Global Transcatheter Embolization and Occlusion Devices Market Overview
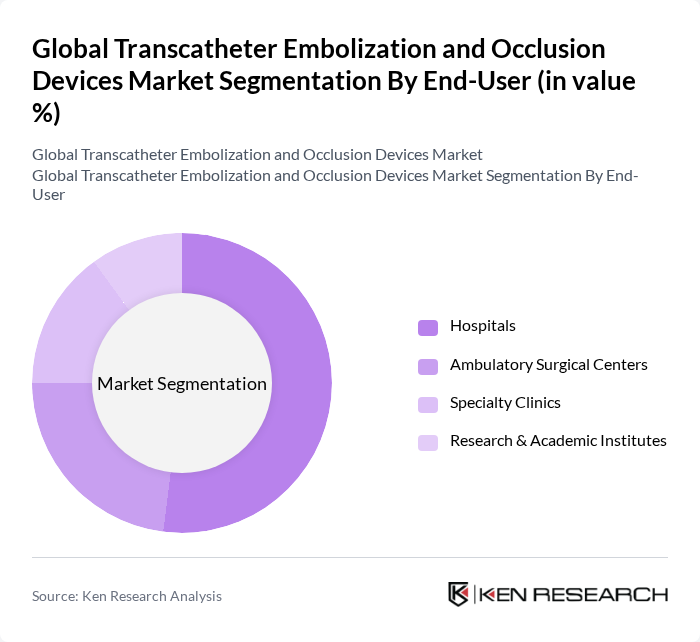
- The Global Transcatheter Embolization and Occlusion Devices Market is valued at USD 5.5 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of vascular diseases, such as peripheral vascular disease and aneurysms, the rising burden of oncology indications treated with embolization (for example, tumor embolization and transarterial chemoembolization), and a growing incidence of neurovascular disorders. Advancements in minimally invasive endovascular and interventional radiology techniques, coupled with continuous device innovations (including hydrogel-coated coils, advanced vascular plugs, and next?generation liquid embolic agents), are further enhancing the efficacy and safety of embolization procedures and supporting market expansion.
- Key players in this market are predominantly located in North America and Europe, with the United States and major European countries such as Germany, the United Kingdom, and France being the most significant contributors. The dominance of these regions is supported by advanced healthcare infrastructure, high healthcare expenditure, broad availability of interventional radiology and neurointerventional suites, and the strong presence of leading medical device manufacturers such as Medtronic, Boston Scientific, Stryker, Terumo, Penumbra, and others. Additionally, favorable reimbursement policies, rapid adoption of innovative medical technologies (including flow diverters and novel liquid embolics), and active clinical research networks in these countries further solidify their market leadership.
- In the United States, regulation of transcatheter embolization and occlusion devices is governed by the Federal Food, Drug, and Cosmetic Act and implementing regulations in Title 21 of the Code of Federal Regulations (CFR), including device classification, premarket approval, and quality system requirements enforced by the U.S. Food and Drug Administration (FDA). Embolization and occlusion devices are generally treated as Class II or Class III medical devices and are subject to premarket notification or Premarket Approval (PMA), clinical investigation requirements under 21 CFR Part 812, and post?market surveillance and adverse event reporting obligations under 21 CFR Part 803. In recent years, FDA has emphasized more robust clinical evidence, adoption of real?world data, and strengthened post?market monitoring for high?risk interventional devices, which collectively aim to enhance safety and effectiveness while supporting innovation in transcatheter embolization technologies.
Global Transcatheter Embolization and Occlusion Devices Market Segmentation
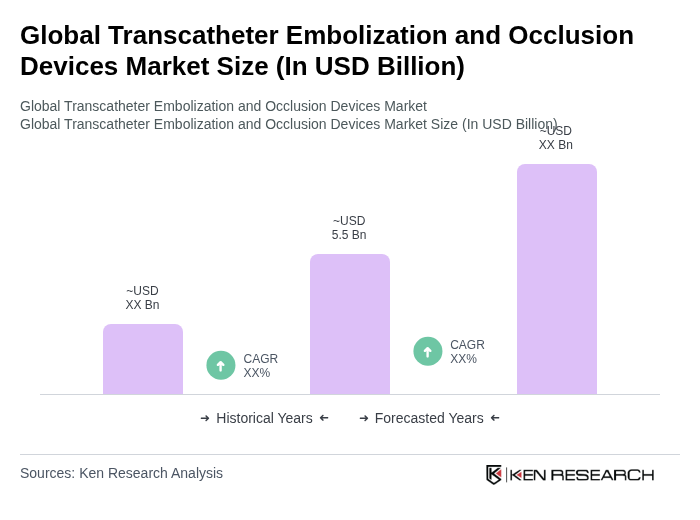
By Product Type:The product type segmentation includes various categories such as embolization coils, embolization particles/microspheres, liquid embolic agents, vascular plugs, flow diverter devices, coiling-assist devices (balloons/stents), and accessories (microcatheters, guidewires, others). Among these, embolization coils continue to account for the largest share of revenue owing to their widespread use in both peripheral and neurovascular procedures and long-standing physician familiarity. Coils are widely used because of their effectiveness in aneurysm and vessel occlusion, predictable deployment, and compatibility with a broad range of microcatheters. At the same time, liquid embolic agents and non?coil devices are growing rapidly driven by their ability to treat complex anatomy, reduce procedure time, and improve long?term occlusion rates, especially in neurovascular and oncology applications.
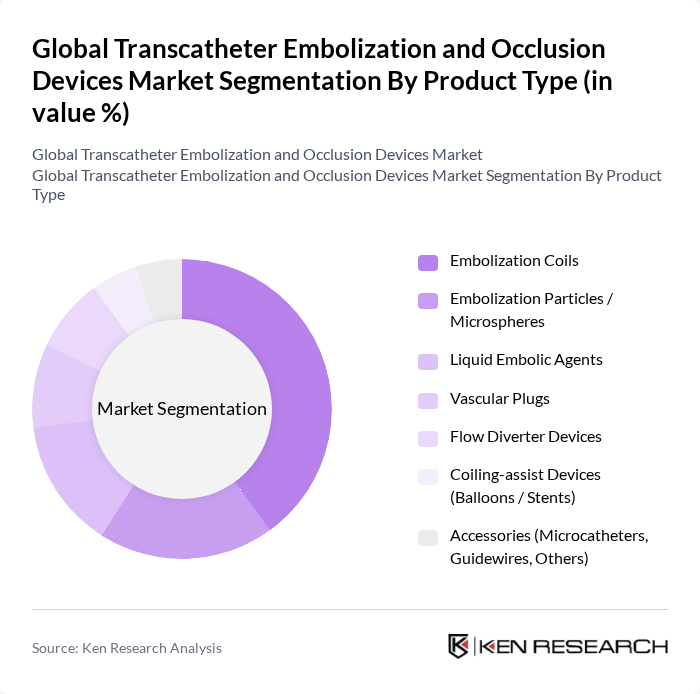
By End-User:The end-user segmentation encompasses hospitals, ambulatory surgical centers, specialty clinics, and research & academic institutes. Hospitals dominate this segment due to their comprehensive facilities, availability of multidisciplinary teams (interventional radiologists, neurosurgeons, vascular surgeons, and anesthesiologists), and the concentration of complex neurovascular and oncology embolization procedures in tertiary care centers. The increasing number of image?guided endovascular procedures performed in hospitals, along with a growing shift of selected interventional cases to ambulatory surgical centers as infrastructure and reimbursement frameworks mature, is driving sustained demand for transcatheter embolization and occlusion devices across care settings.
Global Transcatheter Embolization and Occlusion Devices Market Competitive Landscape
The Global Transcatheter Embolization and Occlusion Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Boston Scientific Corporation, Johnson & Johnson (Cerenovus), Abbott Laboratories, Terumo Corporation, Cook Medical LLC, Stryker Corporation, Penumbra, Inc., Merit Medical Systems, Inc., AngioDynamics, Inc., Balt Group, Kaneka Corporation, Phenox GmbH, Acandis GmbH, MicroPort Scientific Corporation contribute to innovation, geographic expansion, and service delivery in this space.
Global Transcatheter Embolization and Occlusion Devices Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Vascular Diseases:The rise in vascular diseases, such as peripheral artery disease and aneurysms, is a significant growth driver for transcatheter embolization devices. According to the World Health Organization, approximately 17.9 million people died from cardiovascular diseases in the past, with vascular diseases accounting for a substantial portion. This alarming statistic underscores the urgent need for effective treatment options, propelling the demand for embolization procedures and related devices in the healthcare sector.
- Rising Geriatric Population:The global geriatric population is projected to reach around 1.5 billion in future, according to the United Nations. This demographic shift is crucial as older adults are more susceptible to vascular diseases, leading to an increased demand for transcatheter embolization and occlusion devices. In future, the number of individuals aged 65 and older is expected to surpass 700 million, creating a significant market opportunity for healthcare providers and device manufacturers to cater to this vulnerable population.
- Growing Demand for Embolization Procedures:The demand for minimally invasive procedures, including embolization, is on the rise due to their associated benefits such as reduced recovery time and lower complication rates. The American College of Surgeons reported that the number of embolization procedures performed annually has increased by over 30% in the last five years. This trend is expected to continue, driving the need for advanced transcatheter embolization devices to meet the growing procedural volume in hospitals and outpatient settings.
Market Challenges
- High Cost of Devices:The high cost associated with transcatheter embolization devices poses a significant challenge to market growth. For instance, the average price of embolization coils can range from $1,000 to $3,000 per unit, which can limit accessibility for healthcare facilities, especially in developing regions. This financial barrier can hinder the adoption of these devices, impacting overall market expansion and patient access to necessary treatments.
- Stringent Regulatory Requirements:Regulatory hurdles present a considerable challenge for manufacturers of transcatheter embolization devices. In the United States, the FDA requires extensive clinical trials and data submission for device approval, which can take several years and incur significant costs. Similarly, the European Union's CE marking process involves rigorous assessments, delaying product launches and increasing the time to market for innovative solutions, thereby affecting overall industry growth.
Global Transcatheter Embolization and Occlusion Devices Market Future Outlook
The future of the transcatheter embolization and occlusion devices market appears promising, driven by technological advancements and an increasing focus on patient-centric care. As healthcare systems evolve, there is a notable shift towards outpatient procedures, which are expected to enhance patient satisfaction and reduce hospital stays. Additionally, the integration of digital technologies, such as telemedicine and AI, is likely to streamline processes and improve outcomes, further propelling market growth in the coming years.
Market Opportunities
- Expansion in Emerging Markets:Emerging markets present significant growth opportunities for transcatheter embolization devices. Countries like India and Brazil are experiencing rapid urbanization and healthcare infrastructure development, leading to increased access to advanced medical technologies. The World Bank projects healthcare spending in these regions to rise by 10% annually, creating a favorable environment for market players to introduce innovative solutions tailored to local needs.
- Development of Innovative Products:The continuous development of innovative embolization products, such as bioresorbable materials and advanced delivery systems, offers substantial market opportunities. Research indicates that the global market for bioresorbable devices is expected to reach $5 billion in future. This innovation not only enhances treatment efficacy but also aligns with the growing demand for safer and more effective medical solutions, positioning companies favorably in a competitive landscape.