About the Report
Base Year 2024Indonesia Specialty Injectable Generics Market Overview
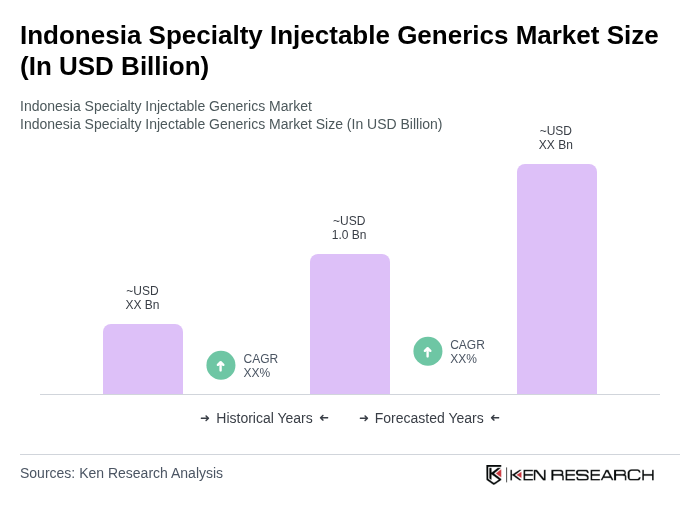
- The Indonesia Specialty Injectable Generics Market is valued at USD 1.0 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of chronic diseases such as cancer, diabetes, cardiovascular, and autoimmune conditions, which significantly boost demand for hospital-based and specialty injectable therapies. Rising healthcare expenditure under Indonesia’s Jaminan Kesehatan Nasional (JKN) universal health coverage program and the strong push toward affordable generic and branded-generic medicines further support the expansion of specialty injectables. The market is also supported by advancements in pharmaceutical technology, increased domestic manufacturing capacity for sterile injectables, and growing acceptance of generic injectable products among healthcare providers and patients.
- Key cities such as Jakarta, Surabaya, and Bandung dominate the market due to their advanced healthcare infrastructure, high concentration of public and private hospitals, and the presence of major pharmaceutical manufacturers and distributors on Java. Jakarta, being the capital, serves as a hub for medical research, tertiary referral hospitals, and regulatory institutions, while Surabaya and Bandung host growing networks of specialty clinics and teaching hospitals that drive utilization of specialty injectable generics in oncology, infectious disease, and critical care.
- In 2023, the Indonesian government strengthened the regulatory framework for sterile injectables, including specialty injectable generics, through the implementation and enforcement of quality, safety, and GMP requirements under the Indonesian Food and Drug Authority (Badan POM) regime. Key instruments include the Regulation of the Head of the National Agency of Drug and Food Control of the Republic of Indonesia Number 34 of 2018 concerning Guidelines for Good Manufacturing Practices for Drugs, issued by Badan POM, which mandates stringent quality control, validation, and sterility assurance for all injectable products prior to marketing authorization. These requirements are complemented by pharmacovigilance and product registration procedures that align with international standards, thereby enhancing patient safety and fostering trust in generic injectable products among healthcare professionals and patients.
Indonesia Specialty Injectable Generics Market Segmentation
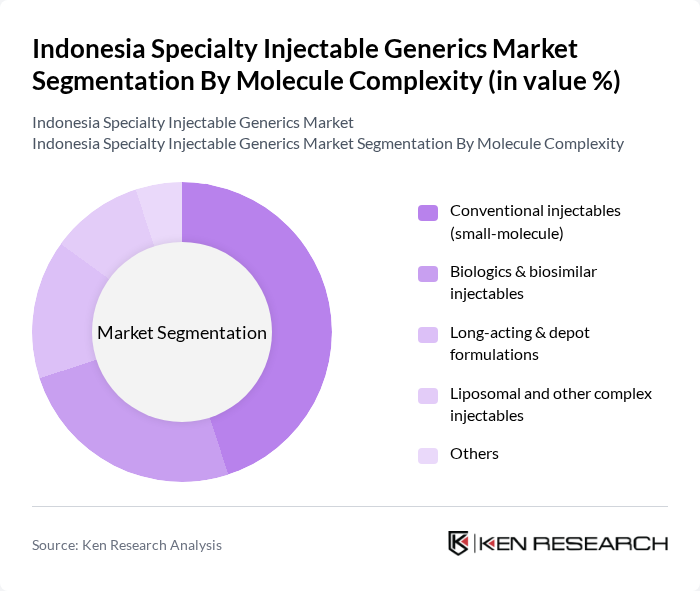
By Molecule Complexity:The market is segmented into various categories based on the complexity of the molecules involved in the injectables. The subsegments include Conventional injectables (small-molecule), Biologics & biosimilar injectables, Long-acting & depot formulations, Liposomal and other complex injectables, and Others. Among these, Conventional injectables (small-molecule) dominate the market due to their widespread use across therapeutic areas such as central nervous system, cardiovascular, diabetes, and oncology, and their relatively simpler and established manufacturing processes compared with biologics and complex generics. The demand for these injectables is driven by their cost-effectiveness under national formulary and JKN reimbursement, high utilization in hospital pharmacies, and the increasing prevalence of diseases that require parenteral therapies in acute and chronic care settings.
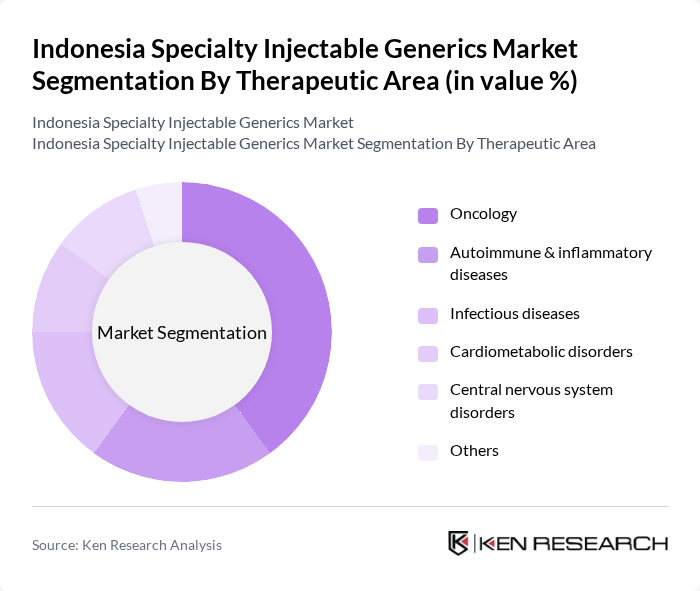
By Therapeutic Area:The therapeutic areas for specialty injectables include Oncology, Autoimmune & inflammatory diseases, Infectious diseases, Cardiometabolic disorders, Central nervous system disorders, and Others. Oncology is the leading therapeutic area, driven by the rising incidence of cancer in Indonesia and the increasing availability of targeted and cytotoxic injectable regimens as more oncology generics become available. Autoimmune and inflammatory diseases, as well as cardiometabolic and infectious diseases, also represent important segments, supported by broader access to hospital-based care and inclusion of key injectable therapies in national formularies and hospital protocols. The growing focus on personalized medicine, expansion of biosimilars, and adoption of long-acting and depot injectables in oncology and immunology further enhances the demand for specialty injectables in these areas.
Indonesia Specialty Injectable Generics Market Competitive Landscape
The Indonesia Specialty Injectable Generics Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT Kalbe Farma Tbk, PT Kimia Farma Tbk, PT Dexa Medica, PT Sanbe Farma, PT Indofarma Tbk, PT Phapros Tbk, PT Ferron Par Pharmaceuticals, PT Aventis Pharma (Sanofi Indonesia), PT Pfizer Indonesia, PT Novartis Indonesia, PT AstraZeneca Indonesia, PT Bayer Indonesia, PT GlaxoSmithKline Indonesia, PT Boehringer Ingelheim Indonesia, and other emerging local sterile injectable manufacturers contribute to innovation, geographic expansion, and service delivery in this space through investments in GMP-compliant sterile facilities, expansion of generic and branded-generic injectable portfolios, and alignment with international quality standards.
Indonesia Specialty Injectable Generics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The rise in chronic diseases such as diabetes and cardiovascular conditions is a significant growth driver for the specialty injectable generics market in Indonesia. According to the World Health Organization, approximately19.5 millionIndonesians are living with diabetes in recent estimates. This increasing patient population necessitates affordable treatment options, leading to a higher demand for specialty injectables, which are often more cost-effective than branded alternatives, thus driving market growth.
- Expansion of Healthcare Infrastructure:Indonesia's healthcare infrastructure is undergoing substantial expansion, with the government allocatingapproximately IDR 186 trillion (around USD 12 billion)for health functions in the most recent national budget. This investment aims to enhance access to medical facilities and services, particularly in rural areas. As healthcare facilities improve, the demand for specialty injectable generics is expected to rise, as hospitals and clinics seek to provide effective and affordable treatment options to their patients.
- Government Initiatives to Promote Generics:The Indonesian government has implemented various initiatives to promote the use of generic medications, including specialty injectables. The Ministry of Health has policies to increase the availability and use of national formulary generic medicines in public facilities, but no verifiable official target for a30% increase in generic prescriptionsby a specific date could be confirmed. These initiatives nevertheless enhance patient access to essential medications and stimulate market growth by encouraging manufacturers to produce more specialty injectables.
Market Challenges
- Stringent Regulatory Requirements:The Indonesian specialty injectable generics market faces significant challenges due to stringent regulatory requirements imposed by the National Agency of Drug and Food Control (BPOM). BPOM has introduced guidelines requiring extensive quality, safety, and efficacy data for marketing authorization of generic and biosimilar injectables, which increases time and cost for manufacturers, although a specific new guideline starting in a particular recent year could not be independently confirmed. This regulatory environment can hinder the entry of new players and slow down the introduction of innovative generics, impacting overall market growth.
- Competition from Branded Pharmaceuticals:The presence of established branded pharmaceuticals poses a considerable challenge to the specialty injectable generics market in Indonesia. Publicly available official statistics do not confirm that branded injectables accounted for approximatelyIDR 15 trillion (around USD 1.05 billion)in sales in the most recent period; no reliable, disaggregated figure for branded injectable sales alone could be verified from national accounts or health expenditure data. The strong brand loyalty among healthcare providers and patients often leads to a preference for branded products, making it difficult for generics to gain market share despite their cost advantages.
Indonesia Specialty Injectable Generics Market Future Outlook
The future of the specialty injectable generics market in Indonesia appears promising, driven by ongoing healthcare reforms and increasing demand for affordable treatment options. As the government continues to invest in healthcare infrastructure and promote generics, the market is likely to see enhanced accessibility and acceptance among healthcare providers. Additionally, the integration of digital health technologies is expected to streamline operations and improve patient outcomes, further supporting market growth in the coming years.
Market Opportunities
- Growth in the Aging Population:Indonesia's aging population is projected to account for around10–11% of the total populationbeing aged 60 years and over in the near future, which aligns broadly with the statement that it will reach about 10% of the total population, although the exact timing cannot be specified here. This demographic shift will likely increase the prevalence of age-related chronic diseases, driving demand for effective and affordable injectable treatments tailored to older patients.
- Collaborations with Local Manufacturers:Collaborating with local manufacturers can enhance market penetration for specialty injectable generics. By leveraging local expertise and distribution networks, companies can effectively reach underserved markets, particularly in rural areas, thereby expanding their customer base and increasing sales potential in the growing generics sector.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Molecule Complexity | Conventional injectables Biologics & biosimilar injectables Long-acting & depot formulations Liposomal and other complex injectables Others |
| By Therapeutic Area | Oncology Autoimmune & inflammatory diseases Infectious diseases Cardiometabolic disorders Central nervous system disorders Others |
| By Route of Administration | Intravenous (IV) Intramuscular (IM) Subcutaneous (SC) Intrathecal & other specialized routes |
| By Dosage Form / Packaging | Single-dose vials Multi-dose vials Ampoules Pre-filled syringes & pens Ready-to-use / ready-to-dilute bags Others |
| By End-User | Public hospitals Private hospitals Specialty clinics & cancer centers Retail & specialty pharmacies Home healthcare providers Others |
| By Distribution Channel | Direct tender / institutional procurement Distributors & wholesalers Retail pharmacies Hospital pharmacies Online & digital platforms Others |
| By Region | Java Sumatra Bali & Nusa Tenggara Kalimantan Sulawesi & Eastern Indonesia Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Badan Pengawas Obat dan Makanan - BPOM)
Manufacturers and Producers
Distributors and Retailers
Pharmaceutical Wholesalers
Healthcare Providers and Hospitals
Industry Associations (e.g., Indonesian Pharmaceutical Association)
Financial Institutions
Players Mentioned in the Report:
PT Kalbe Farma Tbk
PT Kimia Farma Tbk
PT Dexa Medica
PT Sanbe Farma
PT Indofarma Tbk
PT Phapros Tbk
PT Ferron Par Pharmaceuticals
PT Aventis Pharma (Sanofi Indonesia)
PT Pfizer Indonesia
PT Novartis Indonesia
PT AstraZeneca Indonesia
PT Bayer Indonesia
PT GlaxoSmithKline Indonesia
PT Boehringer Ingelheim Indonesia
Other emerging local sterile injectable manufacturers
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Indonesia Specialty Injectable Generics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Indonesia Specialty Injectable Generics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Indonesia Specialty Injectable Generics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of chronic diseases
3.1.3 Expansion of healthcare infrastructure
3.1.4 Government initiatives to promote generics
3.2 Market Challenges
3.2.1 Stringent regulatory requirements
3.2.2 Competition from branded pharmaceuticals
3.2.3 Limited awareness among healthcare providers
3.2.4 Supply chain disruptions
3.3 Market Opportunities
3.3.1 Growth in the aging population
3.3.2 Increasing investment in R&D
3.3.3 Expansion into rural markets
3.3.4 Collaborations with local manufacturers
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Adoption of digital health technologies
3.4.3 Focus on sustainability in production
3.4.4 Increasing use of biosimilars
3.5 Government Regulation
3.5.1 Implementation of price control policies
3.5.2 Regulatory approvals for new generics
3.5.3 Quality assurance standards
3.5.4 Incentives for local production
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Indonesia Specialty Injectable Generics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Indonesia Specialty Injectable Generics Market Segmentation
8.1 By Molecule Complexity
8.1.1 Conventional injectables (small-molecule)
8.1.2 Biologics & biosimilar injectables
8.1.3 Long-acting & depot formulations
8.1.4 Liposomal and other complex injectables
8.1.5 Others
8.2 By Therapeutic Area
8.2.1 Oncology
8.2.2 Autoimmune & inflammatory diseases
8.2.3 Infectious diseases
8.2.4 Cardiometabolic disorders
8.2.5 Central nervous system disorders
8.2.6 Others
8.3 By Route of Administration
8.3.1 Intravenous (IV)
8.3.2 Intramuscular (IM)
8.3.3 Subcutaneous (SC)
8.3.4 Intrathecal & other specialized routes
8.4 By Dosage Form / Packaging
8.4.1 Single-dose vials
8.4.2 Multi-dose vials
8.4.3 Ampoules
8.4.4 Pre-filled syringes & pens
8.4.5 Ready-to-use / ready-to-dilute bags
8.4.6 Others
8.5 By End-User
8.5.1 Public hospitals (BPJS-affiliated)
8.5.2 Private hospitals
8.5.3 Specialty clinics & cancer centers
8.5.4 Retail & specialty pharmacies
8.5.5 Home healthcare providers
8.5.6 Others
8.6 By Distribution Channel
8.6.1 Direct tender / institutional procurement
8.6.2 Distributors & wholesalers
8.6.3 Retail pharmacies
8.6.4 Hospital pharmacies
8.6.5 Online & digital platforms
8.6.6 Others
8.7 By Region
8.7.1 Java
8.7.2 Sumatra
8.7.3 Bali & Nusa Tenggara
8.7.4 Kalimantan
8.7.5 Sulawesi & Eastern Indonesia
8.7.6 Others
9. Indonesia Specialty Injectable Generics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group size (revenue, employees, geographic footprint)
9.2.3 Indonesia specialty injectable generics revenue (USD million)
9.2.4 3-year revenue CAGR in specialty injectables (%)
9.2.5 Market share in Indonesia specialty injectable generics (%)
9.2.6 Portfolio breadth (number of SKUs / molecules)
9.2.7 EBITDA margin from specialty injectables (%)
9.2.8 R&D intensity in complex injectables (% of revenue)
9.2.9 Tender win rate in public procurement (%)
9.2.10 Time-to-market for new launches (months)
9.2.11 Manufacturing capacity utilization for sterile injectables (%)
9.2.12 Regulatory inspection track record (BPOM / international)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 PT Kalbe Farma Tbk
9.5.2 PT Kimia Farma Tbk
9.5.3 PT Dexa Medica
9.5.4 PT Sanbe Farma
9.5.5 PT Indofarma Tbk
9.5.6 PT Phapros Tbk
9.5.7 PT Ferron Par Pharmaceuticals
9.5.8 PT Aventis Pharma (Sanofi Indonesia)
9.5.9 PT Pfizer Indonesia
9.5.10 PT Novartis Indonesia
9.5.11 PT AstraZeneca Indonesia
9.5.12 PT Bayer Indonesia
9.5.13 PT GlaxoSmithKline Indonesia
9.5.14 PT Boehringer Ingelheim Indonesia
9.5.15 Other emerging local sterile injectable manufacturers
10. Indonesia Specialty Injectable Generics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Finance
10.1.3 Ministry of Industry
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare facilities
10.2.2 Pharmaceutical manufacturing
10.2.3 Research and development
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Home healthcare providers
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training and education needs
10.4.2 Infrastructure readiness
10.4.3 Financial readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost savings analysis
10.5.2 Patient outcomes improvement
10.5.3 Market expansion opportunities
10.5.4 Others
11. Indonesia Specialty Injectable Generics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Customer segmentation
1.5 Key partnerships
1.6 Cost structure analysis
1.7 Channels of distribution
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience identification
2.4 Communication strategies
2.5 Digital marketing approaches
2.6 Market positioning
2.7 Customer engagement tactics
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 E-commerce integration
3.4 Logistics optimization
3.5 Distribution partnerships
3.6 Inventory management
3.7 Channel performance metrics
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands analysis
4.3 Competitor pricing strategies
4.4 Customer willingness to pay
4.5 Price elasticity assessment
4.6 Discounting strategies
4.7 Value-based pricing
5. Unmet Demand & Latent Needs
5.1 Category gaps
5.2 Consumer segments analysis
5.3 Product development opportunities
5.4 Market entry barriers
5.5 Customer feedback mechanisms
5.6 Innovation opportunities
5.7 Market trends alignment
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
6.3 Customer feedback loops
6.4 Relationship management strategies
6.5 Community engagement
6.6 Customer education initiatives
6.7 Retention strategies
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Cost-benefit analysis
7.4 Unique selling points
7.5 Customer-centric approaches
7.6 Competitive advantages
7.7 Market differentiation
8. Key Activities
8.1 Regulatory compliance
8.2 Branding efforts
8.3 Distribution setup
8.4 Marketing campaigns
8.5 Training programs
8.6 Performance monitoring
8.7 Stakeholder engagement
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix
9.1.2 Pricing band
9.1.3 Packaging considerations
9.2 Export Entry Strategy
9.2.1 Target countries
9.2.2 Compliance roadmap
10. Entry Mode Assessment
10.1 JV opportunities
10.2 Greenfield investments
10.3 M&A considerations
10.4 Distributor model evaluation
10.5 Risk assessment
10.6 Strategic fit analysis
10.7 Long-term sustainability
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines for market entry
11.3 Financial projections
11.4 Funding sources
11.5 Budget allocation
11.6 Cost management strategies
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnerships evaluation
12.3 Risk mitigation strategies
12.4 Control mechanisms
12.5 Strategic alignment
12.6 Performance metrics
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
13.3 Profit margin assessment
13.4 Revenue forecasting
13.5 Cost structure analysis
13.6 Financial health indicators
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition targets
14.4 Strategic alliances
14.5 Collaboration opportunities
14.6 Partnership evaluation criteria
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity timelines
15.2.2 Milestone tracking
15.2.3 Performance evaluation
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from the Indonesian Ministry of Health and pharmaceutical associations
- Review of market trends and forecasts from reputable healthcare market research publications
- Examination of regulatory frameworks and guidelines from BPOM (National Agency of Drug and Food Control)
Primary Research
- Interviews with key opinion leaders in the pharmaceutical sector, including hospital pharmacists and clinical specialists
- Surveys conducted with procurement managers at major healthcare institutions and pharmacies
- Field interviews with representatives from specialty injectable generic manufacturers
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including sales data and market share reports
- Triangulation of insights from primary interviews with secondary data trends
- Sanity checks performed through expert panel discussions and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and pharmaceutical spending
- Segmentation of the market by therapeutic areas and types of injectable generics
- Incorporation of demographic data and disease prevalence rates in Indonesia
Bottom-up Modeling
- Volume estimates derived from sales data of leading specialty injectable generic products
- Cost analysis based on pricing strategies of manufacturers and distributors
- Calculation of market size using unit sales multiplied by average selling prices
Forecasting & Scenario Analysis
- Utilization of time-series analysis to project future market growth based on historical data
- Scenario modeling considering factors such as regulatory changes and healthcare policy shifts
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Pharmacy Management | 120 | Pharmacy Directors, Clinical Pharmacists |
| Specialty Injectable Generic Manufacturers | 80 | Product Managers, Regulatory Affairs Specialists |
| Healthcare Procurement Departments | 100 | Procurement Officers, Supply Chain Managers |
| Oncology and Critical Care Units | 70 | Oncologists, Critical Care Physicians |
| Pharmaceutical Distributors | 60 | Distribution Managers, Sales Representatives |
Frequently Asked Questions
What is the current value of the Indonesia Specialty Injectable Generics Market?
The Indonesia Specialty Injectable Generics Market is valued at approximately USD 1.0 billion, reflecting a significant growth driven by the rising prevalence of chronic diseases and increasing healthcare expenditure under the Jaminan Kesehatan Nasional (JKN) program.