Region:Global
Author(s):Shubham
Product Code:KRAD2459
Pages:97
Published On:January 2026
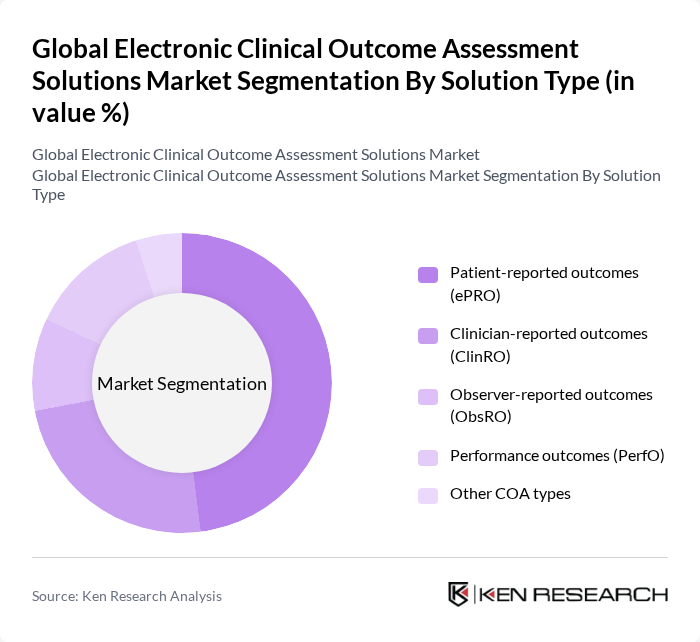
By Solution Type:The market is segmented into various solution types, including Patient-reported outcomes (ePRO), Clinician-reported outcomes (ClinRO), Observer-reported outcomes (ObsRO), Performance outcomes (PerfO), and Other COA types. Among these, Patient-reported outcomes (ePRO) dominate the market due to their ability to provide direct insights from patients regarding their health status and treatment effects, and they represent the largest type segment in leading industry analyses. The increasing emphasis on patient engagement, regulatory support for patient-focused drug development, the use of mobile and bring-your-own-device (BYOD) approaches, and the shift towards personalized medicine have further solidified the importance of ePRO in clinical trials.
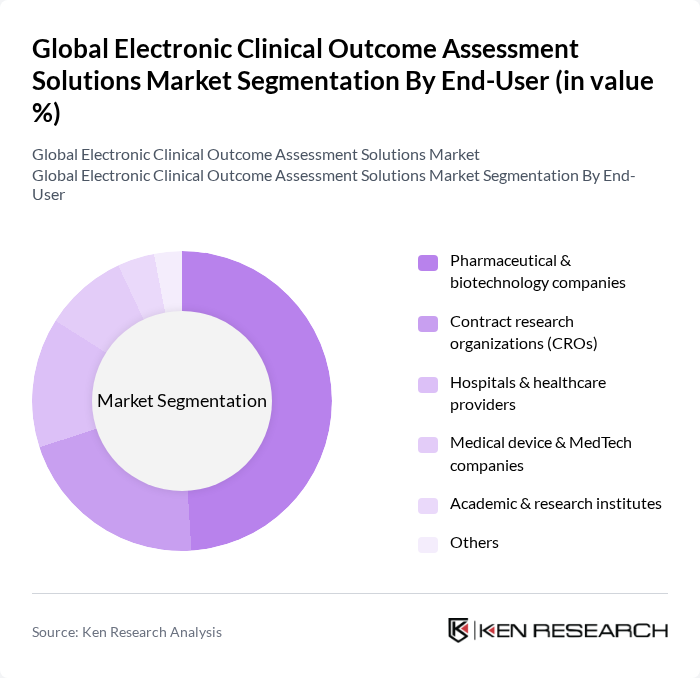
By End-User:The end-user segmentation includes Pharmaceutical & biotechnology companies, Contract research organizations (CROs), Hospitals & healthcare providers, Medical device & MedTech companies, Academic & research institutes, and Others. Pharmaceutical & biotechnology companies lead this segment, driven by their need for efficient clinical trial management, high-volume global study portfolios, and stringent regulatory compliance requirements. The increasing complexity of clinical trials, growing adoption of decentralized and hybrid designs, and the demand for real-time, remote data collection and monitoring have made electronic clinical outcome assessments indispensable for these organizations and their CRO partners.
The Global Electronic Clinical Outcome Assessment Solutions Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medidata Solutions (Dassault Systèmes), Oracle Health Sciences, Parexel International Corporation, Clario (including ERT), CRF Health (Signant Health), Signant Health, Veeva Systems, Clinical Ink, YPrime, Medable, TrialSpark, Bioclinica, ICON plc, Syneos Health, PPD (part of Thermo Fisher Scientific) contribute to innovation, geographic expansion, and service delivery in this space.
The future of eCOA solutions is poised for significant transformation, driven by technological advancements and evolving patient needs. As remote patient monitoring becomes more prevalent, the integration of wearables and mobile applications will enhance data collection and patient engagement. Furthermore, the collaboration between technology providers and pharmaceutical companies is expected to foster innovation, leading to more efficient clinical trials. This synergy will likely result in improved patient outcomes and a more streamlined regulatory process, ultimately shaping the future landscape of eCOA solutions.
| Segment | Sub-Segments |
|---|---|
| By Solution Type | Patient-reported outcomes (ePRO) Clinician-reported outcomes (ClinRO) Observer-reported outcomes (ObsRO) Performance outcomes (PerfO) Other COA types |
| By End-User | Pharmaceutical & biotechnology companies Contract research organizations (CROs) Hospitals & healthcare providers Medical device & MedTech companies Academic & research institutes Others |
| By Application | Clinical trials (onsite, decentralized & hybrid) Real-world evidence & registries Post-marketing surveillance Health economics and outcomes research (HEOR) Others |
| By Deployment / Delivery Mode | Web-hosted solutions Cloud-based solutions On-premise solutions Hybrid deployment |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Modality / Device Type | Smartphones & tablets (native apps) Web-based interfaces Wearables & connected devices Provisioned devices Bring-your-own-device (BYOD) Others |
| By Integration Capability | Standalone eCOA platforms Integrated eCOA with CTMS/EDC/EHR Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Clinical Trials | 120 | Clinical Trial Managers, Regulatory Affairs Specialists |
| Medical Device Evaluations | 90 | Product Development Managers, Clinical Research Coordinators |
| Patient-Reported Outcomes | 70 | Healthcare Providers, Patient Advocacy Representatives |
| Digital Health Solutions | 60 | IT Managers, Digital Health Strategists |
| Regulatory Compliance in Clinical Assessments | 80 | Compliance Officers, Quality Assurance Managers |
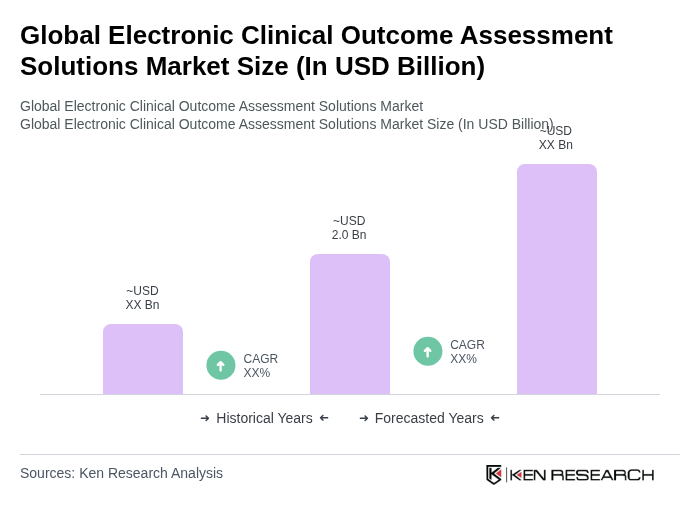
The Global Electronic Clinical Outcome Assessment Solutions Market is valued at approximately USD 2.0 billion, reflecting significant growth driven by the adoption of digital health technologies and the need for efficient data capture in clinical trials.