Japan Alpha 1 Antitrypsin Deficiency Disease Market Overview
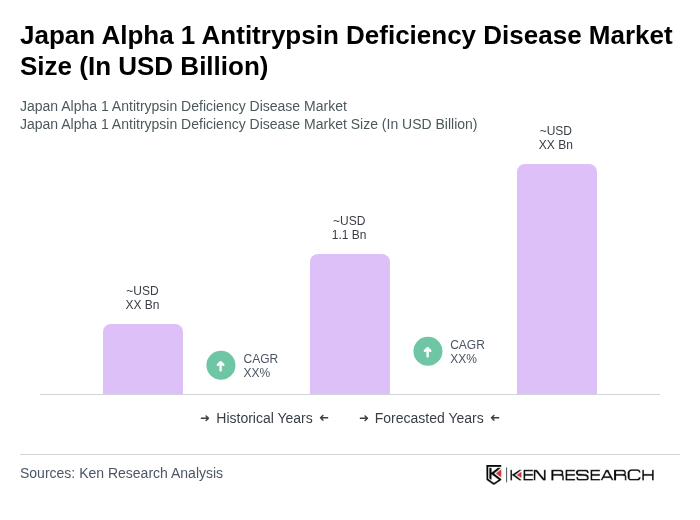
- The Japan Alpha 1 Antitrypsin Deficiency Disease Market is valued at USD 1.1 billion, based on a five-year historical analysis. This growth is primarily driven by increasing awareness of the disease, advancements in treatment options, and a growing patient population. The rising prevalence of respiratory diseases and liver complications associated with Alpha 1 Antitrypsin Deficiency has further fueled demand for effective therapies and healthcare services.
- Key cities such as Tokyo, Osaka, and Kyoto dominate the market due to their advanced healthcare infrastructure, high concentration of medical research institutions, and access to specialized healthcare services. These urban centers are also home to a significant number of healthcare professionals and patients, facilitating better diagnosis and treatment options for Alpha 1 Antitrypsin Deficiency.
- The Japanese government has implemented regulatory frameworks aimed at improving the diagnosis and treatment of Alpha 1 Antitrypsin Deficiency. These regulations mandate that healthcare providers incorporate screening for the deficiency as part of routine respiratory health assessments, ensuring early detection and timely intervention for affected individuals.
Japan Alpha 1 Antitrypsin Deficiency Disease Market Segmentation
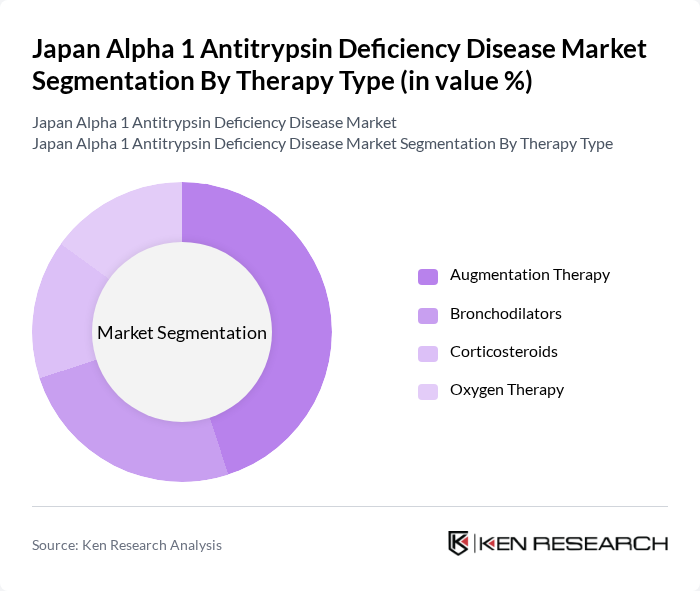
By Therapy Type:The market is segmented into various therapy types, including Augmentation Therapy, Bronchodilators, Corticosteroids, and Oxygen Therapy. Among these, Augmentation Therapy is the leading subsegment due to its effectiveness in replenishing the deficient protein in patients, thereby improving lung function and quality of life. The increasing adoption of this therapy, driven by clinical evidence and patient outcomes, has solidified its position as the preferred treatment option.
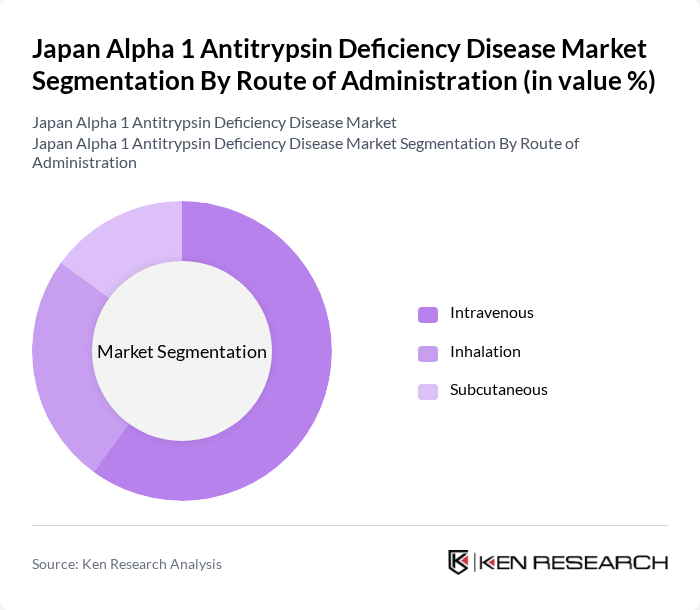
By Route of Administration:The market is categorized based on the route of administration, including Intravenous, Inhalation, and Subcutaneous methods. Intravenous administration is the dominant route due to its rapid onset of action and effectiveness in delivering therapies directly into the bloodstream. This method is particularly favored in clinical settings for patients requiring immediate treatment, thus driving its market share significantly.
Japan Alpha 1 Antitrypsin Deficiency Disease Market Competitive Landscape
The Japan Alpha 1 Antitrypsin Deficiency Disease Market is characterized by a dynamic mix of regional and international players. Leading participants such as Takeda Pharmaceutical Company Limited, Chugai Pharmaceutical Co., Ltd., Mitsubishi Tanabe Pharma Corporation, Daiichi Sankyo Company, Limited, Astellas Pharma Inc., Otsuka Pharmaceutical Co., Ltd., Eisai Co., Ltd., Kyowa Kirin Co., Ltd., Shionogi & Co., Ltd., Sumitomo Dainippon Pharma Co., Ltd., Amgen Inc., Grifols S.A., CSL Behring, Biogen Inc., Vertex Pharmaceuticals Incorporated contribute to innovation, geographic expansion, and service delivery in this space.
Japan Alpha 1 Antitrypsin Deficiency Disease Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Alpha 1 Antitrypsin Deficiency:The prevalence of Alpha 1 Antitrypsin Deficiency (AATD) in Japan is estimated to be around 1 in 2,500 individuals, translating to approximately 52,000 affected people. This rising incidence is attributed to genetic factors and environmental influences, leading to a growing patient population. The increasing number of diagnosed cases is expected to drive demand for diagnostic and therapeutic solutions, thereby expanding the market significantly in the coming years.
- Advancements in Diagnostic Technologies:The Japanese healthcare sector has seen significant advancements in diagnostic technologies, including genetic testing and biomarker identification. In future, the market for diagnostic tools is projected to reach ¥30 billion, reflecting a 15% increase from the prior period. These innovations facilitate early detection of AATD, allowing for timely intervention and management, which is crucial for improving patient outcomes and driving market growth.
- Enhanced Treatment Options and Therapies:The introduction of new therapies, including enzyme replacement therapies and novel medications, has transformed the treatment landscape for AATD. In future, the total expenditure on AATD treatments in Japan is expected to exceed ¥40 billion, driven by the availability of more effective and personalized treatment options. This expansion in therapeutic offerings is likely to enhance patient adherence and improve overall health outcomes, further propelling market growth.
Market Challenges
- High Cost of Treatment:The financial burden of AATD treatment remains a significant challenge, with annual treatment costs averaging ¥10 million per patient. This high cost can limit access to necessary therapies, particularly for patients without adequate insurance coverage. As a result, many patients may delay or forgo treatment, adversely affecting their health and complicating the overall management of the disease, which poses a challenge to market growth.
- Limited Availability of Specialized Healthcare Providers:In Japan, there is a shortage of healthcare providers specializing in AATD, with only about 200 specialists available nationwide. This scarcity can lead to delays in diagnosis and treatment, negatively impacting patient outcomes. The lack of specialized care also hinders the development of comprehensive treatment plans, which is essential for managing this complex disease effectively, thus posing a challenge to market expansion.
Japan Alpha 1 Antitrypsin Deficiency Disease Market Future Outlook
The future of the AATD market in Japan appears promising, driven by ongoing advancements in personalized medicine and increased funding for rare disease research. As healthcare providers adopt more tailored treatment approaches, patient outcomes are expected to improve significantly. Additionally, the expansion of telemedicine services will enhance access to specialized care, particularly in rural areas. These trends indicate a robust growth trajectory for the AATD market, fostering innovation and improving patient management strategies.
Market Opportunities
- Development of New Therapeutic Agents:The ongoing research into novel therapeutic agents presents a significant opportunity for market growth. With an estimated ¥15 billion allocated for AATD research in future, pharmaceutical companies are likely to invest in developing innovative treatments that can address unmet medical needs, thereby enhancing patient care and expanding market potential.
- Expansion of Telemedicine Services:The rise of telemedicine services in Japan is creating new avenues for patient engagement and management. With over 60% of healthcare providers expected to adopt telehealth solutions in future, this trend will facilitate better access to specialists for AATD patients, particularly in underserved regions, ultimately improving treatment adherence and health outcomes.