Region:Global
Author(s):Rebecca
Product Code:KRAD2028
Pages:88
Published On:January 2026
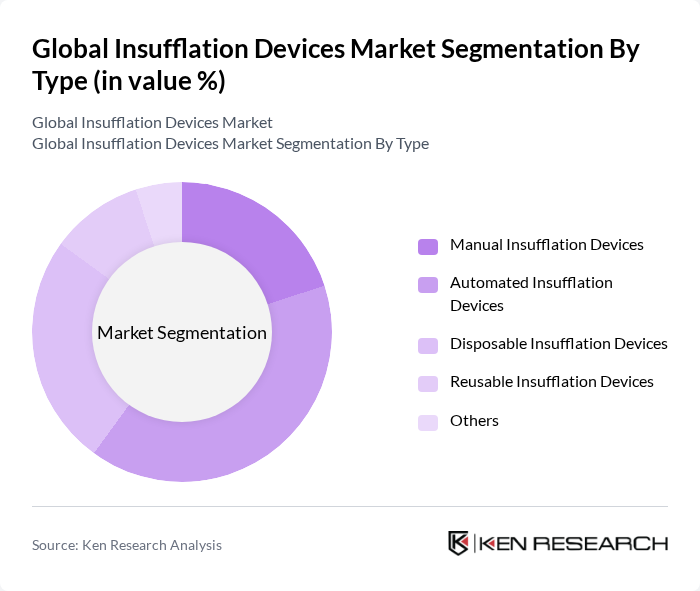
By Type:The insufflation devices market is segmented into various types, including manual, automated, disposable, reusable, and others. Among these, automated insufflation devices are gaining traction due to their precision and efficiency, which significantly reduce the risk of human error during surgical procedures. The demand for disposable devices is also on the rise, driven by the increasing focus on infection control and patient safety in healthcare settings.
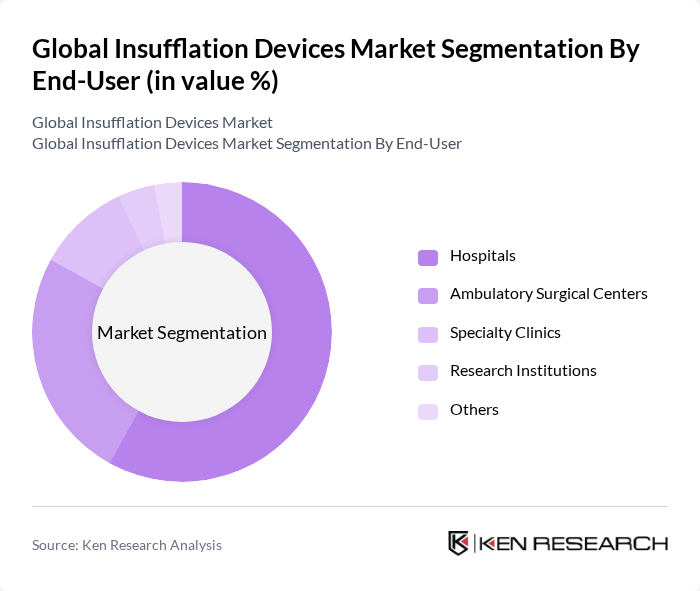
By End-User:The market is categorized by end-users, including hospitals, ambulatory surgical centers, specialty clinics, research institutions, and others. Hospitals are the leading end-users, accounting for approximately 58% market share, primarily due to their extensive surgical facilities and higher patient volumes. Ambulatory surgical centers are also witnessing significant growth, driven by the increasing preference for outpatient procedures and cost-effective healthcare solutions.
The Global Insufflation Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Johnson & Johnson, Stryker Corporation, Olympus Corporation, Karl Storz, Conmed Corporation, B. Braun Melsungen AG, Richard Wolf GmbH, Hologic, Inc., Smith & Nephew, Ambu A/S, Cook Medical, Ethicon, Inc., Medline Industries, Inc., Teleflex Incorporated contribute to innovation, geographic expansion, and service delivery in this space.
The future of the insufflation devices market appears promising, driven by technological advancements and an increasing focus on minimally invasive surgical techniques. The integration of AI and hybrid systems is expected to enhance device functionality, while the growing demand for outpatient procedures will likely lead to innovations in portable and user-friendly designs. As healthcare infrastructure continues to improve, particularly in emerging markets, the market is poised for significant growth, addressing the evolving needs of surgical practices.
| Segment | Sub-Segments |
|---|---|
| By Type | Manual Insufflation Devices Automated Insufflation Devices Disposable Insufflation Devices Reusable Insufflation Devices Others |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Clinics Research Institutions Others |
| By Application | Laparoscopic Surgery Endoscopic Procedures Urological Procedures Gynecological Procedures Others |
| By Product Type | Insufflation Pumps Insufflation Tubes Insufflation Needles Insufflation Valves Others |
| By Technology | Pneumatic Insufflation Electric Insufflation Hybrid Insufflation Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| General Surgery Insufflation Devices | 120 | Surgeons, Operating Room Managers |
| Gynecological Surgery Insufflation Devices | 100 | Gynecologists, Surgical Assistants |
| Urological Surgery Insufflation Devices | 80 | Urologists, Hospital Administrators |
| Endoscopic Insufflation Devices | 70 | Gastroenterologists, Endoscopy Unit Managers |
| Veterinary Insufflation Devices | 50 | Veterinarians, Veterinary Surgeons |
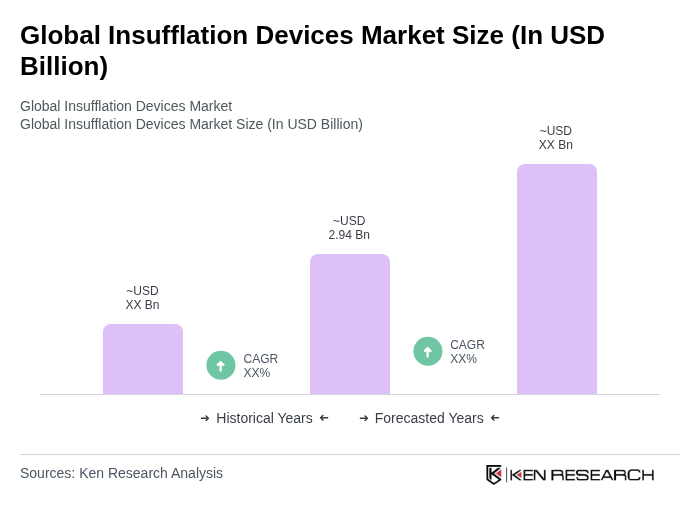
The Global Insufflation Devices Market is valued at approximately USD 2.94 billion, driven by the increasing adoption of minimally invasive surgeries and technological advancements in insufflation devices.