sales
8.5.2 Distributors
8.5.3 Online Pharmacies
8.5.4 Others
8.6 By Region
8.6.1 North America
8.6.2 Europe
8.6.3 Asia-Pacific
8.6.4 Latin America
8.6.5 Middle East & Africa
8.7 By Market Segment
8.7.1 Established Markets
8.7.2 Emerging Markets
8.7.3 Others
9. Global Biobetters Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Product Development Cycle Time
9.2.8 Sales Conversion Rate
9.2.9 Distribution Efficiency
9.2.10 Brand Equity
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Amgen Inc.
9.5.2 AbbVie Inc.
9.5.3 Roche Holding AG
9.5.4 Pfizer Inc.
9.5.5 Johnson & Johnson
9.5.6 Merck & Co., Inc.
9.5.7 Sanofi S.A.
9.5.8 Novartis AG
9.5.9 GSK (GlaxoSmithKline) plc
9.5.10 Eli Lilly and Company
9.5.11 Biogen Inc.
9.5.12 Regeneron Pharmaceuticals, Inc.
9.5.13 Takeda Pharmaceutical Company Limited
9.5.14 Astellas Pharma Inc.
9.5.15 Bayer AG
10. Global Biobetters Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Supplier Selection Criteria
10.1.4 Contracting Practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Priorities
10.2.2 Spending Patterns
10.2.3 Cost Management Strategies
10.2.4 Future Spending Projections
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges Faced by Hospitals
10.3.2 Issues in Specialty Clinics
10.3.3 Concerns of Research Institutions
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training Needs
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of ROI
10.5.2 Use Case Success Stories
10.5.3 Scalability Potential
10.5.4 Others
11. Global Biobetters Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-Ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-Sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-Term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from global health organizations and market research firms
- Review of scientific publications and clinical trial data related to biobetters
- Examination of regulatory guidelines and approval processes from health authorities
Primary Research
- Interviews with biopharmaceutical executives and R&D leaders
- Surveys targeting healthcare professionals and specialists in biobetters
- Focus groups with patients and caregivers to understand treatment experiences
Validation & Triangulation
- Cross-validation of findings through multiple data sources including market reports and expert opinions
- Triangulation of quantitative data with qualitative insights from interviews
- Sanity checks conducted through expert panel discussions and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on global healthcare expenditure and biopharmaceutical growth rates
- Segmentation by therapeutic areas and geographical regions
- Incorporation of trends in patient demographics and disease prevalence
Bottom-up Modeling
- Collection of sales data from leading biopharmaceutical companies
- Estimation of market share based on product launches and pipeline analysis
- Volume and pricing analysis for existing biobetter products
Forecasting & Scenario Analysis
- Multi-variable forecasting models incorporating market drivers such as innovation and regulatory changes
- Scenario analysis based on potential market disruptions and emerging technologies
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Biobetters | 100 | Oncologists, Clinical Researchers |
| Autoimmune Disease Treatments | 80 | Rheumatologists, Immunologists |
| Diabetes Management Solutions | 90 | Endocrinologists, Diabetes Educators |
| Cardiovascular Biobetters | 70 | Cardiologists, Pharmacists |
| Patient Experience Insights | 60 | Patients, Caregivers, Patient Advocacy Groups |
Frequently Asked Questions
What is the current value of the Global Biobetters Market?
The Global Biobetters Market is currently valued at approximately USD 62 billion. This valuation reflects the market's growth driven by factors such as patent expirations of blockbuster biologics and the increasing prevalence of chronic diseases.