About the Report
Base Year 2024Indonesia Surgical Stapling Market Overview
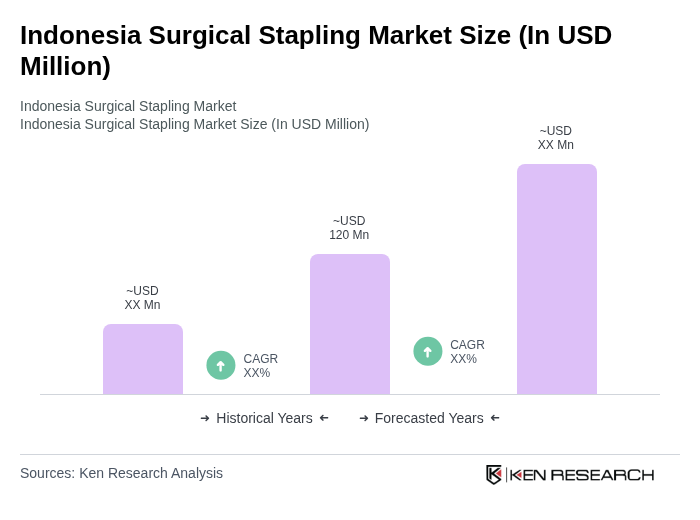
- The Indonesia Surgical Stapling Market is valued at USD 120 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of surgical procedures, advancements in surgical technologies, and a rising demand for minimally invasive surgeries. The healthcare sector's expansion, coupled with government initiatives to improve healthcare infrastructure, has further fueled the market's growth. Notably, the Asia-Pacific region is experiencing robust adoption of advanced stapling devices, with Indonesia contributing significantly due to its expanding hospital network and rising surgical volumes .
- Key cities such as Jakarta, Surabaya, and Bandung dominate the market due to their advanced healthcare facilities and higher patient footfall. Jakarta, being the capital, has a concentration of hospitals and surgical centers equipped with modern technologies, while Surabaya and Bandung are emerging as significant healthcare hubs, attracting investments and skilled professionals .
- The regulation of surgical stapling devices in Indonesia is governed by the “Regulation of the Minister of Health of the Republic of Indonesia Number 62 of 2017 concerning Marketing Authorization for Medical Devices, In Vitro Diagnostic Medical Devices and Household Health Supplies,” issued by the Ministry of Health. This regulation mandates that all surgical stapling devices used in public and private healthcare facilities must obtain marketing authorization, comply with safety and efficacy standards, and be registered with the Ministry of Health before distribution and use in hospitals. The regulation ensures standardized practices and the adoption of advanced medical devices to improve surgical outcomes and patient safety .
Indonesia Surgical Stapling Market Segmentation
By Technology:
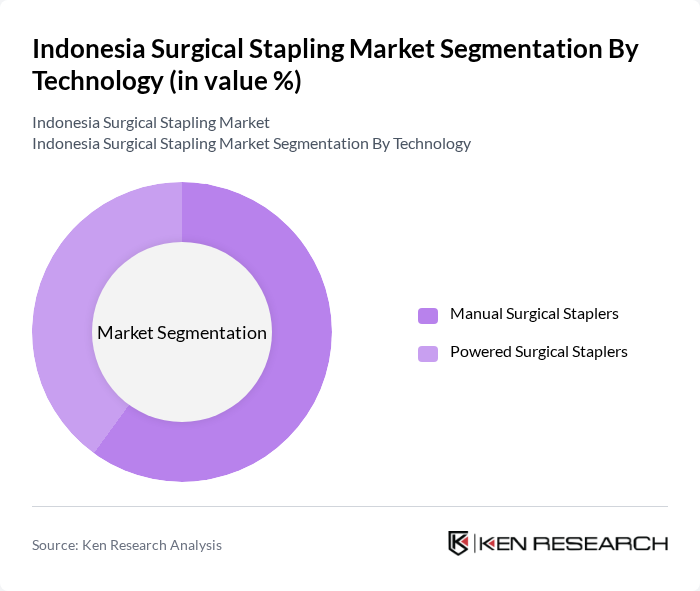
The market is segmented into two primary technologies: Manual Surgical Staplers and Powered Surgical Staplers. Manual surgical staplers are widely used due to their cost-effectiveness and ease of use, particularly in low-resource settings. However, powered surgical staplers are gaining traction due to their precision and efficiency, especially in complex surgeries. The increasing trend towards minimally invasive procedures is driving the demand for powered staplers, which offer enhanced performance and reduced surgical time. While manual staplers currently hold a larger share in the Asia-Pacific region, powered staplers are rapidly increasing in adoption as hospitals modernize their surgical suites .
By Product Type:
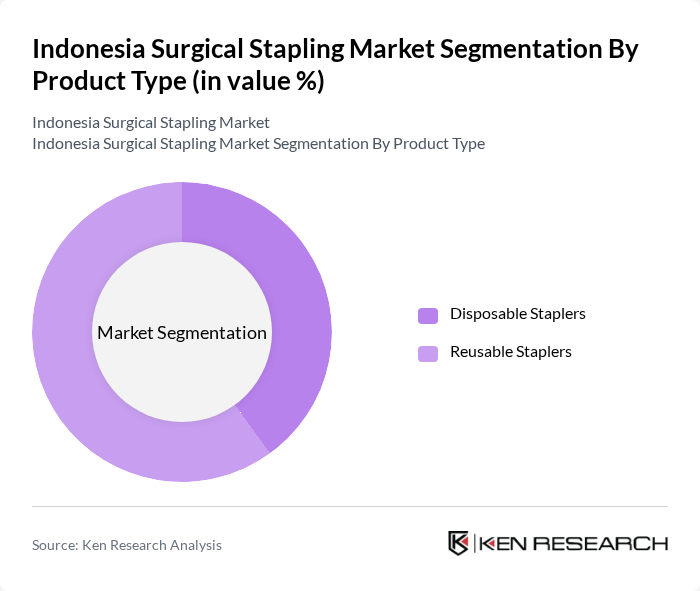
The product type segmentation includes Disposable Staplers and Reusable Staplers. Disposable staplers are preferred in many surgical settings due to their convenience and reduced risk of cross-contamination. However, reusable staplers are favored in hospitals aiming to reduce costs over time. The growing emphasis on sustainability and cost-effectiveness is leading to a balanced demand for both types, with reusable staplers gaining market share as hospitals focus on long-term operational efficiency. In the Asia-Pacific region, reusable staplers currently account for the majority of the market, reflecting a shift towards cost-effective, multi-use devices .
Indonesia Surgical Stapling Market Competitive Landscape
The Indonesia Surgical Stapling Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Johnson & Johnson (Ethicon), B. Braun Melsungen AG, ConMed Corporation, Stryker Corporation, 3M Company, Intuitive Surgical, Inc., Olympus Corporation, Zimmer Biomet Holdings, Inc., Cook Medical LLC, Medline Industries, LP, Smith & Nephew plc, Baxter International Inc., Terumo Corporation, PT Enseval Putera Megatrading Tbk, PT Phapros Tbk, PT Kimia Farma Tbk contribute to innovation, geographic expansion, and service delivery in this space.
Indonesia Surgical Stapling Market Industry Analysis
Growth Drivers
- Increasing Surgical Procedures:The number of surgical procedures in Indonesia has been on the rise, with over 1.6 million surgeries performed annually as of future. This increase is driven by a growing population and improved access to healthcare facilities. The Indonesian government has invested approximately $1.2 billion in healthcare infrastructure, enhancing surgical capabilities. As surgical volumes increase, the demand for surgical stapling devices is expected to grow significantly, supporting market expansion.
- Rising Prevalence of Chronic Diseases:Chronic diseases such as diabetes and cardiovascular conditions are becoming more prevalent in Indonesia, affecting over 11 million people as of future. This trend necessitates more surgical interventions, thereby increasing the demand for surgical stapling devices. The World Health Organization reported that chronic diseases account for 60% of all deaths in Indonesia, highlighting the urgent need for effective surgical solutions to manage these conditions.
- Technological Advancements in Surgical Stapling:The surgical stapling market is witnessing rapid technological advancements, with innovations such as robotic-assisted stapling devices and smart staplers. In future, the adoption of these technologies increased by 30% in major hospitals across Indonesia. These advancements not only improve surgical outcomes but also enhance efficiency, driving the demand for modern stapling solutions in surgical procedures, thus propelling market growth.
Market Challenges
- High Cost of Surgical Stapling Devices:The high cost of advanced surgical stapling devices poses a significant challenge in Indonesia, where healthcare budgets are often constrained. Prices for premium stapling devices can exceed $1,000 per unit, making them less accessible for smaller healthcare facilities. This financial barrier limits the widespread adoption of these devices, hindering overall market growth and innovation in surgical practices.
- Limited Awareness Among Healthcare Professionals:There is a notable lack of awareness and training regarding advanced surgical stapling techniques among healthcare professionals in Indonesia. A survey conducted in future indicated that only 40% of surgeons were familiar with the latest stapling technologies. This knowledge gap can lead to underutilization of effective surgical stapling devices, impacting patient outcomes and limiting market penetration for manufacturers.
Indonesia Surgical Stapling Market Future Outlook
The future of the surgical stapling market in Indonesia appears promising, driven by ongoing advancements in medical technology and increasing healthcare investments. As the government continues to enhance healthcare infrastructure, the accessibility of surgical procedures will improve. Additionally, the rising geriatric population, projected to reach 27 million in future, will further fuel demand for surgical interventions, creating a favorable environment for market growth and innovation in surgical stapling solutions.
Market Opportunities
- Expansion of Healthcare Infrastructure:The Indonesian government plans to invest $2.5 billion in healthcare infrastructure by future, which will enhance surgical facilities and increase the demand for surgical stapling devices. This investment will create opportunities for manufacturers to introduce advanced products and expand their market presence in underserved regions.
- Potential for Product Innovation:There is significant potential for product innovation in the surgical stapling market, particularly in developing bioabsorbable staples. With the increasing focus on patient safety and outcomes, manufacturers can capitalize on this trend by investing in research and development to create safer, more effective stapling solutions that meet evolving healthcare needs.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Technology | Manual Surgical Staplers Powered Surgical Staplers |
| By Product Type | Disposable Staplers Reusable Staplers |
| By Stapler Design | Linear Staplers Circular Staplers Skin Staplers Endoscopic Staplers Specialty Staplers |
| By Application | General Surgery Gastrointestinal Surgery Cardiothoracic Surgery Gynecological Surgery Urological Surgery Others |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Clinics Others |
| By Material | Stainless Steel Polymer Bioabsorbable Materials Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | Java Sumatra Bali Kalimantan Sulawesi Others |
| By Price Range | Low Price Mid Price High Price Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, National Agency of Drug and Food Control)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Wholesalers
Industry Associations
Financial Institutions
Players Mentioned in the Report:
Medtronic plc
Johnson & Johnson (Ethicon)
B. Braun Melsungen AG
ConMed Corporation
Stryker Corporation
3M Company
Intuitive Surgical, Inc.
Olympus Corporation
Zimmer Biomet Holdings, Inc.
Cook Medical LLC
Medline Industries, LP
Smith & Nephew plc
Baxter International Inc.
Terumo Corporation
PT Enseval Putera Megatrading Tbk
PT Phapros Tbk
PT Kimia Farma Tbk
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Indonesia Surgical Stapling Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Indonesia Surgical Stapling Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Indonesia Surgical Stapling Market Analysis
3.1 Growth Drivers
3.1.1 Increasing surgical procedures
3.1.2 Rising prevalence of chronic diseases
3.1.3 Technological advancements in surgical stapling
3.1.4 Growing demand for minimally invasive surgeries
3.2 Market Challenges
3.2.1 High cost of surgical stapling devices
3.2.2 Limited awareness among healthcare professionals
3.2.3 Stringent regulatory requirements
3.2.4 Competition from alternative wound closure methods
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Increasing investment in medical technology
3.3.3 Rising geriatric population
3.3.4 Potential for product innovation
3.4 Market Trends
3.4.1 Shift towards automated surgical stapling devices
3.4.2 Growing preference for bioabsorbable staples
3.4.3 Integration of digital technologies in surgical procedures
3.4.4 Focus on patient safety and outcomes
3.5 Government Regulation
3.5.1 Compliance with Indonesian Ministry of Health standards
3.5.2 Registration requirements for medical devices
3.5.3 Import regulations for surgical stapling products
3.5.4 Guidelines for clinical trials and product testing
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Indonesia Surgical Stapling Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Indonesia Surgical Stapling Market Segmentation
8.1 By Technology
8.1.1 Manual Surgical Staplers
8.1.2 Powered Surgical Staplers
8.2 By Product Type
8.2.1 Disposable Staplers
8.2.2 Reusable Staplers
8.3 By Stapler Design
8.3.1 Linear Staplers
8.3.2 Circular Staplers
8.3.3 Skin Staplers
8.3.4 Endoscopic Staplers
8.3.5 Specialty Staplers
8.4 By Application
8.4.1 General Surgery
8.4.2 Gastrointestinal Surgery
8.4.3 Cardiothoracic Surgery
8.4.4 Gynecological Surgery
8.4.5 Urological Surgery
8.4.6 Others
8.5 By End-User
8.5.1 Hospitals
8.5.2 Ambulatory Surgical Centers
8.5.3 Specialty Clinics
8.5.4 Others
8.6 By Material
8.6.1 Stainless Steel
8.6.2 Polymer
8.6.3 Bioabsorbable Materials
8.6.4 Others
8.7 By Distribution Channel
8.7.1 Direct Sales
8.7.2 Distributors
8.7.3 Online Sales
8.7.4 Others
8.8 By Region
8.8.1 Java
8.8.2 Sumatra
8.8.3 Bali
8.8.4 Kalimantan
8.8.5 Sulawesi
8.8.6 Others
8.9 By Price Range
8.9.1 Low Price
8.9.2 Mid Price
8.9.3 High Price
8.9.4 Others
9. Indonesia Surgical Stapling Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Indonesia Market Revenue (USD, latest year)
9.2.4 Revenue Growth Rate (Indonesia, 3-year CAGR)
9.2.5 Market Share (%)
9.2.6 Product Portfolio Breadth (Number of Stapler SKUs/Types)
9.2.7 Regulatory Approvals (Number of MOH-registered products)
9.2.8 Distribution Network Coverage (Number of provinces/cities served)
9.2.9 Local Partnerships/Distributors (Number and quality)
9.2.10 Pricing Competitiveness (Indexed to market average)
9.2.11 Customer Satisfaction Score (Indonesia, latest survey)
9.2.12 After-Sales Service Capability (Response time, service points)
9.2.13 Product Innovation Rate (New launches in past 3 years)
9.2.14 Brand Recognition (Indonesia, aided/unaided awareness)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Medtronic plc
9.5.2 Johnson & Johnson (Ethicon)
9.5.3 B. Braun Melsungen AG
9.5.4 ConMed Corporation
9.5.5 Stryker Corporation
9.5.6 3M Company
9.5.7 Intuitive Surgical, Inc.
9.5.8 Olympus Corporation
9.5.9 Zimmer Biomet Holdings, Inc.
9.5.10 Cook Medical LLC
9.5.11 Medline Industries, LP
9.5.12 Smith & Nephew plc
9.5.13 Baxter International Inc.
9.5.14 Terumo Corporation
9.5.15 PT Enseval Putera Megatrading Tbk
9.5.16 PT Phapros Tbk
9.5.17 PT Kimia Farma Tbk
10. Indonesia Surgical Stapling Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation for Surgical Supplies
10.1.2 Decision-Making Process
10.1.3 Supplier Selection Criteria
10.1.4 Contract Management Practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Surgical Facilities
10.2.2 Spending on Medical Equipment
10.2.3 Budget for Training and Development
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Supply Chain Disruptions
10.3.2 Quality Assurance Issues
10.3.3 Cost Management Challenges
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training Needs Assessment
10.4.2 Technology Acceptance Levels
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Surgical Outcomes
10.5.2 Cost Savings Analysis
10.5.3 User Feedback Mechanisms
10.5.4 Others
11. Indonesia Surgical Stapling Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Evaluation
1.5 Key Partnerships Exploration
1.6 Customer Segmentation
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Market Identification
2.4 Communication Strategy
2.5 Digital Marketing Approaches
2.6 Trade Show Participation
2.7 Customer Engagement Tactics
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-Ups
3.3 E-commerce Integration
3.4 Logistics and Supply Chain Management
3.5 Distribution Partnerships
3.6 Inventory Management Solutions
3.7 Performance Metrics for Distribution
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
4.4 Customer Willingness to Pay
4.5 Price Sensitivity Assessment
4.6 Discounting Strategies
4.7 Pricing Model Innovation
5. Unmet Demand & Latent Needs
5.1 Category Gaps Identification
5.2 Consumer Segments Analysis
5.3 Product Development Opportunities
5.4 Market Entry Barriers
5.5 Customer Feedback Collection
5.6 Future Trends Anticipation
5.7 Strategic Recommendations
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Support Systems
6.4 Feedback Mechanisms
6.5 Relationship Management Tools
6.6 Community Engagement Initiatives
6.7 Customer Retention Strategies
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Competitive Advantage Analysis
7.4 Customer-Centric Innovations
7.5 Value-Added Services
7.6 Market Differentiation Strategies
7.7 Long-term Value Creation
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
8.4 Training and Development Programs
8.5 Market Research Activities
8.6 Stakeholder Engagement
8.7 Performance Monitoring
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging Considerations
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
10.5 Risk Assessment
10.6 Strategic Fit Evaluation
10.7 Exit Strategy Considerations
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
11.3 Funding Sources
11.4 Financial Projections
11.5 Budget Allocation
11.6 Cost-Benefit Analysis
11.7 Risk Mitigation Strategies
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
12.2 Risk Assessment Framework
12.3 Control Mechanisms
12.4 Partnership Evaluation Criteria
12.5 Long-term Strategic Goals
12.6 Flexibility in Operations
12.7 Exit Strategy Planning
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
13.3 Profit Margin Projections
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from healthcare associations and medical device regulatory bodies in Indonesia
- Review of published market studies and white papers on surgical stapling technologies
- Examination of government health expenditure reports and surgical procedure statistics
Primary Research
- Interviews with surgeons and healthcare professionals involved in surgical procedures
- Surveys targeting hospital procurement managers and medical device distributors
- Field visits to hospitals and surgical centers to observe stapling device usage
Validation & Triangulation
- Cross-validation of data from multiple sources including sales data and clinical usage reports
- Triangulation of findings from primary interviews with secondary research insights
- Sanity checks through expert panels comprising industry veterans and medical professionals
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total healthcare expenditure in Indonesia and its allocation to surgical procedures
- Analysis of the growth rate of surgical procedures requiring stapling devices
- Incorporation of demographic trends and surgical volume forecasts
Bottom-up Modeling
- Collection of sales data from leading surgical stapling device manufacturers operating in Indonesia
- Estimation of average selling prices and market penetration rates for various stapling products
- Volume estimates based on surgical procedure frequency and device usage rates
Forecasting & Scenario Analysis
- Multi-variable forecasting model incorporating factors such as population growth and healthcare access
- Scenario analysis based on potential regulatory changes and technological advancements in surgical devices
- Development of baseline, optimistic, and pessimistic market growth projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| General Surgery Departments | 100 | Surgeons, Department Heads |
| Orthopedic Surgery Units | 60 | Orthopedic Surgeons, Surgical Assistants |
| Gastrointestinal Surgery Clinics | 50 | Gastrointestinal Surgeons, Surgical Nurses |
| Cardiothoracic Surgery Centers | 40 | Cardiothoracic Surgeons, Anesthesiologists |
| Healthcare Procurement Departments | 70 | Procurement Managers, Supply Chain Directors |
Frequently Asked Questions
What is the current value of the Indonesia Surgical Stapling Market?
The Indonesia Surgical Stapling Market is valued at approximately USD 120 million, reflecting a significant growth driven by the increasing prevalence of surgical procedures and advancements in surgical technologies.