Region:Global
Author(s):Shubham
Product Code:KRAC2174
Pages:85
Published On:October 2025
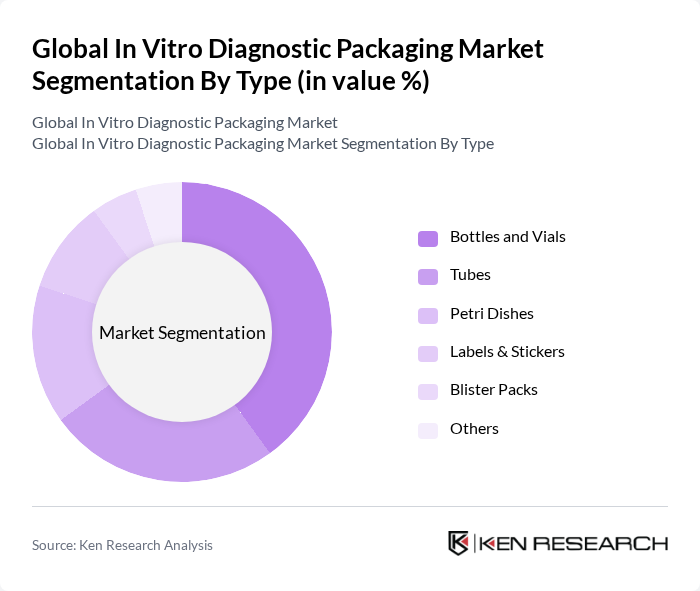
By Type:The market is segmented into various types, including Bottles and Vials, Tubes, Petri Dishes, Labels & Stickers, Blister Packs, and Others. Bottles and Vials are the most dominant sub-segment, accounting for over 40% of market share, due to their extensive use in storing and transporting liquid samples for a wide range of diagnostic tests. Tubes are also in high demand, primarily for blood collection and specimen handling, with safety and contamination prevention driving adoption. Petri Dishes, Labels & Stickers, and Blister Packs serve specialized roles in sample analysis, identification, and sterile transport, respectively.
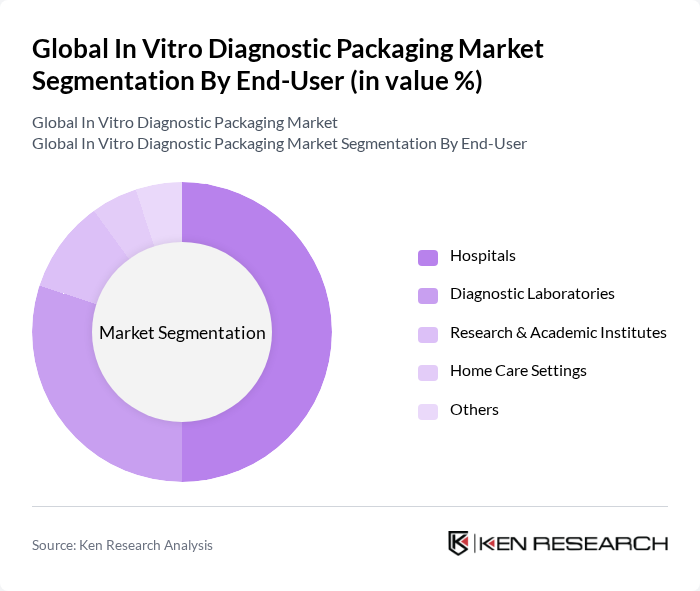
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Research & Academic Institutes, Home Care Settings, and Others. Hospitals are the leading end-user segment, representing over 43% of market share, due to the high volume of diagnostic tests performed and the need for rapid, reliable sample handling. Diagnostic Laboratories follow, driven by outsourcing trends and increased test volumes. Research & Academic Institutes utilize specialized packaging for experimental and clinical studies, while Home Care Settings are growing with the rise of self-testing and point-of-care diagnostics.
The Global In Vitro Diagnostic Packaging Market is characterized by a dynamic mix of regional and international players. Leading participants such as Thermo Fisher Scientific Inc., Becton, Dickinson and Company, Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Agilent Technologies, Inc., QIAGEN N.V., Bio-Rad Laboratories, Inc., Hologic, Inc., PerkinElmer, Inc., Danaher Corporation, Merck KGaA, Sysmex Corporation, Ortho Clinical Diagnostics, Greiner Bio-One International GmbH, Corning Incorporated contribute to innovation, geographic expansion, and service delivery in this space.
The future of the in vitro diagnostic packaging market appears promising, driven by ongoing innovations and a growing emphasis on patient-centric solutions. As the healthcare landscape evolves, the integration of digital technologies and smart packaging will enhance product tracking and patient engagement. Additionally, the shift towards personalized medicine will necessitate tailored packaging solutions, ensuring that diagnostic products meet specific patient needs while maintaining compliance with regulatory standards and sustainability goals.
| Segment | Sub-Segments |
|---|---|
| By Type | Bottles and Vials Tubes Petri Dishes Labels & Stickers Blister Packs Others |
| By End-User | Hospitals Diagnostic Laboratories Research & Academic Institutes Home Care Settings Others |
| By Material | Plastic Glass Paper & Paperboard Metal Others |
| By Application | Infectious Disease Testing Cancer Diagnostics Blood Glucose Monitoring Genetic Testing Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Retail Pharmacies Others |
| By Region | North America (U.S., Canada, Mexico) Europe (Germany, U.K., France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe) Asia-Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia-Pacific) Latin America (Brazil, Argentina, Rest of Latin America) Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa) |
| By Price Range | Low Price Mid Price High Price Premium Price Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| IVD Packaging Innovations | 100 | Product Development Managers, R&D Directors |
| Regulatory Compliance in IVD Packaging | 80 | Quality Assurance Managers, Regulatory Affairs Specialists |
| Supply Chain Management for IVD Products | 60 | Logistics Coordinators, Supply Chain Analysts |
| Market Trends in Diagnostic Packaging | 90 | Market Research Analysts, Business Development Managers |
| End-user Feedback on IVD Packaging | 50 | Healthcare Professionals, Laboratory Managers |
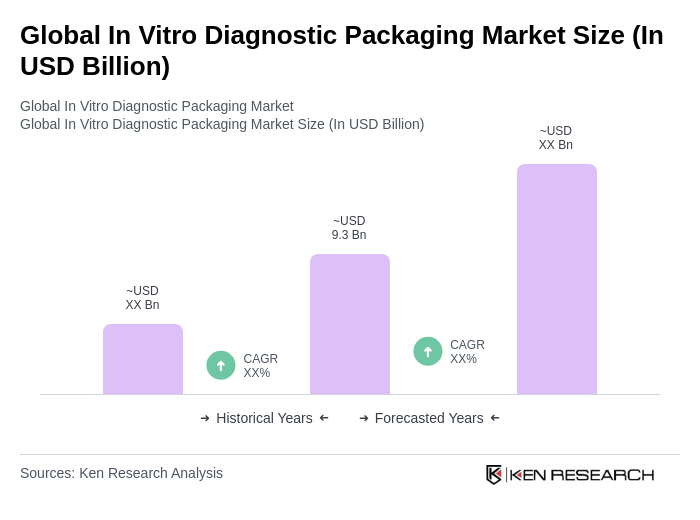
The Global In Vitro Diagnostic Packaging Market is valued at approximately USD 9.3 billion, driven by increasing demand for diagnostic testing, advancements in healthcare technologies, and the rising prevalence of chronic diseases.