Region:Asia
Author(s):Rebecca
Product Code:KRAE3367
Pages:86
Published On:February 2026
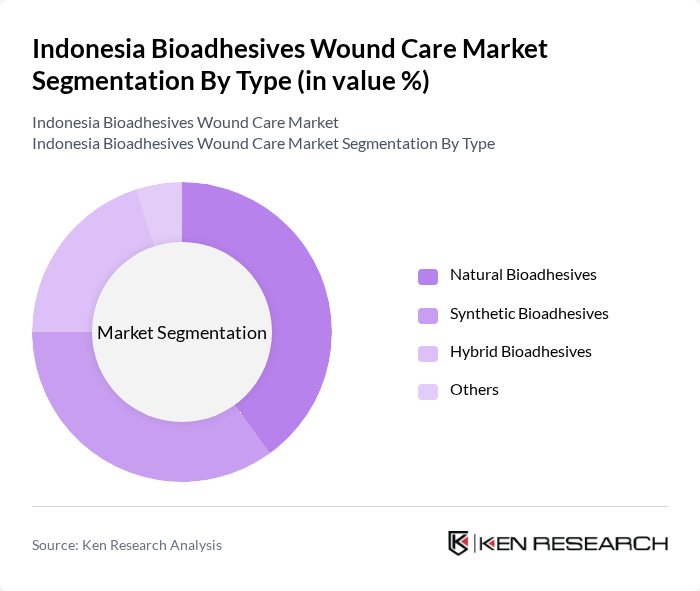
By Type:The market is segmented into various types of bioadhesives, including natural, synthetic, hybrid, and others. Among these, natural bioadhesives are gaining traction due to their biocompatibility and reduced risk of adverse reactions. Synthetic bioadhesives, on the other hand, are favored for their durability and effectiveness in various applications. The hybrid category combines the benefits of both natural and synthetic options, catering to diverse consumer needs.
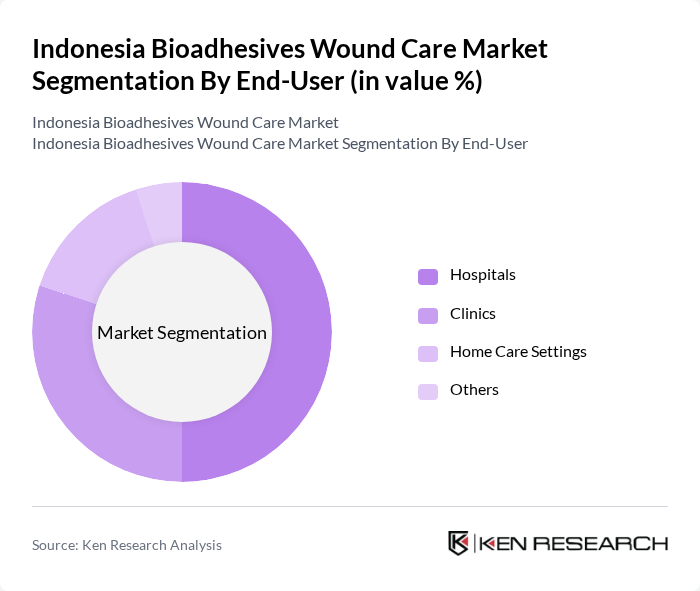
By End-User:The end-user segmentation includes hospitals, clinics, home care settings, and others. Hospitals are the leading end-users due to their extensive use of bioadhesives in surgical procedures and wound management. Clinics also contribute significantly, particularly in outpatient care. Home care settings are emerging as a growing segment as patients increasingly manage their wounds at home, aided by bioadhesive products.
The Indonesia Bioadhesives Wound Care Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT. Medika Sarana Tiara, PT. Kimia Farma Tbk, PT. Indofarma Tbk, PT. Biofarma, PT. Sido Muncul, PT. Kalbe Farma Tbk, PT. Darya-Varia Laboratoria Tbk, PT. Harsen Laboratories, PT. Anugerah Pharmindo Lestari, PT. Surya Dermato Medica, PT. Medisafe Technologies, PT. Prodia Widyahusada Tbk, PT. Novell Pharmaceutical Laboratories, PT. Bintang Toedjoe, PT. Murni Solusindo contribute to innovation, geographic expansion, and service delivery in this space.
The future of the bioadhesives wound care market in Indonesia appears promising, driven by increasing healthcare investments and a growing focus on advanced medical technologies. As the government continues to enhance healthcare infrastructure, the adoption of bioadhesives is expected to rise. Furthermore, the integration of smart technologies in wound management will likely create new avenues for product development, catering to the evolving needs of healthcare providers and patients alike, ultimately improving treatment outcomes.
| Segment | Sub-Segments |
|---|---|
| By Type | Natural Bioadhesives Synthetic Bioadhesives Hybrid Bioadhesives Others |
| By End-User | Hospitals Clinics Home Care Settings Others |
| By Application | Surgical Wounds Traumatic Wounds Chronic Wounds Others |
| By Distribution Channel | Direct Sales Online Retail Distributors Others |
| By Region | Java Sumatra Bali Others |
| By Product Form | Gels Films Sprays Others |
| By Patient Demographics | Pediatric Adult Geriatric Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Professionals in Wound Care | 150 | Doctors, Nurses, Wound Care Specialists |
| Manufacturers of Bioadhesives | 100 | Product Managers, R&D Directors |
| Patients Using Wound Care Products | 80 | Patients, Caregivers, Health Advocates |
| Healthcare Facility Administrators | 70 | Hospital Administrators, Procurement Officers |
| Regulatory Bodies and Health Authorities | 50 | Policy Makers, Regulatory Affairs Managers |
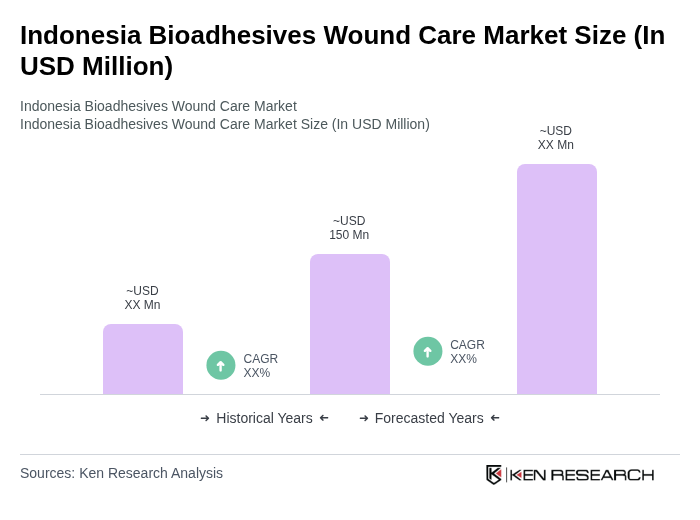
The Indonesia Bioadhesives Wound Care Market is valued at approximately USD 150 million, reflecting a significant growth trend driven by the increasing prevalence of chronic wounds and advancements in wound care technologies.