About the Report
Base Year 2024Indonesia External Counterpulsation Devices Market Overview
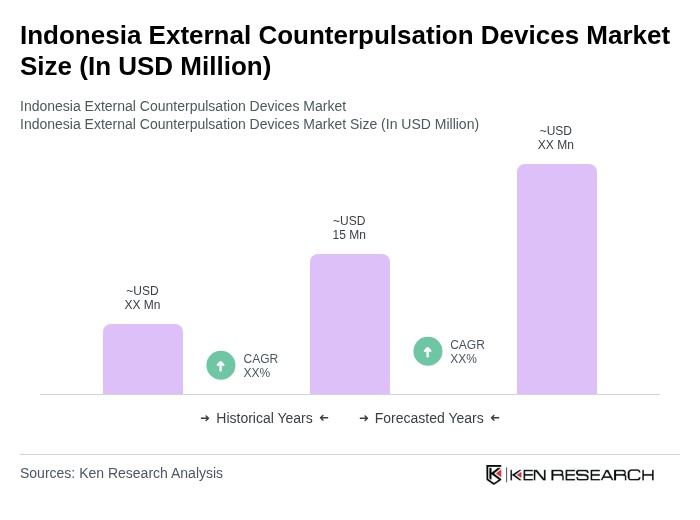
- The Indonesia External Counterpulsation Devices Market is valued at USD 15 million, based on a five-year historical analysis of the adoption of non-invasive cardiac therapies and the overall size of the cardiovascular devices segment in the country. This growth is primarily driven by the increasing prevalence and burden of cardiovascular diseases, including ischemic heart disease and stroke, which together account for a substantial share of mortality and disability in Indonesia. Rising healthcare expenditure, expanding national health insurance coverage, and advancements in medical technology are further supporting the adoption of advanced cardiac care solutions. The demand for non-invasive treatment options, such as external counterpulsation devices, has increased as healthcare providers seek cost-effective, outpatient-based therapies for patients with chronic heart conditions who are not optimal candidates for invasive revascularization.
- Key cities dominating the market include Jakarta, Surabaya, and Bandung. Jakarta, as the capital, concentrates a large share of Indonesia’s tertiary and referral hospitals, specialist cardiac centers, and private hospital chains, combined with higher population density and purchasing power, which drives demand for innovative medical devices and advanced cardiovascular care. Surabaya and Bandung also contribute significantly due to the rapid expansion of hospital infrastructure on Java Island, growing numbers of cardiologists and interventional cardiology units, and increasing awareness and diagnosis of coronary artery disease and heart failure among the urban population.
- In 2023, the Indonesian government implemented regulations to enhance the quality, safety, and performance of medical devices, including external counterpulsation systems. The key binding framework is the Regulation of the Minister of Health of the Republic of Indonesia Number 14 of 2021 concerning the Standards of Business Activities and Products in the Implementation of Risk-Based Business Licensing in the Health Sector, issued by the Ministry of Health, which is operationalized for medical devices through the mandatory registration and licensing regime under the Indonesia Medical Device Registration (Regalkes) and Online Single Submission system. This regulatory framework requires that medical devices be classified by risk, obtain marketing authorization from the Ministry of Health, comply with applicable quality and safety standards (including testing, clinical evaluation where required, and post?market surveillance), and be distributed only through licensed establishments, thereby reinforcing trust among healthcare providers and patients in the use of technologies such as external counterpulsation devices.
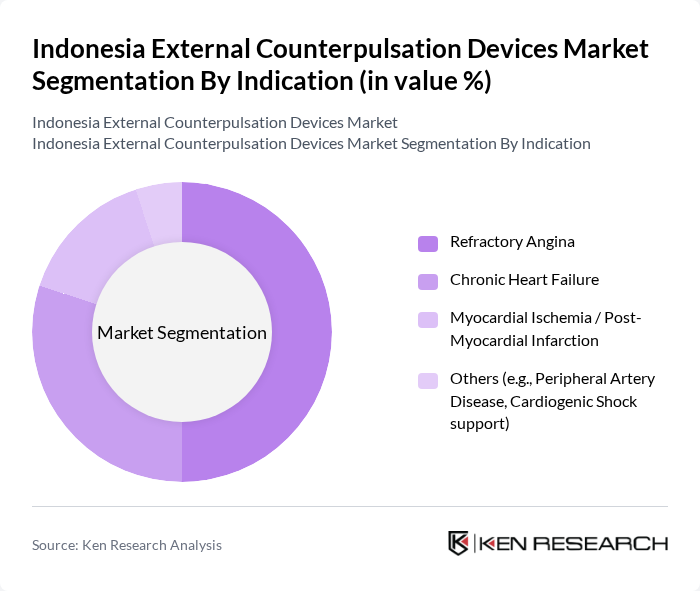
Indonesia External Counterpulsation Devices Market Segmentation
By Device Type:The market is segmented into various device types, including Fixed External Counterpulsation Systems, Portable / Mobile External Counterpulsation Systems, Multi-channel EECP Systems, and Others. Among these, Fixed External Counterpulsation Systems dominate the market due to their widespread use in hospitals and dedicated cardiac care centers, where they can be integrated into comprehensive programs for refractory angina and chronic coronary syndromes. Their reliability, ability to deliver standardized treatment protocols, and suitability for higher-throughput clinical environments make them the preferred choice for many healthcare providers. The increasing number of cardiac specialty centers and referral hospitals managing complex ischemic heart disease and heart failure patients further supports the demand for these systems, while portable and multi?channel configurations are gradually gaining traction in private clinics and wellness-oriented cardiovascular programs.

By Indication:The market is categorized based on indications such as Refractory Angina, Chronic Heart Failure, Myocardial Ischemia / Post-Myocardial Infarction, and Others. Refractory Angina is the leading indication, reflecting the substantial burden of ischemic heart disease in Indonesia and the clinical need for adjunctive therapies in patients who remain symptomatic or are not ideal candidates for additional revascularization. The high and rising prevalence of coronary artery disease, combined with growing awareness of non?invasive options among cardiologists, supports the use of external counterpulsation to improve exercise tolerance and quality of life in these patients. Chronic Heart Failure and post?myocardial infarction ischemia also represent important segments, as heart failure admissions and post?acute coronary syndrome management account for a large share of cardiovascular healthcare utilization and costs in Indonesia, encouraging interest in supportive therapies that can be delivered on an outpatient basis.
Indonesia External Counterpulsation Devices Market Competitive Landscape
The Indonesia External Counterpulsation Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Vasomedical, Inc., PSK-Health (PSK International), Guangzhou Yinghui Medical Equipment Co., Ltd., Chongqing PSK-Health Sci-Tech Development Co., Ltd., NCP International (NeoCardial External Counterpulsation), Advanced Cardiac Therapeutics Pte. Ltd. (Regional Distributor), Medtronic plc, Abbott Laboratories, Boston Scientific Corporation, GE Healthcare, Philips Healthcare, Siemens Healthineers, Terumo Corporation, Local Indonesian Distributors (e.g., PT Enseval Putera Megatrading Tbk, PT Kimia Farma Tbk – medical devices division), Other Emerging Regional EECP Vendors contribute to innovation, geographic expansion, and service delivery in this space.
Indonesia External Counterpulsation Devices Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Cardiovascular Diseases:The World Health Organization reported that cardiovascular diseases (CVDs) account for approximately 33% of all deaths in Indonesia, translating to over 650,000 fatalities annually. This alarming statistic underscores the urgent need for effective treatment options, including external counterpulsation devices. As the population ages and lifestyle-related health issues rise, the demand for innovative cardiovascular treatments is expected to surge, driving market growth significantly.
- Rising Awareness About Non-Invasive Treatment Options:A recent survey indicated that 60% of Indonesian patients prefer non-invasive treatments for cardiovascular issues, reflecting a growing awareness of alternative therapies. This shift is supported by educational campaigns from healthcare providers and NGOs, which have increased public knowledge about the benefits of external counterpulsation devices. As more patients seek these options, the market is poised for expansion, with an estimated 20% increase in device adoption expected in future.
- Technological Advancements in Medical Devices:The Indonesian medical device market is projected to reach about USD 3 billion in value in future, driven by rapid technological advancements. Innovations in external counterpulsation devices, such as improved efficacy and user-friendly designs, are attracting healthcare providers. The integration of smart technologies, including remote monitoring capabilities, is enhancing patient outcomes and driving demand. As hospitals adopt these advanced devices, the market is expected to experience robust growth, with a projected increase of 15% in device installations.
Market Challenges
- High Cost of External Counterpulsation Devices:The average cost of external counterpulsation devices in Indonesia ranges from about USD 15,000 to about USD 25,000, which poses a significant barrier to widespread adoption. Many healthcare facilities, especially in rural areas, struggle to allocate budgets for such expensive equipment. This financial constraint limits access to advanced treatment options for patients, hindering market growth. As a result, only a small percentage of hospitals currently offer these devices, impacting overall market penetration.
- Limited Reimbursement Policies:The Indonesian healthcare system has limited reimbursement policies for external counterpulsation treatments, which discourages healthcare providers from investing in these devices. Currently, only 30% of hospitals receive adequate reimbursement for such treatments, leading to financial losses. This lack of support from insurance providers creates a significant challenge for market growth, as many patients are unable to afford out-of-pocket expenses for these non-invasive therapies, limiting their accessibility.
Indonesia External Counterpulsation Devices Market Future Outlook
The future of the external counterpulsation devices market in Indonesia appears promising, driven by increasing healthcare investments and a growing focus on preventive care. As the government enhances healthcare infrastructure, more facilities are expected to adopt advanced medical technologies. Additionally, the rising trend of telemedicine and home-based healthcare solutions will likely facilitate greater access to these devices, improving patient outcomes. Overall, the market is set to evolve significantly, aligning with global healthcare trends and technological advancements.
Market Opportunities
- Expansion of Healthcare Facilities in Rural Areas:The Indonesian government plans to invest about USD 1 billion in healthcare infrastructure by future, focusing on rural areas. This initiative will increase access to external counterpulsation devices, enabling more patients to benefit from advanced cardiovascular treatments. As healthcare facilities expand, the demand for these devices is expected to rise, creating significant market opportunities.
- Increasing Demand for Preventive Healthcare Solutions:With a growing emphasis on preventive healthcare, the demand for non-invasive treatments is on the rise. The Indonesian Ministry of Health reported a significant growth in preventive health programs in recent periods. This trend is likely to drive the adoption of external counterpulsation devices, as more patients seek proactive solutions to manage cardiovascular health, presenting a lucrative opportunity for market players.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Device Type | Fixed External Counterpulsation Systems Portable / Mobile External Counterpulsation Systems Multi-channel EECP Systems Others |
| By Indication | Refractory Angina Chronic Heart Failure Myocardial Ischemia / Post-Myocardial Infarction Others (e.g., Peripheral Artery Disease, Cardiogenic Shock support) |
| By End-User | Tertiary Care Hospitals Cardiac Specialty Centers Multi-specialty Clinics Others (including Research & Academic Institutes) |
| By Distribution Channel | Direct Sales to Healthcare Providers Local Distributors / Importers Tender-based Institutional Procurement Others |
| By Region | Java (Jakarta, West Java, Central Java, East Java, Banten, Yogyakarta) Sumatra Kalimantan Sulawesi & Eastern Indonesia (Bali, Nusa Tenggara, Maluku, Papua) |
| By Facility Ownership | Public / Government Hospitals Private Hospitals Independent Cardiac Clinics Others |
| By Patient Profile | Elderly Patients (?65 years) Middle-aged Patients (45–64 years) High-risk Co-morbid Patients (Diabetes, Hypertension, Obesity) Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, National Agency of Drug and Food Control)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Importers
Industry Associations
Health Insurance Companies
Players Mentioned in the Report:
Vasomedical, Inc.
PSK-Health (PSK International)
Guangzhou Yinghui Medical Equipment Co., Ltd.
Chongqing PSK-Health Sci-Tech Development Co., Ltd.
NCP International (NeoCardial External Counterpulsation)
Advanced Cardiac Therapeutics Pte. Ltd. (Regional Distributor)
Medtronic plc
Abbott Laboratories
Boston Scientific Corporation
GE Healthcare
Philips Healthcare
Siemens Healthineers
Terumo Corporation
Local Indonesian Distributors (e.g., PT Enseval Putera Megatrading Tbk, PT Kimia Farma Tbk medical devices division)
Other Emerging Regional EECP Vendors
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Indonesia External Counterpulsation Devices Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Indonesia External Counterpulsation Devices Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Indonesia External Counterpulsation Devices Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of cardiovascular diseases
3.1.2 Rising awareness about non-invasive treatment options
3.1.3 Technological advancements in medical devices
3.1.4 Government initiatives to improve healthcare infrastructure
3.2 Market Challenges
3.2.1 High cost of external counterpulsation devices
3.2.2 Limited reimbursement policies
3.2.3 Lack of trained professionals
3.2.4 Regulatory hurdles in product approval
3.3 Market Opportunities
3.3.1 Expansion of healthcare facilities in rural areas
3.3.2 Increasing demand for preventive healthcare solutions
3.3.3 Collaborations with local healthcare providers
3.3.4 Growth in medical tourism
3.4 Market Trends
3.4.1 Shift towards home-based healthcare solutions
3.4.2 Integration of AI in medical devices
3.4.3 Focus on patient-centric care
3.4.4 Rise in telemedicine services
3.5 Government Regulation
3.5.1 Implementation of stricter quality control measures
3.5.2 Introduction of new healthcare policies
3.5.3 Guidelines for clinical trials
3.5.4 Regulations on medical device marketing
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Indonesia External Counterpulsation Devices Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Indonesia External Counterpulsation Devices Market Segmentation
8.1 By Device Type
8.1.1 Fixed External Counterpulsation Systems
8.1.2 Portable / Mobile External Counterpulsation Systems
8.1.3 Multi-channel EECP Systems
8.1.4 Others
8.2 By Indication
8.2.1 Refractory Angina
8.2.2 Chronic Heart Failure
8.2.3 Myocardial Ischemia / Post-Myocardial Infarction
8.2.4 Others (e.g., Peripheral Artery Disease, Cardiogenic Shock support)
8.3 By End-User
8.3.1 Tertiary Care Hospitals
8.3.2 Cardiac Specialty Centers
8.3.3 Multi-specialty Clinics
8.3.4 Others (including Research & Academic Institutes)
8.4 By Distribution Channel
8.4.1 Direct Sales to Healthcare Providers
8.4.2 Local Distributors / Importers
8.4.3 Tender-based Institutional Procurement
8.4.4 Others
8.5 By Region
8.5.1 Java (Jakarta, West Java, Central Java, East Java, Banten, Yogyakarta)
8.5.2 Sumatra
8.5.3 Kalimantan
8.5.4 Sulawesi & Eastern Indonesia (Bali, Nusa Tenggara, Maluku, Papua)
8.6 By Facility Ownership
8.6.1 Public / Government Hospitals
8.6.2 Private Hospitals
8.6.3 Independent Cardiac Clinics
8.6.4 Others
8.7 By Patient Profile
8.7.1 Elderly Patients (?65 years)
8.7.2 Middle-aged Patients (45–64 years)
8.7.3 High-risk Co-morbid Patients (Diabetes, Hypertension, Obesity)
8.7.4 Others
9. Indonesia External Counterpulsation Devices Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Indonesia Revenue (USD million) and 3-year CAGR
9.2.4 Installed Base of EECP Systems in Indonesia
9.2.5 Market Share in Indonesia EECP Segment (%)
9.2.6 EBITDA Margin (%) from Indonesia Operations
9.2.7 Average Selling Price (ASP) per System in Indonesia
9.2.8 R&D Spend as % of Global Revenue
9.2.9 Number of Key Hospital Accounts in Indonesia
9.2.10 Payback Period for Providers (Years) & Typical ROI (%)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Vasomedical, Inc.
9.5.2 PSK-Health (PSK International)
9.5.3 Guangzhou Yinghui Medical Equipment Co., Ltd.
9.5.4 Chongqing PSK-Health Sci-Tech Development Co., Ltd.
9.5.5 NCP International (NeoCardial External Counterpulsation)
9.5.6 Advanced Cardiac Therapeutics Pte. Ltd. (Regional Distributor)
9.5.7 Medtronic plc
9.5.8 Abbott Laboratories
9.5.9 Boston Scientific Corporation
9.5.10 GE Healthcare
9.5.11 Philips Healthcare
9.5.12 Siemens Healthineers
9.5.13 Terumo Corporation
9.5.14 Local Indonesian Distributors (e.g., PT Enseval Putera Megatrading Tbk, PT Kimia Farma Tbk – medical devices division)
9.5.15 Other Emerging Regional EECP Vendors
10. Indonesia External Counterpulsation Devices Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation for Medical Devices
10.1.2 Decision-Making Process
10.1.3 Supplier Selection Criteria
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Healthcare Facilities
10.2.2 Spending on Medical Equipment
10.2.3 Budget for Training and Development
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges Faced by Hospitals
10.3.2 Issues in Rehabilitation Centers
10.3.3 Concerns in Home Care Settings
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training Needs
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of ROI
10.5.2 Expansion of Use Cases
10.5.3 Feedback Mechanisms
10.5.4 Others
11. Indonesia External Counterpulsation Devices Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from health organizations and medical device associations
- Review of government publications and healthcare regulations specific to external counterpulsation devices
- Examination of market trends and forecasts from reputable market research firms
Primary Research
- Interviews with cardiologists and healthcare professionals specializing in cardiovascular treatments
- Surveys with hospital procurement managers regarding purchasing decisions for medical devices
- Field interviews with medical device distributors and suppliers in Indonesia
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including sales data and clinical studies
- Triangulation of insights from primary interviews with secondary research findings
- Sanity checks conducted through expert panel reviews and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and cardiovascular disease prevalence
- Segmentation of the market by device type, application, and end-user demographics
- Incorporation of government health initiatives promoting non-invasive treatment options
Bottom-up Modeling
- Collection of sales data from leading manufacturers of external counterpulsation devices
- Estimation of market penetration rates based on hospital and clinic adoption rates
- Volume x price analysis to determine revenue potential across different market segments
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating demographic trends and healthcare access improvements
- Scenario modeling based on potential regulatory changes and technological advancements
- Development of baseline, optimistic, and pessimistic market growth projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiology Clinics | 100 | Cardiologists, Clinic Managers |
| Hospitals with Cardiac Units | 80 | Procurement Officers, Medical Directors |
| Medical Device Distributors | 60 | Sales Managers, Product Specialists |
| Health Insurance Providers | 50 | Policy Analysts, Claims Managers |
| Research Institutions | 40 | Research Scientists, Clinical Trial Coordinators |
Frequently Asked Questions
What is the current value of the Indonesia External Counterpulsation Devices Market?
The Indonesia External Counterpulsation Devices Market is valued at approximately USD 15 million, reflecting the growing adoption of non-invasive cardiac therapies in response to the increasing prevalence of cardiovascular diseases in the country.