Region:Global
Author(s):Shubham
Product Code:KRAC3652
Pages:100
Published On:January 2026
By Product Type:The market is segmented into Disposable Custom Procedure Kits and Reusable Custom Procedure Kits. Disposable kits are gaining traction due to their convenience and reduced risk of infection, particularly in high-volume surgical centers and ambulatory settings where rapid turnover and strict infection control are critical. Reusable kits are favored for their cost-effectiveness over multiple procedures and alignment with hospital sustainability initiatives when effective reprocessing and sterilization infrastructure is in place.
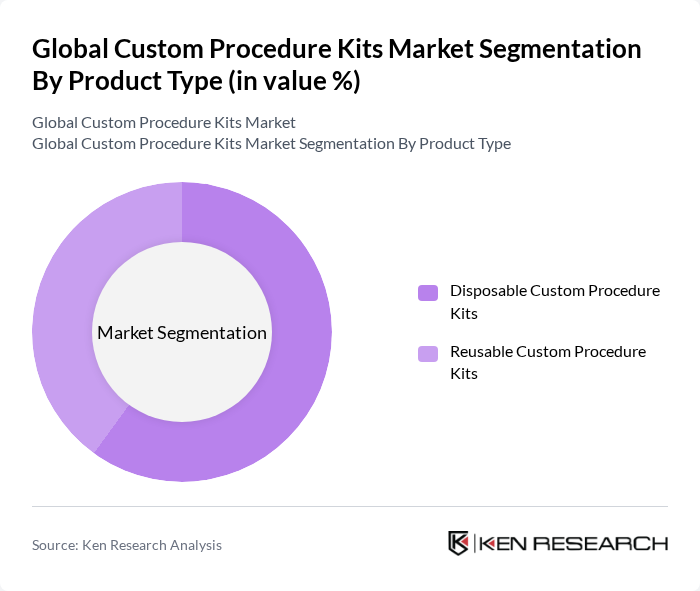
The Disposable Custom Procedure Kits segment is currently leading the market due to the increasing emphasis on infection control and patient safety in healthcare settings, supported by guidelines and policies that encourage the use of single-use components to reduce healthcare-associated infections. The convenience of using single-use kits, which eliminate the need for on-site sterilization and reduce the risk of cross-contamination, has made them a preferred choice among hospitals and ambulatory surgery centers. Additionally, the growing trend of outpatient and same-day surgeries, rising procedural volumes in orthopedics, cardiology, and gynecology, and the need to optimize procedure room turnover further bolster the demand for disposable kits.
By Procedure / Specialty:The market is categorized into Colorectal Surgery Kits, Thoracic Surgery Kits, Orthopedic Surgery Kits, Ophthalmology Procedure Kits, Neurosurgery Kits, Cardiac Surgery Kits, Gynecology & OB/GYN Kits, General Surgery Kits, Urology Procedure Kits, and Others. Each specialty requires specific kits tailored to the unique needs of the procedures, including procedure-specific instruments, drapes, implants or accessories, and standardized consumables to support efficiency and consistency of care.
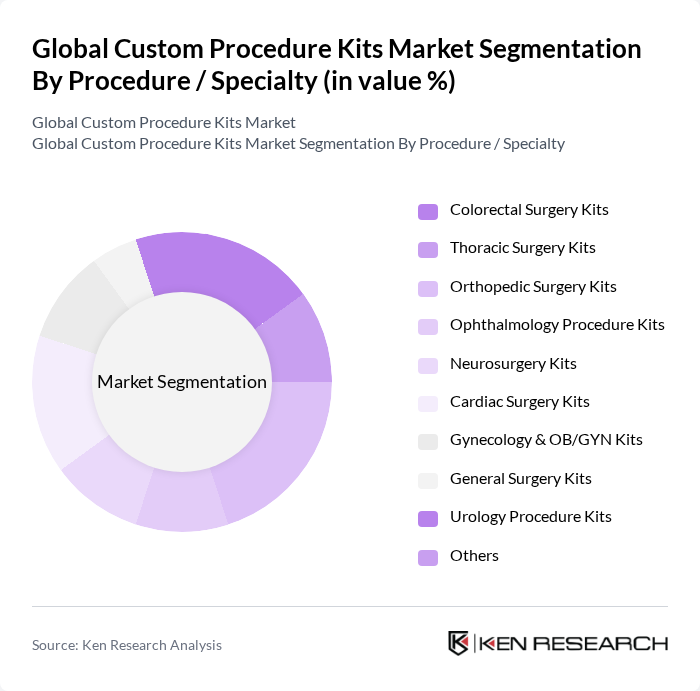
The Orthopedic Surgery Kits segment is currently the dominant player in the market, consistent with industry analyses that identify orthopedic procedures as holding the largest share among procedures, driven by the increasing incidence of joint replacements, trauma surgeries, and spine procedures in aging populations. The demand for specialized kits that cater to various orthopedic procedures, such as hip and knee arthroplasty, arthroscopy, and fracture fixation, is on the rise as providers seek to standardize protocols and reduce operating room setup time. Additionally, advancements in surgical techniques, including minimally invasive and robotic-assisted orthopedic surgery, and the push toward enhanced recovery pathways are further propelling the growth of this segment and increasing the need for highly tailored orthopedic custom packs.
The Global Custom Procedure Kits Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medline Industries, LP, Cardinal Health, Inc., Owens & Minor, Inc., Medtronic plc, McKesson Corporation, Teleflex Incorporated, Terumo Corporation, Mölnlycke Health Care AB, OneMed Group, Santex S.p.A., B. Braun Melsungen AG, 3M Company, Smiths Medical (ICU Medical, Inc.), Hologic, Inc., Olympus Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the custom procedure kits market appears promising, driven by technological advancements and an increasing focus on personalized healthcare. As healthcare systems worldwide invest in digital health technologies, the integration of telemedicine and AI will enhance the customization of kits. Furthermore, the growing emphasis on sustainability will likely lead to the development of eco-friendly kits, aligning with global health initiatives and consumer preferences for environmentally responsible products.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Disposable Custom Procedure Kits Reusable Custom Procedure Kits |
| By Procedure / Specialty | Colorectal Surgery Kits Thoracic Surgery Kits Orthopedic Surgery Kits Ophthalmology Procedure Kits Neurosurgery Kits Cardiac Surgery Kits Gynecology & OB/GYN Kits General Surgery Kits Urology Procedure Kits Others |
| By End-Use | Hospitals Ambulatory Surgical Centers Clinics Homecare Settings Others |
| By Distribution Channel | Direct Sales (Manufacturers to Providers) Group Purchasing Organizations (GPOs) Distributors / Wholesalers Online / E-commerce Channels Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Surgical Departments | 120 | Surgeons, Surgical Coordinators |
| Outpatient Clinics | 90 | Clinic Managers, Healthcare Administrators |
| Medical Device Manufacturers | 70 | Product Managers, R&D Specialists |
| Healthcare Procurement Agencies | 60 | Procurement Officers, Supply Chain Managers |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
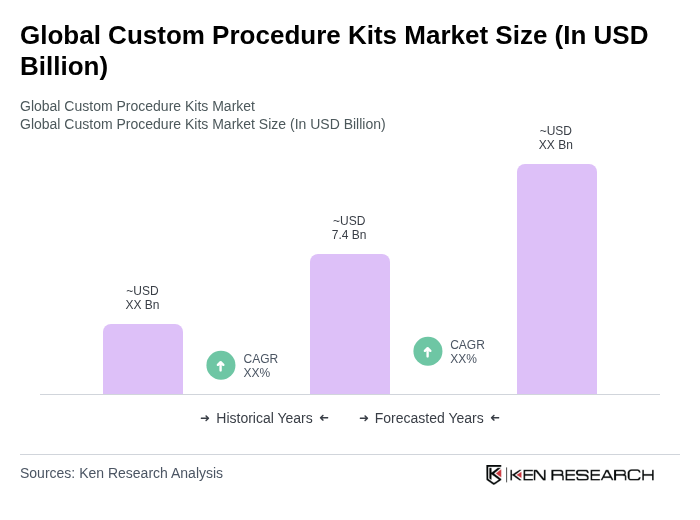
The Global Custom Procedure Kits Market is valued at approximately USD 7.4 billion, reflecting a significant growth trend driven by the demand for personalized healthcare solutions and procedure-specific kits that enhance operational efficiency in healthcare settings.