Region:Asia
Author(s):Dev
Product Code:KRAA0649
Pages:82
Published On:January 2026
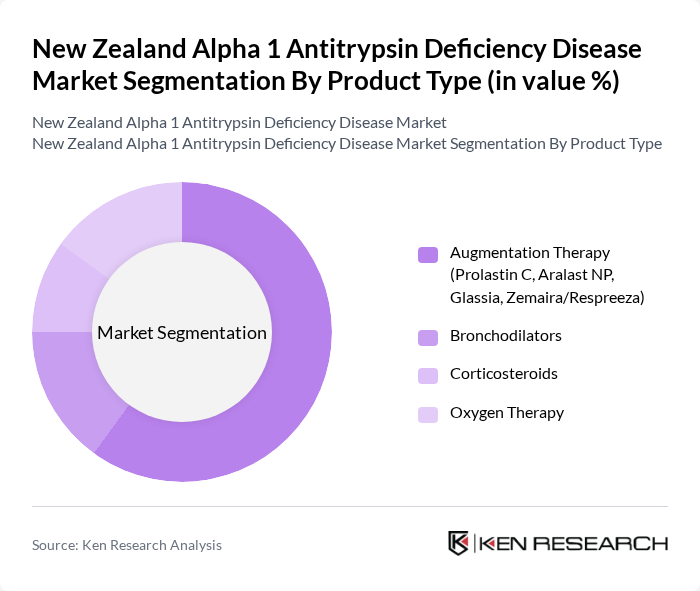
By Product Type:The product type segmentation includes various therapeutic options available for managing Alpha 1 Antitrypsin Deficiency. Among these, Augmentation Therapy is the leading sub-segment, as it directly addresses the deficiency by providing the missing protein. This therapy is crucial for patients with severe deficiency, leading to its high adoption rate. Other products like Bronchodilators, Corticosteroids, and Oxygen Therapy also play significant roles in managing symptoms and improving quality of life, but they do not replace the missing protein.
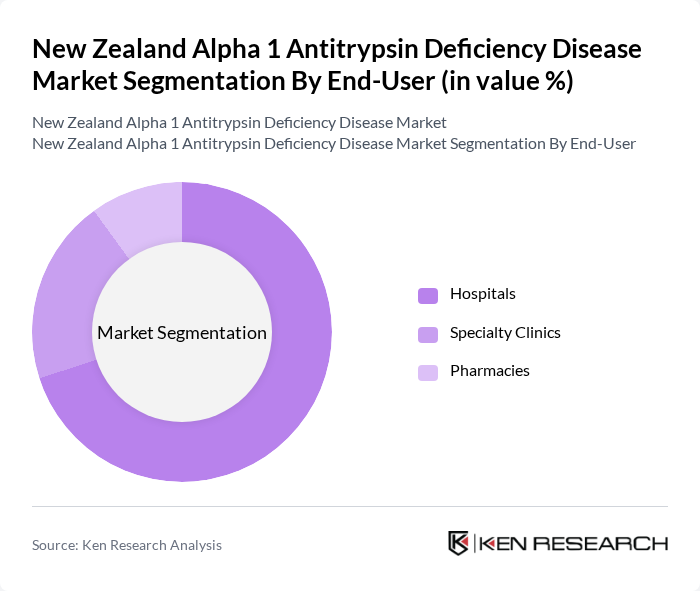
By End-User:The end-user segmentation highlights the primary settings where treatments for Alpha 1 Antitrypsin Deficiency are administered. Hospitals are the dominant end-user segment, as they provide comprehensive care and access to specialized treatments. Specialty clinics also play a vital role, offering targeted therapies and follow-up care for patients. Pharmacies contribute to the market by providing medications for symptom management, but their impact is less significant compared to hospitals and specialty clinics.
The New Zealand Alpha 1 Antitrypsin Deficiency Disease Market is characterized by a dynamic mix of regional and international players. Leading participants such as CSL Behring, Grifols, Takeda Pharmaceutical Company Limited, AstraZeneca, Kamada Pharmaceuticals, LFB Biomedicaments S.A., Pfizer Inc., Boehringer Ingelheim, Roche, Sanofi, Regeneron Pharmaceuticals, Alnylam Pharmaceuticals, Amgen, Novartis, Merck & Co. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the New Zealand Alpha 1 Antitrypsin Deficiency market appears promising, driven by ongoing advancements in treatment modalities and increased government support. As awareness continues to grow, more patients are likely to be diagnosed and treated effectively. Additionally, the integration of telemedicine and digital health solutions is expected to enhance patient engagement and access to care, particularly in underserved regions. These trends indicate a positive trajectory for patient outcomes and overall market growth in future.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Augmentation Therapy (Prolastin C, Aralast NP, Glassia, Zemaira/Respree) Bronchodilators Corticosteroids Oxygen Therapy |
| By End-User | Hospitals Specialty Clinics Pharmacies |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies |
| By Route of Administration | Injection Inhalation Oral |
| By Geographic Region | North Island South Island Urban Areas Rural Areas |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 120 | Pulmonologists, Hepatologists, General Practitioners |
| Patient Advocacy Groups | 50 | Patient Advocates, Support Group Leaders |
| Pharmaceutical Stakeholders | 75 | Market Access Managers, Medical Affairs Professionals |
| Health Insurance Representatives | 60 | Policy Analysts, Claims Managers |
| Research Institutions | 40 | Clinical Researchers, Epidemiologists |
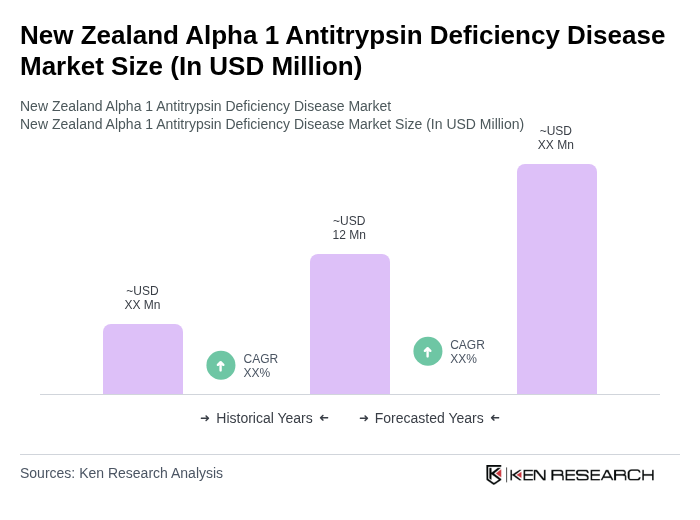
The New Zealand Alpha 1 Antitrypsin Deficiency Disease market is valued at approximately USD 12 million, reflecting growth driven by increased awareness, advancements in treatment options, and a growing patient population requiring management for respiratory conditions associated with the deficiency.